Abstract
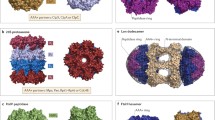
ATPases associated with diverse cellular activities (AAA+ proteins) are macromolecular machines that convert the chemical energy contained in ATP molecules into powerful mechanical forces to remodel a vast array of cellular substrates, including protein aggregates, macromolecular complexes and polymers. AAA+ proteins have key functionalities encompassing unfolding and disassembly of such substrates in different subcellular localizations and, hence, power a plethora of fundamental cellular processes, including protein quality control, cytoskeleton remodelling and membrane dynamics. Over the past 35 years, many of the key elements required for AAA+ activity have been identified through genetic, biochemical and structural analyses. However, how ATP powers substrate remodelling and whether a shared mechanism underlies the functional diversity of the AAA+ superfamily were uncertain. Advances in cryo-electron microscopy have enabled high-resolution structure determination of AAA+ proteins trapped in the act of processing substrates, revealing a conserved core mechanism of action. It has also become apparent that this common mechanistic principle is structurally adjusted to carry out a diverse array of biological functions. Here, we review how substrate-bound structures of AAA+ proteins have expanded our understanding of ATP-driven protein remodelling.







Similar content being viewed by others
References
Neuwald, A. F., Aravind, L., Spouge, J. L. & Koonin, E. V. AAA+: a class of chaperone-like ATPases associated with the assembly, operation, and disassembly of protein complexes. Genome Res. 9, 27–43 (1999).
Ogura, T. & Wilkinson, A. J. AAA+ superfamily ATPases: common structure-diverse function. Genes Cells 6, 575–597 (2001).
White, S. R. & Lauring, B. AAA+ ATPases: achieving diversity of function with conserved machinery. Traffic 8, 1657–1667 (2007).
Lyubimov, A. Y., Strycharska, M. & Berger, J. M. The nuts and bolts of ring-translocase structure and mechanism. Curr. Opin. Struct. Biol. 21, 240–248 (2011).
Erzberger, J. P. & Berger, J. M. Evolutionary relationships and structural mechanisms of AAA+ proteins. Annu. Rev. Biophys. Biomol. Struct. 35, 93–114 (2006).
Hanson, P. I. & Whiteheart, S. W. AAA+ proteins: have engine, will work. Nat. Rev. Mol. Cell Biol. 6, 519–529 (2005).
Iyer, L. M., Leipe, D. D., Koonin, E. V. & Aravind, L. Evolutionary history and higher order classification of AAA+ ATPases. J. Struct. Biol. 146, 11–31 (2004).
Monroe, N. & Hill, C. P. Meiotic Ccade AAA ATPases: protein polymer disassembly machines. J. Mol. Biol. 428, 1897–1911 (2016).
Roll-Mecak, A. & McNally, F. J. Microtubule-severing enzymes. Curr. Opin. Cell Biol. 22, 96–103 (2010).
Hartman, J. J. et al. Katanin, a microtubule-severing protein, is a novel AAA ATPase that targets to the centrosome using a WD40-containing subunit. Cell 93, 277–287 (1998).
Roll-Mecak, A. & Vale, R. D. Structural basis of microtubule severing by the hereditary spastic paraplegia protein spastin. Nature 451, 363–367 (2008).
Lata, S. et al. Helical structures of ESCRT-III are disassembled by VPS4. Science 321, 1354–1357 (2008).
Scott, A. et al. Structural and mechanistic studies of VPS4 proteins. EMBO J. 24, 3658–3669 (2005).
Frickey, T. & Lupas, A. N. Phylogenetic analysis of AAA proteins. J. Struct. Biol. 146, 2–10 (2004).
Brunger, A. T. & DeLaBarre, B. NSF and p97/VCP: similar at first, different at last. FEBS Lett. 555, 126–133 (2003).
Saffert, P., Enenkel, C. & Wendler, P. Structure and function of p97 and Pex1/6 type II AAA+ complexes. Front. Mol. Biosci. 4, 33 (2017).
