Abstract
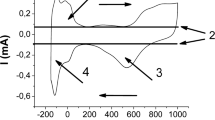
Accumulation of electrolytic hydrogen in alkaline medium (5 M KOH) by multiwall carbon nanotubes (MWCNTs) 20–60 nm in inner diameter and 2 μm in length obtained by catalytic pyrolysis of propane/butane mixture has been studied by means of the electrochemical diffusion technique, cyclic voltammetry, and impedance spectroscopy. MWCNTs were applied on a steel membrane and were encapsulated by a 10-nm electrolytic nickel layer. Cyclic voltammograms were recorded in the range of potentials from −1.2 to +0.2 V and contained a current peak in the cathode region corresponding to hydrogen absorption by nanotubes at −0.9 V and current peak in the anode region corresponding to oxidation of absorbed hydrogen at −0.6 V. Hydrogen storage capacity of MWCNTs varies from 4.6 to 6.5% depending on the amount of nanotubes according to electrochemical diffusion data. The electrochemical impedance data correlate with the results of the above methods.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Iijima, S., Nature, 1991, vol. 354, p. 56.
Fialkov, A.S., Formirovanie struktury i svoistva uglegrafitovykh materialov (Structuring and Properties of Coal-Graphite Materials), Moscow: Metallurgiya, 1965.
Fialkov, A.S., Uglegrafitovye materialy (Coal-Graphite Materials), Moscow: Energiya, 1979.
Pechkovskaya, K.A., Sazha kak usilitel’ kauchuka (The Implementation of Soot for Reinforcement of Caoutchouc), Moscow: Khimiya, 1968.
Tarasevich, Yu.G., Elektrokhimiya uglerodnykh materialov (Electrochemistry of Carbo Materials), Moscow: Nauka, 1984.
Shvartsman, A.S. and Fialkov, A.S., Zh. Prikl. Khim., 1987, vol. 60, p. 1559.
Varentsov, V.K. and Varentsova, V.I., Khim. Interesakh Ustoich. Razvit., 2000, vol. 8, no. 3, p. 353.
Varentsov, V.K. and Varentsova, V.I., Russ. J. Appl. Chem., 2000, vol. 73, no. 10, p. 1742.
Varenstov, V.K. and Varentsova, V.I., Russ. J. Electrochem., 2001, vol. 37, no. 7, p. 690.
Varenstov, V.K., Varentsova, V.I., Bataev I.A. Yusin S.I., Prot. Met. Phys. Chem. Surf., 2011, vol. 47, no. 1, p. 43.
Shibaev, D.A., Orlov, V.Yu., Bazlov, D.A., and Vaganov, V.Yu., Khim. Khim. Tekhnol., 2011, vol. 54, no. 7, p. 38.
Vigdorovich, V.I., Tsygankova, L.E., Aleksashina, E.V., and Gladysheva, I.E., Korroz.: Mater., Zashch., 2010, no. 1, p. 8.
Vigdorovich, V.I., Tsygankova, L.E., Kichigin, V.I., and Gladysheva, I.E., Prot. Met. Phys. Chem. Surf., 2012, vol. 48, p. 207.
Vigdorovich. V.I., Tsygankova, L.E., Kichigin, V.I., and Gladysheva, I.E., Prot. Met. Phys. Chem. Surf., 2012, vol. 48, p. 438.
Tsygankova, L.E., Aleksashina, E.V., Gladysheva, I.E., and Vigdorovich, V.I., Kondens. Sredy Mezhfaznye Granitsy, 2009, vol. 11, no. 3, p. 249.
Bannov, A.G., Varentsov, V.K., Chukanov, I.S., Gorodilova, E.V., and Kuvshinov, G.G., Prot. Met. Phys. Chem. Surf., 2012, vol. 48, no. 2, p. 199.
Pan, W., Zhang, X., Li, S., et al., Int. J. Hydrogen Energy, 2005, vol. 30, p. 719.
Zhou, L., Zhou, Y., and Sun, Y., Int. J. Hydrogen Energy, 2006, vol. 31, p. 259.
Dillon, A.C., Jones, K.M., Bekkedahl, T.A., et al., Nature, 1997, vol. 386, p. 377.
Ye, Y., Ahn, C.C., Witham, C., et al., Appl. Phys. Lett., 1999, vol. 74, p. 2307.
