Abstract
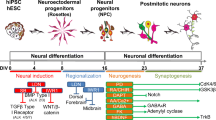
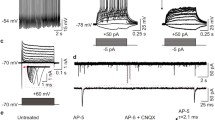
Ionotropic glutamate and GABA receptors regulate the differentiation and determine the functional properties of mature neurons. Both insufficient and excessive activity of these neurotransmission systems are associated with various nervous system diseases. Our knowledge regarding the expression profiles of these receptors and the mechanisms of their regulation during the differentiation of specialized human neuron subtypes is limited. Here the expression profiles of the NMDA and GABAA receptor subunits were explored during in vitro differentiation of human induced pluripotent stem cells (iPSCs) into ventral mesencephalic neurons. The correlation between the neuronal maturation and the expression dynamics of these genes was investigated, and the functional activity of these receptors was assessed by calcium imaging. The role of NMDA and GABAA receptors in neurite outgrowth and the development of spontaneous activity was analyzed using the viral transduction of neural progenitors with the reporter genes TagGFP and TagRFP. The data indicate that agonists of the investigated receptors can be employed for optimization of existing protocols for neural differentiation of iPSCs, in particular for acceleration of neuronal maturation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BDNF:
-
brain derived nerve growth factor
- (c)NPs:
-
(committed) neural progenitors
- eNPs:
-
early neural progenitors
- ESCs:
-
embryonic stem cells
- GABA:
-
α-aminobutyric acid
- GABAA-R:
-
GABAA receptors
- GDNF:
-
glial cell derived nerve growth factor
- iPSCs:
-
induced pluripotent stem cells
- NMDA:
-
N-methyl-D-aspartic acid
- NMDA-R:
-
NMDA receptors
- RT-PCR:
-
reverse transcription polymerase chain reaction
- TD:
-
terminal differentiation
- VM neurons:
-
ventral mesencephalic neurons
References
Takahashi, K., Tanabe, K., Ohnuki, M., Narita, M., Ichisaka, T., Tomoda, K., and Yamanaka, S. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors, Cell, 131, 861–872.
Novosadova, E. V., and Grivennikov, I. A. (2014) Induced pluripotent stem cells: from derivation to application in biochemical and biomedical research, Biochemistry (Moscow), 79, 1425–1441.
Schaarschmidt, G., Schewtschik, S., Kraft, R., Wegner, F., Eilers, J., Schwarz, J., and Schmidt, H. (2009) A new cul–turing strategy improves functional neuronal development of human neural progenitor cells, J. Neurochem., 109, 238–247.
Zhou, Q., and Sheng, M. (2013) NMDA receptors in nerv–ous system diseases, Neuropharmacology, 74, 69–75.
Koutsilieri, E., and Riederer, P. (2007) Excitotoxicity and new antiglutamatergic strategies in Parkinson’s disease and Alzheimer’s disease, Parkinsonism Relat. Disord., 13, S329–S331.
Dong, X. X., Wang, Y., and Qin, Z. H. (2009) Molecular mechanisms of excitotoxicity and their relevance to patho–genesis of neurodegenerative diseases, Acta Pharmacol. Sin., 30, 379–387.
Rowley, N. M., Madsen, K. K., Schousboe, A., and Steve White, H. (2012) Glutamate and GABA synthesis, release, transport and metabolism as targets for seizure control, Neurochem. Int., 61, 546–558.
Nacher, J., and McEwen, B. S. (2006) The role of N–methyl–D–asparate receptors in neurogenesis, Hippocampus, 16, 267–270.
Galanopoulou, A. S. (2008) GABAA receptors in normal development and seizures: friends or foes? Curr. Neuropharmacol., 6, 1–20.
Young, A., Machacek, D. W., Dhara, S. K., Macleish, P. R., Benveniste, M., Dodla, M. C., Sturkie, C. D., and Stice, S. L. (2011) Ion channels and ionotropic receptors in human embryonic stem cell derived neural progenitors, Neuroscience, 192, 793–805.
Zhang, W. B., Ross, P. J., Tu, Y., Wang, Y., Beggs, S., Sengar, A. S., Ellis, J., and Salter, M. W. (2016) Fyn kinase regulates GluN2B subunit–dominant NMDA receptors in human induced pluripotent stem cell–derived neurons, Sci. Rep., 6, 23837.
Ishii, M. N., Yamamoto, K., Shoji, M., Asami, A., and Kawamata, Y. (2017) Human induced pluripotent stem cell (hiPSC)–derived neurons respond to convulsant drugs when co–cultured with hiPSC–derived astrocytes, Toxicology, 389, 130–138.