Verma, R., Oania, R. S., Kolawa, N. J. & Deshaies, R. J. Cdc48/p97 promotes degradation of aberrant nascent polypeptides bound to the ribosome. eLife 2, e00308 (2013).
Vaz, B., Halder, S. & Ramadan, K. Role of p97/VCP (Cdc48) in genome stability. Front. Genet. 4, 60 (2013).
Torrecilla, I., Oehler, J. & Ramadan, K. The role of ubiquitin-dependent segregase p97 (VCP or Cdc48) in chromatin dynamics after DNA double strand breaks. Phil. Trans. R. Soc. B Biol. Sci. 372, 20160282 (2017).
Platta, H. W. et al. Pex2 and pex12 function as protein–ubiquitin ligases in peroxisomal protein import. Mol. Cell. Biol. 29, 5505–5516 (2009).
Doyle, S. M., Genest, O. & Wickner, S. Protein rescue from aggregates by powerful molecular chaperone machines. Nat. Rev. Mol. Cell Biol. 14, 617–629 (2013).
Parsell, D. A., Kowal, A. S., Singer, M. A. & Lindquist, S. Protein disaggregation mediated by heat-shock protein Hsp104. Nature 372, 475–478 (1994).
Parsell, D. A., Sanchez, Y., Stitzel, J. D. & Lindquist, S. Hsp104 is a highly conserved protein with two essential nucleotide-binding sites. Nature 353, 270–273 (1991).
Sweeny, E. A. & Shorter, J. Mechanistic and structural insights into the prion-disaggregase activity of Hsp104. J. Mol. Biol. 428, 1870–1885 (2016).
Bodnar, N. & Rapoport, T. Toward an understanding of the Cdc48/p97 ATPase. F1000Res. 6, 1318 (2017).
Glover, J. R. & Lindquist, S. Hsp104, Hsp70, and Hsp40: a novel chaperone system that rescues previously aggregated proteins. Cell 94, 73–82 (1998).
Sauer, R. T. & Baker, T. A. AAA+ proteases: ATP-fueled machines of protein destruction. Annu. Rev. Biochem. 80, 587–612 (2011).
Sauer, R. T. et al. Sculpting the proteome with AAA(+) proteases and disassembly machines. Cell 119, 9–18 (2004).
Gottesman, S. Proteolysis in bacterial regulatory circuits. Annu. Rev. Cell Dev. Biol. 19, 565–587 (2003).
Glynn, S. E. Multifunctional mitochondrial AAA proteases. Front. Mol. Biosci. 4, 34 (2017).
Livneh, I., Cohen-Kaplan, V., Cohen-Rosenzweig, C., Avni, N. & Ciechanover, A. The life cycle of the 26S proteasome: from birth, through regulation and function, and onto its death. Cell Res. 26, 869–885 (2016).
Saeki, Y. Ubiquitin recognition by the proteasome. J. Biochem. 161, 113–124 (2017).
Bashore, C. et al. Ubp6 deubiquitinase controls conformational dynamics and substrate degradation of the 26S proteasome. Nat. Struct. Mol. Biol. 22, 712–719 (2015).
Besche, H. C. et al. Autoubiquitination of the 26S proteasome on Rpn13 regulates breakdown of ubiquitin conjugates. EMBO J. 33, 1159–1176 (2014).
Rossmann, M. G., Moras, D. & Olsen, K. W. Chemical and biological evolution of nucleotide-binding protein. Nature 250, 194–199 (1974).
Walker, J. E., Saraste, M., Runswick, M. J. & Gay, N. J. Distantly related sequences in the α- and β-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1, 945–951 (1982).
Erdmann, R. et al. PAS1, a yeast gene required for peroxisome biogenesis, encodes a member of a novel family of putative ATPases. Cell 64, 499–510 (1991).
Saraste, M., Sibbald, P. R. & Wittinghofer, A. The P-loop-a common motif in ATP- and GTP-binding proteins. Trends Biochem. Sci. 15, 430–434 (1990).
Leipe, D. D., Koonin, E. V. & Aravind, L. Evolution and classification of P-loop kinases and related proteins. J. Mol. Biol. 333, 781–815 (2003).
