Abstract
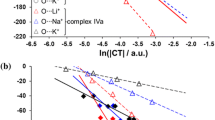
The optimal geometry and wave functions of the complexes [M(Solv)]+ (M = Li, Na, K; Solv is an aprotic solvent molecule) were calculated and the topological characteristics of the electron density distribution at the (3,–1) critical points (CP) of ion–molecule bonds were analyzed by the density functional theory in the B3LYP/6-31+G(d, p) approximation. The parametric dependences for the energy of ion–molecule bonds in terms of the local kinetic and potential electron energy densities at the bond CTs were proposed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. D. Berezin, in Solution Thermodynamics and Structure (Ivanovo, 1973), Iss. 1, p. 5 [in Russian].
R. F. W. Bader, Atoms in Molecules. A Quantum Theory (Clarendon Press, Oxford, 1990).
E. Espinosa, E. Molins, and C. Lecomte, Chem. Phys. Lett. 285, 170 (1998).
E. Espinosa, I. Alkorta, and I. Rozas, Chem. Phys. Lett. 336, 457 (2001).
I. Mata, E. Molins, I. Alkorta, and E. Espinosa, J. Chem. Phys. 130, 044104 (2009).
R. Parthasarathi, V. Subramanian, and N. Sathyamurthy, J. Phys. Chem. A 110, 3349 (2006).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., GAUSSIAN 09, Revision A.1, Gaussian, Inc., Wallingford, CT, 2009.
T. A. Keith, AIMAll (Version 10.05.04). http://aim. tkgristmill.com5.
A. Zalewska, I. Pruszczyk, E. Sulek, and W. Wieczorek, Solid State Ionics 157, 233 (2003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.P. Mikhailov, 2018, published in Zhurnal Neorganicheskoi Khimii, 2018, Vol. 63, No. 2, pp. 186–189.
Rights and permissions
About this article
Cite this article
Mikhailov, G.P. The Relationship of the Energy of Interaction of Alkali Metal Cations with an Aprotic Solvent Molecule with Quantum Topological Electron Density Characteristics. Russ. J. Inorg. Chem. 63, 197–200 (2018). https://doi.org/10.1134/S0036023618020158
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023618020158