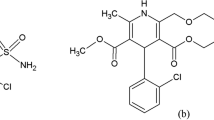
Abstract
An accurate, sensitive and precise HPLC method was developed and validated for the routine analysis of pioglitazone hydrochloride in dosage forms. The analyte was chromatographed on a C-18 column using a mixture of acetonitrile and ammonium acetate buffer (pH 5.0) as mobile phase in ratio 60: 40 (v/v) at flow rate 1.0 mL/min. Linearity range was found to be 10–100 μg/mL with a correlation coefficient r 2 = 0.9984. The method was validated for precision, accuracy, robustness, specificity and sensitivity, using bulk drug samples. Application of method in assay of bulk drug and tablets revealed mean recoveries range from 99.88–100.32%. Due to its simplicity, rapidity, high precision and accuracy, the proposed method may be used for determining pioglitazone hydrochloride in bulk and dosage forms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Prigeon, R.L., Kahn, S.E., and Porte, J.D., J. Clin. Endocrinol. Metab., 1998, vol. 83, p. 819.
Cavaghan, M.K., Ehrmann, D.A., Byrne, M.M., and Polonsky, K.S., J. Clin. Invest., 1997, vol. 100, p. 530.
Sevgi, T.U. and Fikriye, T.E., Anal. Lett., 2009, vol. 42, p. 2254.
Shankar, M.B., Modi, V.D., Shah, D.A., Bhatt, K.K., Mehta, R.S., and Geetha, M., J. AOAC Int., 2005, vol. 88, no. 4, p. 1167.
Radhakrishna, T., Sreenivas, R.D., and Om, R.G., J. Pharm. Biomed. Anal., 2002, vol. 29, p. 593.
Zhong, W.Z. and Williams, M.G., J. Pharm. Biomed. Anal., 1996, vol. 14, no. 4, p. 465.
Ho, E.N., Yiu, K.C., Wan, T.S., Stewart, B.D., and Watkins, K.L., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2004, vol. 811, no. 1, p. 65.
Lin, Z.J., Ji, W., Desai, K.D., and Shum, L., J. Pharm. Biomed. Anal., 2003, vol. 33, no. 1, p. 101.
Wang, M. and Miksa, I.R., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2007, vol. 856, no. 1, p. 318.
Yamashita, K., Murakami, H., Okuda, T., and Motohashi, M., J. Chromatogr. B: Biomed. Sci. Appl., 1996, vol. 677, no. 1, p. 141.
Souri, E., Jalalizadeh, H., and Saremi, S., J. Chromatogr. Sci., 2008, vol. 46, no. 9, p. 809.
Zhong, W.Z. and Lakings, D.B., J. Chromatogr., 1989, vol. 490, no. 2, p. 377.
Sripalakit, P. and Neamhom, P., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2006, vol. 843, no. 2, p. 164.
Kolte, B.L., Raut, B.B., Deo, A.A., Bagool, M.A., and Shinde, D.B., J. Chromatogr. Sci., 2004, vol. 42, p. 27.
Jain, D., Jain, S., Jain, D., and Amin, M., J. Chromatogr. Sci., 2008, vol. 46, p. 501.
Mostafa, G.A. and Majed, A., J. Pharm. Biomed. Anal., 2008, vol. 48, no. 1, p. 57.
ICH. Validation of Analytical Procedure; Methodology, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceutical for Human Use, Geneva, 2000.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Prasad, P.S., Imam, S.S., Aqil, M. et al. Validated reversed phase HPLC method for determination of pioglitazone hydrochloride in bulk drug and tablet formulations. J Anal Chem 70, 744–746 (2015). https://doi.org/10.1134/S106193481506012X
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106193481506012X