Abstract
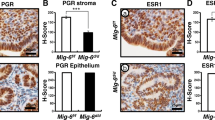
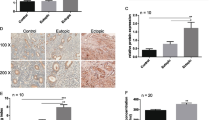
Objective: To investigate the role of phosphatase of regenerating liver-3 (PRL-3) in the 17β-estradiol (E2)- and interleukin 6 (IL-6)-induced migration of endometrial stromal cells (ESCs) from ectopic endometrium. Methods: Ectopic endometrial tissues were collected from patients with endometriosis, and PRL-3 expression in ectopic and eutopic endometrium was examined by immunohistochemistry. Endometrial stromal cells isolated from ectopic endometrium were treated with E2, progesterone (P), IL-6, or sodium orthovanadate (Sov) to inhibit PRL-3. Total RNA and protein were extracted from ESCs after treatment for quantitative real-time polymerase chain reaction and Western blot analyses. Cell migration was assessed using a scratch wound assay. Results: Phosphatase of regenerating liver 3 protein was highly expressed in the endometrial glandular cells (EGCs) and ESCs in ectopic endometrium, whereas its weak expression was observed only in EGCs in eutopic endometrium. Both E2 and IL-6 treatment significantly increased PRL-3 messenger RNA and protein expression, and P treatment significantly inhibited PRL-3 expression. However, E2-induced PRL-3 expression in ESCs from ectopic endometrium was significantly blocked by IL-6 antibody. Moreover, E2- and IL-6-enhanced cell migration was completely abrogated by Sov treatment. Furthermore, Sov treatment could significantly promote PTEN expression but inhibit E2- and IL-6-induced p-AKT activation. Conclusion: Phosphatase of regenerating liver 3 plays a key role in the E2- and IL-6-induced migration of ESCs from ectopic endometrium, a process that is involved in the PTEN-AKT signaling pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Quaas AM, Weedin EA, Hansen KR. On-label and off-label drug use in the treatment of endometriosis. Fertil Steril. 2015;103(3): 612–625.
Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3): 511–519.
Cramer DW, Missmer SA. The epidemiology of endometriosis. Ann N Y Acad Set 2002;955: 11–22; discussion 34–16, 396–406.
Gao X, Outley J, Botteman M, Spalding J, Simon JA, Pashos CL. Economic burden of endometriosis. Fertil Steril. 2006;86(6): 1561–1572.
Bulun SE. Endometriosis. N Engl J Med. 2009;360(3): 268–279.
Nezhat F, Datta MS, Hanson V, Pejovic T, Nezhat C, Nezhat C. The relationship of endometriosis and ovarian malignancy: a review. Fertil Steril. 2008;90(5): 1559–1570.
Van Gorp T, Amant F, Neven P, Vergote I, Moerman P. Endome-triosis and the development of malignant tumours of the pelvis. A review of literature. Best Pract Res Clin Obstet Gynaecol. 2004;18(2): 349–371.
Shao R, Cao S, Wang X, Feng Y, Billig H. The elusive and controversial roles of estrogen and progesterone receptors in human endometriosis. Am J Transl Res. 2014;6(2): 104–113.
Brandenberger AW, Lebovic DI, Tee MK, et al. Oestrogen receptor (ER)-alpha and ER-beta isoforms in normal endometrial and endometriosis-derived stromal cells. Mol Hum Reprod. 1999;5(7): 651–655.
Cavallini A, Resta L, Caringella AM, Dinaro E, Lippolis C, Loverro G. Involvement of estrogen receptor-related receptors in human ovarian endometriosis. Fertil Steril. 2011;96(1): 102–106.
Bergqvist A, Ferno M, Skoog L. Quantitative enzyme immunoassay and semiquantitative immunohistochemistry of oestrogen and progesterone receptors in endometriotic tissue and endometrium. J Clin Pathol. 1997;50(6): 496–500.
Nisolle M, Casanas-Roux F, Wyns C, de Menten Y, Mathieu PE, Donnez J. Immunohistochemical analysis of estrogen and proges-terone receptors in endometrium and peritoneal endometriosis: a new quantitative method. Fertil Steril. 1994;62(4): 751–759.
Zeitoun K, Takayama K, Michael MD, Bulun SE. Stimulation of aromatase P450 promoter (II) activity in endometriosis and its inhibition in endometrium are regulated by competitive binding of steroidogenic factor-1 and chicken ovalbumin upstream promoter transcription factor to the same cis-acting element. Mol Endocrinol. 1999;13(2): 239–253.
Gentilini D, Busacca M, Di Francesco S, Vignali M, Vigano P, Di Blasio AM. PI3K/Akt and ERK1/2 signalling pathways are involved in endometrial cell migration induced by 17beta-estradiol and growth factors. Mol Hum Reprod. 2007;13(5): 317–322.
Bersinger NA, Dechaud H, McKinnon B, Mueller MD. Analysis of cytokines in the peritoneal fluid of endometriosis patients as a function of the menstrual cycle stage using the Bio-Plex(R) platform. Arch Physiol Biochem. 2012;118(4): 210–218.
Agic A, Xu H, Finas D, Banz C, Diedrich K, Hornung D. Is endometriosis associated with systemic subclinical inflammation? Gynecol Obstet Invest. 2006;62(3): 139–147.