Wendler, P., Ciniawsky, S., Kock, M. & Kube, S. Structure and function of the AAA+ nucleotide binding pocket. Biochim. Biophys. Acta 1823, 2–14 (2012).
Beyer, A. Sequence analysis of the AAA protein family. Protein Sci. 6, 2043–2058 (1997).
Karata, K., Inagawa, T., Wilkinson, A. J., Tatsuta, T. & Ogura, T. Dissecting the role of a conserved motif (the second region of homology) in the AAA family of ATPases. Site-directed mutagenesis of the ATP-dependent protease FtsH. J. Biol. Chem. 274, 26225–26232 (1999).
Ogura, T., Whiteheart, S. W. & Wilkinson, A. J. Conserved arginine residues implicated in ATP hydrolysis, nucleotide-sensing, and inter-subunit interactions in AAA and AAA+ ATPases. J. Struct. Biol. 146, 106–112 (2004).
Wang, J. et al. Crystal structures of the HslVU peptidase–ATPase complex reveal an ATP-dependent proteolysis mechanism. Structure 9, 177–184 (2001).
White, S. R., Evans, K. J., Lary, J., Cole, J. L. & Lauring, B. Recognition of C-terminal amino acids in tubulin by pore loops in Spastin is important for microtubule severing. J. Cell Biol. 176, 995–1005 (2007).
Martin, A., Baker, T. A. & Sauer, R. T. Pore loops of the AAA+ ClpX machine grip substrates to drive translocation and unfolding. Nat. Struc. Mol. Biol. 15, 1147–1151 (2008).
Yamada-Inagawa, T., Okuno, T., Karata, K., Yamanaka, K. & Ogura, T. Conserved pore residues in the AAA protease FtsH are important for proteolysis and its coupling to ATP hydrolysis. J. Biol. Chem. 278, 50182–50187 (2003).
Hinnerwisch, J., Fenton, W. A., Furtak, K. J., Farr, G. W. & Horwich, A. L. Loops in the central channel of ClpA chaperone mediate protein binding, unfolding, and translocation. Cell 121, 1029–1041 (2005).
Weibezahn, J. et al. Thermotolerance requires refolding of aggregated proteins by substrate translocation through the central pore of ClpB. Cell 119, 653–665 (2004).
Zolkiewski, M. A camel passes through the eye of a needle: protein unfolding activity of Clp ATPases. Mol. Microbiol. 61, 1094–1100 (2006).
Glynn, S. E., Martin, A., Nager, A. R., Baker, T. A. & Sauer, R. T. Structures of asymmetric ClpX hexamers reveal nucleotide-dependent motions in a AAA+ protein-unfolding machine. Cell 139, 744–756 (2009).
Olivares, A. O., Baker, T. A. & Sauer, R. T. Mechanistic insights into bacterial AAA+ proteases and protein-remodelling machines. Nat. Rev. Microbiol. 14, 33–44 (2016).
Lum, R., Tkach, J. M., Vierling, E. & Glover, J. R. Evidence for an unfolding/threading mechanism for protein disaggregation by Saccharomyces cerevisiae Hsp104. J. Biol. Chem. 279, 29139–29146 (2004).
Lee, C., Schwartz, M. P., Prakash, S., Iwakura, M. & Matouschek, A. ATP-dependent proteases degrade their substrates by processively unraveling them from the degradation signal. Mol. Cell 7, 627–637 (2001).
Bai, X. C., McMullan, G. & Scheres, S. H. How cryo-EM is revolutionizing structural biology. Trends Biochem. Sci. 40, 49–57 (2015).
Kuhlbrandt, W. Biochemistry. The resolution revolution. Science 343, 1443–1444 (2014).
Cheng, Y. Single-particle cryo-EM at crystallographic resolution. Cell 161, 450–457 (2015).
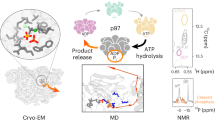
Huang, R. et al. Unfolding the mechanism of the AAA+ unfoldase VAT by a combined cryo-EM, solution NMR study. Proc. Natl Acad. Sci. USA 113, E4190–E4199 (2016).