Nekrasov, E. D., Lebedeva, O. S., Chestkov, I. V., Syusina, M. A., Fedotova, E. Yu., Lagar’kova, M. A., Kiselev, S. L., Grivennikov, I. A., and Illarioshkin, S. N. (2011) Obtain–ing and characterization of induced pluripotent stem cells from human skin fibroblasts of patients with neurodege–nerative diseases, Klet. Transplant. Tkan. Inzhener., 6, 1–7.
Lebedeva, O. S., Novosadova, E. V., Manuilova, E. S., Arsen’eva, E. L., Kiselev, S. L., Lagar’kova, M. A., Khaspekov, L. G., Illarioshkin, S. N., and Grivennikov, I. A. (2014) Obtaining and characterization of the induced pluripotent stem cell–based model of Parkinson disease, in Stem Cells and Regenerative Medicine (Tkachuk, V. A., ed.) [in Russian], Moscow University Publishers, Moscow, pp. 154–168.
Antonov, S. A., Novosadova, E. V., Arsenyeva, E. L., Grefenstein, M. A., Zykova, A. A., Kobylyansky, A. G., Manuilova, E. S., Grivennikov, I. A., Illarioshkin, S. N., and Myasoedov, N. F. (2016) Investigation of the effects of GABA receptor agonists in the differentiation of human induced pluripotent stem cells into dopaminergic neurons, Dokl. Biol. Sci., 470, 244–246.
Livak, K. J., and Schmittgen, T. D. (2001) Analysis of rela–tive gene expression data using real–time quantitative PCR and the 2–ΔΔCT method, Methods, 25, 402–408.
Zhao, H., and Darzynkiewicz, Z. (2017) Rapid detection of DNA strand breaks in apoptotic cells by flow–and image–cytometry, Methods Mol. Biol., 1644, 139–149.
Verpelli, C., Carlessi, L., Bechi, G., Fusar Poli, E., Orellana, D., Heise, C., Franceschetti, S., Mantegazza, R., Mantegazza, M., Delia, D., and Sala, C. (2013) Comparative neuronal differentiation of self–renewing neu–ral progenitor cell lines obtained from human induced pluripotent stem cells, Front. Cell. Neurosci., 7, 1–14.
Liu, X. B., Murray, K. D., and Jones, E. G. (2004) Switching of NMDA receptor 2A and 2B subunits at thala–mic and cortical synapses during early postnatal develop–ment, J. Neurosci., 24, 8885–8895.
Elkabetz, Y., Panagiotakos, G., Al Shamy, G., Socci, N. D., Tabar, V., and Studer, L. (2008) Human ES cell–derived neural rosettes reveal a functionally distinct early neural stem cell stage, Genes Dev., 22, 152–165.
Dingledine, R., Borges, K., Bowie, D., and Traynelis, S. F. (1999) The glutamate receptor ion channels, Pharmacol. Rev., 51, 7–61.
Ben–Ari, Y. (2002) Excitatory actions of gaba during devel–opment: the nature of the nurture, Nat. Rev. Neurosci., 3, 728–739.
Herlenius, E., and Lagercrantz, H. (2010) Neurotransmit–ters and neuromodulators during brain development, in The Newborn Brain: Neuroscience and Clinical Applications (Lagercrantz, H., ed.) 2nd Edn., Cambridge University Press, pp. 99–119.
Rogawski, M. A. (2011) Revisiting AMPA receptors as an antiepileptic drug target, Epilepsy Curr., 11, 56–63.
Gupta, K., Hardingham, G. E., and Chandran, S. (2013) NMDA receptor–dependent glutamate excitotoxicity in human embryonic stem cell–derived neurons, Neurosci. Lett., 543, 95–100.
Bergey, G. K., Fitzgerald, S. C., Schrier, B. K., and Nelson, P. G. (1981) Neuronal maturation in mammalian cell culture is dependent on spontaneous electrical activity, Brain Res., 207, 49–58.
Lam, R. S., Topfer, F. M., Wood, P. G., Busskamp, V., and Bamberg, E. (2017) Functional maturation of human stem cell–derived neurons in long–term cultures, PLoS One, 12, e0169506.
Gunhanlar, N., Shpak, G., van der Kroeg, M., Gouty–Colomer, L. A., Munshi, S. T., Lendemeijer, B., Ghazvini, M., Dupont, C., Hoogendijk, W. J. G., Gribnau, J., de Vrij, F. M. S., and Kushner, S. A. (2017) A simplified protocol for differentiation of electrophysiologically mature neu–ronal networks from human induced pluripotent stem cells, Mol. Psychiatry, 23, 1336–1344.