Liu, C., Fan, Y.Y., Liu, M., et al., Science, 1999, vol. 286, p. 1127.
Darkrim, F.L., Malbrunot, P., and Tartaglia, G.P., Int. J. Hydrogen Energy, 2002, vol. 27, p. 193.
Nutzenadel, C., Zuttel, A., Chartouni, D., and Schlapbach, L., Electrochem. Solid-State Lett., 1999, vol. 2, p. 30.
Vix-Guterl, C., Frackowiak, E., Jurewicz, K., et al., Carbon, 2005, vol. 43, p. 1293.
Zhang, H., Fu, X., Chen, Y., et al., Physica B, 2004, vol. 352, p. 66.
Chen, X., Zhang, Y., Gao, X.P., et al., Int. J. Hydrogen Energy, 2004, vol. 29, p. 743.
Qin, X., Gao, X.P., Liu, H., et al., Electrochem. Solid- State Lett., 2000, vol. 3, p. 532.
Rajalakshmi, N., Dhathathreyan, K.S., Govindaraj, A., and Satishkumar, B.C., Electrochim. Acta, 2000, vol. 45, p. 4511.
Fazle Kibria, A.K.M., Mo, Y.H., Park, K.S., et al., Int. J. Hydrogen Energy, 2001, vol. 26, p. 823.
Gundiah, G., Govindaraj, A., Rajalakshmi, N., et al., J. Mater. Chem., 2003, vol. 13, p. 209.
Lee, S.M., Park, K.S., Choi, Y.C., et al., Synth. Met., 2000, vol. 113, p. 209.
Zuttel, A., Sudan, P., Mauron, P., et al., Int. J. Hydrogen Energy, 2002, vol. 27, p. 203.
Jones, C.P., Jurkschat, K., Crossley, A., et al., Langmuir, 2007, vol. 23, p. 9501.
Niessen, R.A.H., de Longe, J., and Nottena, P.H.L., J. Electrochem. Soc., 2006, vol. 153, p. A1484.
Solodkova, L.N., Lyakhov, B.F., Lipson, A.G., and Tsivadze, A.Yu., Prot. Met. Phys. Chem. Surf., 2010, vol. 46, no. 5, p. 524.
Tkachev, A.G., Perspekt. Mater., 2007, no. 3, p. 5.
Devanathan, M.A.V. and Stachurski, Z., Zashch. Met., 1995, vol. 31, p. 441.
Tsygankova, L.E., Vigdorovich, V.I., and Zvereva, A.A., Prot. Met. Phys. Chem. Surf., 2013, vol. 49, p. 669.
Lim, C. and Pyun, S.-I., Electrochim. Acta, 1993, vol. 38, p. 2645.
Lasia, A., in Modern Aspects of Electrochemistry, Conway, B. and White, R., Eds., New York: Kluwer, 2002, vol. 35, p. 1.
Gabrielli, C., Grand, P.P., Lasia, A., and Perrot, H., J. Electrochem. Soc., 2004, vol. 151, p. A1925.
Bockris, J.O.M., McBreen, J., and Nanis, L., J. Electrochem. Soc., 1965, vol. 112, p. 1025.
Harrington, D.A. and Conway, B.E., Electrochim. Acta, 1987, vol. 32, p. 1703.
Dull, D.L. and Nobe Ken, Corrosion, 1979, vol. 35, p. 535.
Saito, Y. and Nobe Ken, Corrosion, 1980, vol. 36, p. 178.
Zakroczymski, T., Scr. Mater., 1985, vol. 19, p. 521.
Diard, J.-P., J. Electroanal. Chem., 2003, vol. 557, p. 19.
Bóbics, L., Sziráki, L., and Láng, G.G., Electrochem. Comm., 2008, vol. 10, p. 283.
McNabb, A. and Foster, P.K., Trans. Metall. Soc. AIME, 1963, vol. 227, p. 618.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.E. Tsygankova, V.I. Vigdorovich, A.A. Zvereva, V.I. Kichigin, 2016, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2016, Vol. 52, No. 2, pp. 142–149.
Rights and permissions
About this article
Cite this article
Tsygankova, L.E., Vigdorovich, V.I., Zvereva, A.A. et al. A Study of Hydrogen Accumulation in Multiwall Carbon Nanotubes by Electrochemical Techniques. Prot Met Phys Chem Surf 52, 211–217 (2016). https://doi.org/10.1134/S2070205116020301
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205116020301