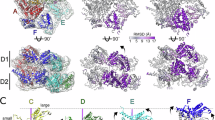
Ripstein, Z. A., Huang, R., Augustyniak, R., Kay, L. E. & Rubinstein, J. L. Structure of a AAA+ unfoldase in the process of unfolding substrate. eLife 6, e25754 (2017). This paper shows that substrate-free and substrate-bound structures of an archaeal type II AAA+ protein, VAT, reveal the conformational rearrangements associated with substrate processing, which suggests hand-over-hand substrate translocation through the central pore.
Monroe, N., Han, H., Shen, P. S., Sundquist, W. I. & Hill, C. P. Structural basis of protein translocation by the Vps4–Vta1 AAA ATPase. eLife 6, e24487 (2017).
Gates, S. N. et al. Ratchet-like polypeptide translocation mechanism of the AAA+ disaggregase Hsp104. Science 357, 273–279 (2017). This paper shows that structures of type II AAA+ protein Hsp104 in two different substrate-bound states support a rotary mechanism for ATP hydrolysis and a hand-over-hand substrate translocation.
Puchades, C. et al. Structure of the mitochondrial inner membrane AAA+ protease YME1 gives insight into substrate processing. Science 358, eaao0464 (2017). This paper shows that coexisting nucleotide states in the substrate-bound structure of AAA+ protease YME1 provide evidence for a sequential ATP hydrolysis cycle powering hand-over-hand substrate translocation. This study establishes the ISS motif as an allosteric modulator of both ATP hydrolysis and pore loop conformation.
Puchades, C. et al. Unique structural features of the mitochondrial AAA+ protease AFG3L2 reveal the molecular basis for activity in health and disease. Mol. Cell 75, 1073–1085.e6 (2019).
de la Pena, A. H., Goodall, E. A., Gates, S. N., Lander, G. C. & Martin, A. Substrate-engaged 26S proteasome structures reveal mechanisms for ATP-hydrolysis-driven translocation. Science 362, eaav0725 (2018). This paper shows that the structures of the actively hydrolysing AAA+ motor of the 26S proteasome stalled during substrate translocation provide a series of snapshots that show the different ATPase subunits proceeding along the staircase.
Dong, Y. et al. Cryo-EM structures and dynamics of substrate-engaged human 26S proteasome. Nature 565, 49–55 (2019).
Majumder, P. et al. Cryo-EM structures of the archaeal PAN-proteasome reveal an around-the-ring ATPase cycle. Proc. Natl Acad. Sci. USA 116, 534–539 (2019).
Zhao, M. et al. Mechanistic insights into the recycling machine of the SNARE complex. Nature 518, 61–67 (2015).
White, K. I., Zhao, M., Choi, U. B., Pfuetzner, R. A. & Brunger, A. T. Structural principles of SNARE complex recognition by the AAA+ protein NSF. eLife 7, e38888 (2018). This paper presents the structure of the 20S complex (type II AAA+ protein NSF bound to its endogenous substrate, the SNARE complex, via the adaptor protein αSNAP) and shows that only D1 of NSF engages the substrate.
Twomey, E. C. et al. Substrate processing by the Cdc48 ATPase complex is initiated by ubiquitin unfolding. Science 365, eaax1033 (2019). This paper shows the structure of type II AAA+ protein Cdc48 in complex with a polyubiquitylated substrate, trapped in the process of unfolding a ubiquitin molecule.
Cooney, I. et al. Structure of the Cdc48 segregase in the act of unfolding an authentic substrate. Science 365, 502–505 (2019).
Yokom, A. L. et al. Spiral architecture of the Hsp104 disaggregase reveals the basis for polypeptide translocation. Nat. Struct. Mol. Biol. 23, 830–837 (2016).
Rizo, A. N. et al. Structural basis for substrate gripping and translocation by the ClpB AAA+ disaggregase. Nat. Commun. 10, 2393 (2019).
Yu, H. et al. ATP hydrolysis-coupled peptide translocation mechanism of Mycobacterium tuberculosis ClpB. Proc. Natl Acad. Sci. USA 115, E9560–E9569 (2018).
Ho, C. M. et al. Malaria parasite translocon structure and mechanism of effector export. Nature 561, 70–75 (2018).
