Abstract
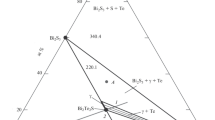
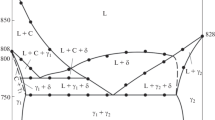
Phase relations in the Bi2O3–SnO2 system have been studied using differential thermal analysis, differential scanning calorimetry, X-ray diffraction, and the sessile drop method. The C p (T) curve obtained in the range 350–960 K has a number of extrema, which are attributable to structural changes in Bi2Sn2O7. The experimental C p data have been used to evaluate the thermodynamic properties of the oxide compound.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kargin, Yu.F., Nelyapina, N.I., and Skorikov, V.M., System Bi2O3–SnO2, in Fiziko-khimicheskie issledovaniya ravnovesii v rastvorakh (Physicochemical Studies of Equilibria in Solutions), Yaroslavl: YaGRI im. K.D. Ushinskogo, 1988, pp. 81–83.
Mao, Y., Li, G., Xu, W., et al., Hydrothermal synthesis and characterization of nanocrystalline pyrochlore oxides M2Sn2O7 (M = La, Bi, Gd or Y), J. Mater. Chem., 2000, vol. 10, pp. 479–482.
Shannon, R.D., Bierlein, J.D., Gillson, J.L., et al., Polymorphism in Bi2Sn2O7, J. Phys. Chem. Solids, 1980, vol. 41, pp. 117–122.
Jones, R.H. and Knight, K.S., The structure of γ-Bi2Sn2O7 at 725 by high-resolution neutron diffraction: implications for bismuth(III)-containing pyrochlores, J. Chem. Soc., Dalton Trans., 1977, pp. 2551–2555.
Moens, L., Ruiz, P., Delmon, B., et al., Cooperation effects towards partial oxidation of isobutene in multiphasic catalysts based on bismuth pyrostannate, Appl. Catal., A, 1998, vol. 171, pp. 131–143.
Mims, C.A., Jacobson, A.J., Hall, R.B., et al., Methane oxidative coupling over nonstoichiometric bismuth–tin pyrochlore catalysts, J. Catal., 1995, vol. 153, pp. 197–207.
Devi, G.S., Manorama, S.V., and Rao, V.J., SnO2/Bi2O3: a suitable system for selective carbon monoxide detection, J. Electrochem. Soc., 1988, vol. 145, no. 3, pp. 1039–1044.
Diagrammy sostoyaniya sistem tugoplavkikh oksidov: Spravochnik (Phase Diagrams of Refractory Oxide Systems: A Handbook), Dvoinye sistemy (Binary Systems), Galakhov, F.Ya., Ed., Leningrad: Nauka, 1986, part 2, pp. 298–299.
Asryan, N.A., Kol’tsova, T.N., Alikhanyan, A.S., and Nipan, G.D., Thermodynamics and phase diagram of the Bi2O3–SnO2 system, Inorg. Mater., 2002, vol. 38, no. 11, pp. 1141–1147.
Asryan, N.A., Kol’tsova, T.N., Alikhanyan, A.S., and Nipan, G.D., Phase equilibria in the Bi2O3–SnO2 system, Russ. J. Phys. Chem. A, 2003, vol. 77, no. 11, pp. 1740–1744.
Denisov, V.M., Kuchumova, O.V., Denisova, L.T., et al., Contact interaction of PbO–GeO2 melts with silver, Rasplavy, 2010, no. 1, pp. 3–8.
Roth, R.S., Pyrochlore-type compounds containing double oxides of trivalent and tetravalent ions, J. Res. Natl. Bur. Stand., 1956, vol. 56, pp. 17–25.
Vetter, G., Queyroux, F., and Gilles, J.-C., Preparation, stabilite et etude cristallographique preliminaire du compose Bi2Sn2O7, Mater. Res. Bull., 1978, vol. 13, pp. 211–216.
Udod, L.V., Aplesnin, S.S., Sitnikov, M.N., and Molokoev, M.S., Dielectric and electrical properties of polymorphic bismuth pyrostannate Bi2Sn2O7, Phys. Solid State, 2014, vol. 56, no. 7, pp. 1315–1319.
Kahlenberg, V. and Zeiske, Th., Structure of γ-Bi2Sn2O7 by high-temperature powder neutron diffraction, Z. Kristallogr., 1997, vol. 212, no. 4, pp. 297–301.
Walsh, A. and Watson, G.W., Polymorphism in bismuth stannate: a first-principles study, Chem. Mater., 2007, vol. 19, pp. 5158–5164.
Evans, I.R., Howard, J.A.K., and Evans, S.O., α-Bi2Sn2O7—a 176 atom crystal structure from powder diffraction, J. Mater. Chem., 2003, vol. 13, pp. 2098–2103.
Belousova, N.V. and Arkhipova, E.O., Thermodynamic properties of bismuth pyrostannate, Polzunovsk. Vestn., 2009, no. 3, pp. 56–59.