Hirasawa, T., Wada, H., Kohsaka, S., and Uchino, S. (2003) Inhibition of NMDA receptors induces delayed neuronal maturation and sustained proliferation of progen–itor cells during neocortical development, J. Neurosci. Res., 74, 676–687.
Yamasaki, M., Okada, R., Takasaki, C., Toki, S., Fukaya, M., Natsume, R., Sakimura, K., Mishina, M., Shirakawa, T., and Watanabe, M. (2014) Opposing role of NMDA receptor GluN2B and GluN2D in somatosensory develop–ment and maturation, J. Neurosci., 34, 11534–11548.
Sernagor, E., Chabrol, F., Bony, G., and Cancedda, L. (2010) GABAergic control of neurite outgrowth and remodeling during development and adult neurogenesis: general rules and differences in diverse systems, Front. Cell Neurosci., 4, 11.
Rushton, D. J., Mattis, V. B., Svendsen, C. N., Allen, N. D., and Kemp, P. J. (2013) Stimulation of GABA–induced Ca2+ influx enhances maturation of human induced pluripotent stem cell–derived neurons, PLoS One, 8, e81031.
Paoletti, P., Bellone, C., and Zhou, Q. (2013) NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease, Nat. Rev. Neurosci., 14, 383–400.
Hartfield, E. M., Yamasaki–Mann, M., Ribeiro Fernandes, H. J., Vowles, J., James, W. S., Cowley, S. A., and Wade–Martins, R. (2014) Physiological characterisation of human iPS–derived dopaminergic neurons, PLoS One, 9, e87388.
Stanslowsky, N., Haase, A., Martin, U., Naujock, M., Leffler, A., Dengler, R., and Wegner, F. (2014) Functional differentiation of midbrain neurons from human cord blood–derived induced pluripotent stem cells, Stem Cell Res. Ther., 5, 35.
Bagasrawala, I., Memi, F., Radonjic, N., and Zecevic, N. (2017) N–Methyl–D–aspartate receptor expression patterns in the human fetal cerebral cortex, Cereb. Cortex, 27, 5041–5053.
Skilbeck, K. J., Johnston, G. A., and Hinton, T. (2010) Stress and GABA receptors, J. Neurochem., 112, 1115–1130.
Momcilovic, O., Liu, Q., Swistowski, A., Russo–Tait, T., Zhao, Y., Rao, M. S., and Zeng, X. (2014) Genome wide profiling of dopaminergic neurons derived from human embryonic and induced pluripotent stem cells, Stem Cells Dev., 23, 406–420.
Gulacsi, A., Lee, C. R., Sik, A., Viitanen, T., Kaila, K., Tepper, J. M., and Freund, T. F. (2003) Cell type–specific differences in chloride–regulatory mechanisms and GABA(A) receptor–mediated inhibition in rat substantia nigra, J. Neurosci., 23, 8237–8246.
Kaila, K., Price, T. J., Payne, J. A., Puskarjov, M., and Voipio, J. (2014) Cation–chloride cotransporters in neu–ronal development, plasticity and disease, Nat. Rev. Neurosci., 15, 637–654.
Wang, C., Liu, F., Patterson, T. A., Paule, M. G., and Slikker, W., Jr. (2017) Relationship between ketamine–induced developmental neurotoxicity and NMDA recep–tor–mediated calcium influx in neural stem cell–derived neurons, Neurotoxicology, 60, 254–259.
Kritis, A. A., Stamoula, E. G., Paniskaki, K. A., and Vavilis, T. D. (2015) Researching glutamate–induced cyto–toxicity in different cell lines: a comparative/collective analysis/study, Front. Cell Neurosci., 9, 1–18.
Kikuchi, S., and Kim, S. U. (1993) Glutamate neurotoxic–ity in mesencephalic dopaminergic neurons in culture, J. Neurosci. Res., 36, 558–569.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2019, Vol. 84, No. 3, pp. 436–447.
Rights and permissions
About this article
Cite this article
Antonov, S.A., Novosadova, E.V., Kobylyansky, A.G. et al. Expression and Functional Properties of NMDA and GABAA Receptors during Differentiation of Human Induced Pluripotent Stem Cells into Ventral Mesencephalic Neurons. Biochemistry Moscow 84, 310–320 (2019). https://doi.org/10.1134/S0006297919030131
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297919030131