Alfieri, C., Chang, L. & Barford, D. Mechanism for remodelling of the cell cycle checkpoint protein MAD2 by the ATPase TRIP13. Nature 559, 274–278 (2018).
Gardner, B. M. et al. The peroxisomal AAA-ATPase Pex1/Pex6 unfolds substrates by processive threading. Nat. Commun. 9, 135 (2018).
Blok, N. B. et al. Unique double-ring structure of the peroxisomal Pex1/Pex6 ATPase complex revealed by cryo-electron microscopy. Proc. Natl Acad. Sci. USA 112, E4017–E4025 (2015).
Sun, S. et al. Cryo-EM structures of the ATP-bound Vps4(E233Q) hexamer and its complex with Vta1 at near-atomic resolution. Nat. Commun. 8, 16064 (2017).
Han, H., Monroe, N., Sundquist, W. I., Shen, P. S. & Hill, C. P. The AAA ATPase Vps4 binds ESCRT-III substrates through a repeating array of dipeptide-binding pockets. eLife 6, e31324 (2017). This article presents an in-depth analysis of the interactions with the substrate along the central pore of the type I AAA+ protein Vps4. This study defines class I and class II substrate binding pockets established by pore loop 1 and pore loop 2, respectively.
Zehr, E. et al. Katanin spiral and ring structures shed light on power stroke for microtubule severing. Nat. Struct. Mol. Biol. 24, 717–725 (2017).
Sandate, C. R., Szyk, A., Zehr, E. A., Lander, G. C. & Roll-Mecak, A. An allosteric network in spastin couples multiple activities required for microtubule severing. Nat. Struct. Mol. Biol. 26, 671–678 (2019). This paper shows that the substrate-bound structure of type I AAA+ protein spastin reveals a salt bridge-based allosteric mechanism that connects the pore loops of one subunit to the nucleotide-binding pocket of its neighbour.
Lander, G. C. et al. Complete subunit architecture of the proteasome regulatory particle. Nature 482, 186–191 (2012).
Matyskiela, M. E., Lander, G. C. & Martin, A. Conformational switching of the 26S proteasome enables substrate degradation. Nat. Struct. Mol. Biol. 20, 781–788 (2013).
Lasker, K. et al. Molecular architecture of the 26S proteasome holocomplex determined by an integrative approach. Proc. Natl Acad. Sci. USA 109, 1380–1387 (2012).
Iosefson, O., Nager, A. R., Baker, T. A. & Sauer, R. T. Coordinated gripping of substrate by subunits of a AAA+ proteolytic machine. Nat. Chem. Biol. 11, 201–206 (2015).
Han, H. et al. Binding of substrates to the central pore of the Vps4 ATPase is autoinhibited by the microtubule interacting and trafficking (MIT) domain and activated by MIT interacting motifs (MIMs). J. Biol. Chem. 290, 13490–13499 (2015).
Doyle, S. M. et al. Asymmetric deceleration of ClpB or Hsp104 ATPase activity unleashes protein-remodeling activity. Nat. Struct. Mol. Biol. 14, 114–122 (2007).
Barkow, S. R., Levchenko, I., Baker, T. A. & Sauer, R. T. Polypeptide translocation by the AAA+ ClpXP protease machine. Chem. Biol. 16, 605–612 (2009).
Bell, T. A., Baker, T. A. & Sauer, R. T. Interactions between a subset of substrate side chains and AAA+ motor pore loops determine grip during protein unfolding. eLife 8, e46808 (2019).
Shorter, J. & Southworth, D. R. Spiraling in control: structures and mechanisms of the Hsp104 disaggregase. Cold Spring Harb. Perspect. Biol. 11, a034033 (2019).
Banerjee, S. et al. 2.3 A resolution cryo-EM structure of human p97 and mechanism of allosteric inhibition. Science 351, 871–875 (2016).
Hartman, J. J. & Vale, R. D. Microtubule disassembly by ATP-dependent oligomerization of the AAA enzyme katanin. Science 286, 782–785 (1999).