Roy, M., Bala, I., and Barbar, S.K., Synthesis, structural, electrical and thermal properties of Ti-doped Bi2Sn2O7, J. Therm. Anal. Calorim., 2012, vol. 110, pp. 559–565.
Kargin, Yu.F., Burkov, V.I., Mar’in, A.A., et al., Kristally Bi12MxO20 ± δ so strukturoi sillenita. Sintez, stroenie, svoistva (Sillenite-Structure Bi12MxO20 ± δ Crystals: Synthesis, Structure, and Properties), Moscow: Inst. Obshchei i Neorgsnicheskoi Khimii Ross. Akad. Nauk, 2004.
Tret’yakov, Yu.D. and Putlyaev, V.I., Vvedenie v khimiyu tverdofaznykh materialov (Introduction to the Chemistry of Solid Materials), Moscow: Mosk. Gos. Univ., Nauka, 2006.
Tret’yakov, Yu.D., Tverdofaznye reaktsii (Solid-State Reactions), Moscow: Khimiya, 1978.
Solovyov, L.A., Full-profile refinement by derivative difference minimization, J. Appl. Crystallogr., 2004, vol. 37, pp. 743–749.
Denisov, V.M., Denisova, L.T., Irtyugo, L.A., and Biront, V.S., Thermal physical properties of Bi4Ge3O12 single crystals, Phys. Solid State, 2010, vol. 52, no. 7, pp. 1362–1365.
Denisova, L.T., Kargin, Yu.F., Chumilina, L.G., et al., High-temperature heat capacity of Sc2Cu2O5, Inorg. Mater., 2014, vol. 50, no. 5, pp. 482–484.
Denisova, L.T., Krinitsyn, D.O., Denisov, V.M., et al., Wetting of the noble metals by Bi2O3–CaO melts, Rasplavy, 2014, no. 5, pp. 3–6.
Vonsovskii, S.V., Magnetizm (Magnetism), Moscow: Nauka, 1971.
Potashinskii, A.Z. and Pokrovskii, V.L., Fluktuatsionnaya teoriya fazovykh perekhodov (Fluctuation Theory of Phase Transitions), Moscow: Nauka, 1982.
Gusev, A.I., Nestekhiometriya, besporyadok, blizhnii i dal’nii poryadok v tverdom tele (Nonstoichiometry, Disorder, and Short- and Long-Range Order in Solids), Moscow: Fizmatlit, 2007.
Strukov, B.A. and Levanyuk, A.P., Fizicheskie osnovy segnetoelektricheskikh yavlenii v kristallakh (Physical Principles of Ferroelectricity in Crystals), Moscow: Fizmatlit, 1983.
Bush, A.A. and Popova, E.A., Heat capacity of the Pb5(Ge1 − xSix)3O11 ferroelectric system, Phys. Solid State, 2004, vol. 46, no. 5, pp. 902–907.
Holland, T.J.B. and Powell, R., An enlarged and updated internally consistent thermodynamic dataset with uncertainties and correlations: the system K2O–FeO–Fe2O3–Al2O3–TiO2–SiO2–C–H2–O2, J. Metamorph. Geol., 1990, vol. 8, pp. 89–124.
Chudnenko, K.V., Termodinamicheskoe modelirovanie v geokhimii: teoriya, algoritmy, programmnoe obespechenie, prilozheniya (Thermodynamic Modeling in Geochemistry: Theory, Algorithms, Software, and Applications), Novosibirsk: Geo, 2010.
Walsh, A., Watson, G.W., Payne, D.J., et al., A theoretical and experimental study of the distorted pyrochlore Bi2Sn2O7, J. Mater. Chem., 2006, vol. 16, pp. 3452–3458.
Naidich, Yu.V., Perevertailo, V.M., and Obushchak, L.P., Properties of solid–liquid interfaces in binary alloy crystallization processes, in Adgeziya rasplavov (Melt Adhesion), Kiev: Naukova Dumka, 1974, pp. 27–32.
Naidich, Yu.V., Perevertailo, V.M., Lebovich, E.M., et al., Interfacial and capillary phenomena in unary crystal–melt systems, in Adgeziya rasplavov (Melt Adhesion), Kiev: Naukova Dumka, 1974, pp. 3–7.
Summ, B.D. and Goryunov, Yu.V., Fiziko-khimicheskie osnovy smachivaniya i rastekaniya (Physicochemical Principles of Wetting and Spreading), Moscow: Khimiya, 1976.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.T. Denisova, Yu.F. Kargin, L.A. Irtyugo, V.M. Denisov, 2015, published in Neorganicheskie Materialy, 2015, Vol. 51, No. 7, pp. 714–718.
Rights and permissions
About this article
Cite this article
Denisova, L.T., Kargin, Y.F., Irtyugo, L.A. et al. Phase relations in the Bi2O3-SnO2 system and thermodynamic properties of bismuth pyrostannate. Inorg Mater 51, 650–654 (2015). https://doi.org/10.1134/S0020168515070043
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168515070043