Khan KN, Kitajima M, Inoue T, Fujishita A, Nakashima M, Masuzaki H. 17beta-estradiol and lipopolysaccharide additively promote pelvic inflammation and growth of endometriosis. Reprod Sci. 2015;22(5): 585–594.
Kozlov G, Cheng J, Ziomek E, Banville D, Gehring K, Ekiel I. Structural insights into molecular function of the metastasis-associated phosphatase PRL-3. J Biol Chem. 2004;279(12): 11882–11889.
Saha S, Bardelli A, Buckhaults P, et al. A phosphatase associated with metastasis of colorectal cancer. Science. 2001;294(5545): 1343–1346.
Miskad UA, Semba S, Kato H, Yokozaki H. Expression of PRL-3 phosphatase in human gastric carcinomas: close correlation with invasion and metastasis. Pathobiology. 2004;71(4): 176–184.
Li ZR, Wang Z, Zhu BH, et al. Association of tyrosine PRL-3 phosphatase protein expression with peritoneal metastasis of gas-tric carcinoma and prognosis. Surg Today. 2007;37(8): 646–651.
Wang Z, He YL, Cai SR, et al. Expression and prognostic impact of PRL-3 in lymph node metastasis of gastric cancer: its molecular mechanism was investigated using artificial microRNA inter-ference. Int J Cancer. 2008;123(6): 1439–1447.
Ruan F, Lin J, Wu RJ, et al. Phosphatase of regenerating liver-3: a novel and promising marker in human endometriosis. Fertil Steril. 2010;94(6): 1980–1984.
Wu X, Zeng H, Zhang X, et al. Phosphatase of regenerating liver-3 promotes motility and metastasis of mouse melanoma cells. Am J Pathol. 2004;164(6): 2039–2054.
Ryan IP, Schriock ED, Taylor RN. Isolation, characterization, and comparison of human endometrial and endometriosis cells in vitro. J Clin Endocrinol Metab. 1994;78(3): 642–649.
Akoum A, lemay A, Paradis I, Rheault N, Maleux R. Secretion of interleukin-6 by human endometriotic cells and regulation by proinflammatory cytokines and sex steroid. Hum Reprod. 1996; 11(10):2269–2275.
Shain KH, Yarde DN, Meads MB, et al. Betal integrin adhesion enhances IL-6-mediated STAT3 signaling in myeloma cells: implications for microenvironment influence on tumor survival and proliferation. Cancer Res. 2009;69(3): 1009–1015.
Younis A, Hawkins K, Mahini H, Butler W, Garelnabi M. Serum tumor necrosis factor-alpha, interleukin-6, monocyte chemotactic protein-1 and paraoxonase-1 profiles in women with endometriosis, PCOS, or unexplained infertility. J Assist Reprod Genet. 2014;31(11): 1445–1451.
Govatati S, Kodati VL, Deenadayal M, Chakravarty B, Shivaji S, Bhanoori M. Mutations in the PTEN tumor gene and risk of endometriosis: a case-control study. Hum Reprod. 2014;29(2): 324–336.
Guzeloglu-Kayisli O, Kayisli UA, Al-Rejjal R, Zheng W, Luleci G, Arici A. Regulation of PTEN (phosphatase and tensin homolog deleted on chromosome 10) expression by estradiol and proges-terone in human endometrium. J Clin Endocrinol Metab. 2003;88(10): 5017–5026.
Wang H, Quah SY, Dong JM, Manser E, Tang JP, Zeng Q. PRL-3 downregulates PTEN expression and signals through PI3K to promote epithelial-mesenchymal transition. Cancer Res. 2007;67(7): 2922–2926.
Georgescu MM. PTEN Tumor Suppressor Network in PI3K-Akt Pathway Control. Genes Cancer. 2010;1(12): 1170–1177.
Kao LC, Tulac S, Lobo S, et al. Global gene profiling in human endometrium during the window of implantation. Endocrinology. 2002;143(6): 2119–2138.
Kim JJ, Kurita T, Bulun SE. Progesterone action in endometrial cancer, endometriosis, uterine fibroids, and breast cancer. Endocr Rev. 2013;34(1): 130–162.
Weiss G, Goldsmith LT, Taylor RN, Bellet D, Taylor HS. Inflammation in reproductive disorders. Reprod Sci. 2009;16(2): 216–229.
Lee SK, Han YM, Yun J, et al. Phosphatase of regenerating liver-3 promotes migration and invasion by upregulating matrix metalloproteinases-7 in human colorectal cancer cells. Int J Cancer. 2012;131(3):E190–E203.
Ruan F, Wu RJ, Xu KH, et al. Effects of PRL-3 siRNA on the migration of endometriotic stromal cells. Zhonghua Yi Xue Za Zhi. 2011;91(23): 1613–1616.
Gentilini D, Vigano P, Somigliana E, et al. Endometrial stromal cells from women with endometriosis reveal peculiar migratory behavior in response to ovarian steroids. Fertil Steril. 2010;93(3): 706–715.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, S., Zhou, Y., Fang, X. et al. PRL-3 Is Involved in Estrogen- and IL-6–Induced Migration of Endometrial Stromal Cells From Ectopic Endometrium. Reprod. Sci. 24, 124–132 (2017). https://doi.org/10.1177/1933719116650751
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116650751