Burton, R. E., Baker, T. A. & Sauer, R. T. Nucleotide-dependent substrate recognition by the AAA+ HslUV protease. Nat. Struct. Mol. Biol. 12, 245–251 (2005).
Weibezahn, J., Schlieker, C., Bukau, B. & Mogk, A. Characterization of a trap mutant of the AAA+ chaperone ClpB. J. Biol. Chem. 278, 32608–32617 (2003).
Dalal, S., Rosser, M. F., Cyr, D. M. & Hanson, P. I. Distinct roles for the AAA ATPases NSF and p97 in the secretory pathway. Mol. Biol. Cell 15, 637–648 (2004).
Liu, S., Chistol, G. & Bustamante, C. Mechanical operation and intersubunit coordination of ring-shaped molecular motors: insights from single-molecule studies. Biophys. J. 106, 1844–1858 (2014). This article reviews single-molecule studies of AAA+ proteins from diverse clades, highlighting the operational versatility and different mechanochemical properties of AAA+ motors.
Rodriguez-Aliaga, P., Ramirez, L., Kim, F., Bustamante, C. & Martin, A. Substrate-translocating loops regulate mechanochemical coupling and power production in AAA+ protease ClpXP. Nat. Struct. Mol. Biol. 23, 974–981 (2016). This paper presents a mutational study demonstrating that the bulkiness of pore loop 1 residues influences substrate grip as well as the mechano-chemical properties of AAA+ motors.
Shin, M. et al. Distinct structural features of the Lon protease drive conserved hand-over-hand substrate translocation. Preprint at bioRxiv https://doi.org/10.1101/617159 (2019).
Gatsogiannis, C., Balogh, D., Merino, F., Sieber, S. & Raunser, S. Cryo-EM structure of the ClpXP protein degradation machinery. Nat. Struct. Mol. Biol. 26, 946–954 (2019).
Fei, X. et al. Structures of the ATP-fueled ClpXP proteolytic machine bound to protein substrate. Preprint at bioRxiv https://doi.org/10.1101/704999 (2019).
Enemark, E. J. & Joshua-Tor, L. Mechanism of DNA translocation in a replicative hexameric helicase. Nature 442, 270–275 (2006).
Thomsen, N. D. & Berger, J. M. Running in reverse: the structural basis for translocation polarity in hexameric helicases. Cell 139, 523–534 (2009).
Itsathitphaisarn, O., Wing, R. A., Eliason, W. K., Wang, J. & Steitz, T. A. The hexameric helicase DnaB adopts a nonplanar conformation during translocation. Cell 151, 267–277 (2012).
Gai, D., Zhao, R., Li, D., Finkielstein, C. V. & Chen, X. S. Mechanisms of conformational change for a replicative hexameric helicase of SV40 large tumor antigen. Cell 119, 47–60 (2004).
Maillard, R. A. et al. ClpX(P) generates mechanical force to unfold and translocate its protein substrates. Cell 145, 459–469 (2011).
Johjima, A. et al. Microtubule severing by katanin p60 AAA+ ATPase requires the C-terminal acidic tails of both α- and β-tubulins and basic amino acid residues in the AAA+ ring pore. J. Biol. Chem. 290, 11762–11770 (2015).
Olivares, A. O., Nager, A. R., Iosefson, O., Sauer, R. T. & Baker, T. A. Mechanochemical basis of protein degradation by a double-ring AAA+ machine. Nat. Struct. Mol. Biol. 21, 871–875 (2014).
Djuranovic, S. et al. Structure and activity of the N-terminal substrate recognition domains in proteasomal ATPases. Mol. Cell 34, 580–590 (2009).
Ryu, J. K. et al. Spring-loaded unraveling of a single SNARE complex by NSF in one round of ATP turnover. Science 347, 1485–1489 (2015).
Augustin, S. et al. An intersubunit signaling network coordinates ATP hydrolysis by m-AAA proteases. Mol. Cell 35, 574–585 (2009). This article presents the first biochemical study to identify the ISS motif and describe its functional relevance.
McNally, F. J. & Vale, R. D. Identification of katanin, an ATPase that severs and disassembles stable microtubules. Cell 75, 419–429 (1993).
