Abstract
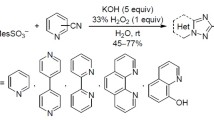
Treatment of N-aryl-8-hydrazinylpyrano[3,4-c]pyridine-5-carbonitriles with sodium nitrite in acetic acid afforded the corresponding 8-azido derivatives existing in solution as equilibrium mixtures with cyclic tetrazole tautomer whose fraction attains 48%. Lower solvent polarity and elevated temperature favor increased fraction of the azido tautomer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kumar, R.R., Perumol, S., Menandes, J.C., Yugeeswari, P., and Sriram, D., Bioorg. Med. Chem., 2011, vol. 19, p. 3444.
Abdel-Wadood, F.K., Abdel-Monem, M.I., Fahamy, A.M., and Geies, A.A., Z. Naturforsch., Teil B, 2008, vol. 63, p. 303.
Lee, S., Chae, S.M., Yi, K.Y., Kim, N., and Oh, Ch.H., Bull. Korean Chem. Soc., 2005, vol. 26, p. 619.
Kwak, W.J., Kim, J.H., Cho, Y.B., Jeon, S.D., and Moon, C.K., Biol. Pharm. Bull., 2005, vol. 28, p. 750.
Opperman, T.J., Kwasny, S.M., Kim, H.S., Nguyen, S.T., Houseweart, C., D’Souza, S., Walker, G.C., Peet, N.P., Nikaido, H., and Bowlin, T.L., Antimicrob. Agents Chemother., 2014, vol. 58, p. 722.
Nguyen, S.T., Kwasny, S.M., Ding, X., Cardinale, S.C., McCarthy, C.T., Kim, H.S., Nikaido, H., Peet, N.P., Williams, J.D., Bowlin, T.L., and Opperman, T.J., Bioorg. Med. Chem., 2015, vol. 23, p. 2024.
Panda, B. and Sarkar, T.K., Tetrahedron Lett., 2008, vol. 49, p. 6701.
Subba Reddy, B.V., Reddy, S.G., Reddy, M.R., Spidhar, B., and Bhadra, M.P., Tetrahedron Lett., 2014, vol. 55, p. 4817.
Lowe-Ma, Ch.K., Nissan, R.A., and Wilson, W.S., J. Org. Chem., 1990, vol. 55, p. 3755.
Keith, J.M., J. Org. Chem., 2006, vol. 71, p. 9540.
Liu, Sh., Lentz, D., and Tzschucke, C.Ch., J. Org. Chem., 2014, vol. 79, p. 3249.
Paronikyan, E.G., Dashyan, Sh.Sh., Noravyan, A.S., Tamazyan, R.A., Ayvazyan, A.G., and Panosyan, H.A., Tetrahedron, 2015, vol. 71, p. 2686.
Paronikyan, E.G., Dashyan, Sh.Sh., Dzhagatspanyan, I.A., Paronikyan, R.G., Nazaryan, I.M., Akopyan, A.G., Minasyan, N.S., Ayvazyan, A.G., Tamazyan, R.A., and Babaev, E.V., Russ. J. Bioorg. Chem., 2016, vol. 42, no. 2, p. 215.
Paronikyan, E.G., Sirakanyan, S.N., Noravyan, A.S., Paronikyan, R.G., and Dzhagatspanyan, I.A., Pharm. Chem. J., 2001, vol. 35, no. 1, p. 8.
Paronikyan, E.G., Noravyan, A.S., Dzhagatspanyan, I.A., Nazaryan, I.M., and Paronikyan, R.G., Pharm. Chem. J., 2002, vol. 36, no. 9, p. 465.
Dashyan, Sh.Sh., Paronikyan, E.G., Noravyan, A.S., and Paronikyan, R.G., Pharm. Chem. J., 2016, vol. 50, no. 4, p. 221.
Paronikyan, E.G., Dashyan, Sh.Sh., Minasyan, N.S., Stepanyan, G.M., Ayvazyan, A.G., and Tamazyan, R.A., Chem. Heterocycl. Compd., 2016, vol. 52, no. 12, p. 1078.
Bellamy, L.J., Advances in Infrared Group Frequencies, London: Methuen, 1968, p. 70.
Pochinok, V.Y., Avramenko, L.F., Grigorenko, P.S., and Skopenko, V.N., Russ. Chem. Rev., 1975, vol. 44, no. 6, p. 481.
Chattopadhyay, B., Vera, C.I.R., Chuprakov, S., and Gevorgyan, V., Org. Lett., 2010, vol. 12, p. 2166.
Chan, J. and Faul, M., Tetrahedron Lett., 2006, vol. 47, p. 3361.
Temple, C. and Montgomery, J.A., J. Am. Chem. Soc., 1964, vol. 86, p. 2946.
Temple, C. and Montgomery, J.A., J. Org. Chem., 1965, vol. 30, p. 826.
Lioux, Th., Gosselin, G., and Mathe, Ch., Eur. J. Org. Chem., 2003, p. 3997.
Mironov, A.N., Rukovodstvo po provedeniyu doklinicheskikh issledovanii lekarstvennykh sredstv (A Guide to Preclinical Trials of Medicines), Moscow Meditsina, 2012.
Mashkovskii, M.D., Lekarstvennye sredstva (Medicines), Moscow: Novaya Volna, 2010, 16th ed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.G. Paronikyan, Sh.Sh. Dashyan, N.S. Minasyan, G.M. Stepanyan, 2017, published in Zhurnal Organicheskoi Khimii, 2017, Vol. 53, No. 6, pp. 925–929.
Rights and permissions
About this article
Cite this article
Paronikyan, E.G., Dashyan, S.S., Minasyan, N.S. et al. Azido–tetrazole tautomerism of pyrano[3,4-c]pyridine derivatives. Russ J Org Chem 53, 941–945 (2017). https://doi.org/10.1134/S1070428017060215
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428017060215