Deville, C., Franke, K., Mogk, A., Bukau, B. & Saibil, H. R. Two-step activation mechanism of the ClpB disaggregase for sequential substrate threading by the main ATPase motor. Cell Rep. 27, 3433–3446.e4 (2019).
Hattendorf, D. A. & Lindquist, S. L. Cooperative kinetics of both Hsp104 ATPase domains and interdomain communication revealed by AAA sensor-1 mutants. EMBO J. 21, 12–21 (2002).
DeSantis, M. E. et al. Operational plasticity enables hsp104 to disaggregate diverse amyloid and nonamyloid clients. Cell 151, 778–793 (2012).
Eckert, T. et al. Subunit Interactions and cooperativity in the microtubule-severing AAA ATPase spastin. J. Biol. Chem. 287, 26278–26290 (2012).
Martin, A., Baker, T. A. & Sauer, R. T. Rebuilt AAA+ motors reveal operating principles for ATP-fuelled machines. Nature 437, 1115–1120 (2005).
Martin, A., Baker, T. A. & Sauer, R. T. Protein unfolding by a AAA+ protease is dependent on ATP-hydrolysis rates and substrate energy landscapes. Nat. Struct. Mol. Biol. 15, 139–145 (2008).
Sen, M. et al. The ClpXP protease unfolds substrates using a constant rate of pulling but different gears. Cell 155, 636–646 (2013).
Lee, S. et al. Cryo-EM structures of the Hsp104 protein disaggregase captured in the ATP conformation. Cell Rep. 26, 29–36.e3 (2019).
Moffitt, J. R. et al. Intersubunit coordination in a homomeric ring ATPase. Nature 457, 446–450 (2009).
Acknowledgements
The authors thank M. Shin for helpful discussions. Preparation of this Review was supported by an American Heart Association predoctoral fellowship to C.P. (17PRE32910005), a National Science Foundation predoctoral fellowship to C.R.S. (2016219351) and a Pew Scholarship in the Biomedical Sciences from the Pew Charitable Trusts and the National Institutes of Health (DP2EB020402 and R21AG06169701) to G.C.L.
Author information
Authors and Affiliations
Contributions
C.P. conceived and drafted the manuscript, C.R.S and C.P. prepared the figures. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Molecular Cell Biology thanks C. Hill and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- ESCRT-III
-
(Endosomal sorting complex required for transport III). ESCRT-III proteins are recruited to membrane constriction sites, including nearly all subcellular membrane compartments, where they are activated and assemble into filaments, which in turn recruit VPS4, a type I AAA+ protein. ATP-dependent VPS4 activity remodels and disassembles ESCRT-III polymers, thereby powering ESCRT-dependent membrane fission reactions that are required for diverse biological processes, such as vesicle formation in the secretory system, budding of enveloped viruses from the plasma membrane and membrane repair.
- SNARE complexes
-
Protein complexes consisting of syntaxin, synaptobrevin and SNAP25 (synaptosome-associated protein), which assemble into a four-helix bundle that aids in the fusion of membranes.
- ER-associated degradation
-
A cellular pathway that targets misfolded proteins for selective ubiquitylation by endoplasmic reticulum (ER)-resident ubiquitin ligases. The type II AAA+ protein p97/Cdc48 recognizes and dislocates these polyubiquitylated substrates from the ER membrane and into the cytosol. The resulting unfolded polypeptides are subsequently degraded by the 26S proteasome.
- Mitochondria-associated degradation
-
The process by which the AAA+ protein p97/Cdc48 recognizes and retrotranslocates polyubiquitylated substrates from the outer mitochondrial membrane for subsequent degradation by the 26S proteasome.
- Retrotranslocation
-
Following translation in the cytosol, proteins are translocated into the respective cellular subcompartments. When a protein is misfolded, AAA+ proteins extract these proteins from the membrane, dislocating them into the cytosol. This process is known as retrotranslocation (from the subcompartment back into the cytosol).
- 26S proteasome
-
A large multisubunit complex located in the cytosol of eukaryotes with numerous ubiquitin receptors that selectively bind polyubiquitylated protein substrates for degradation. Targeted substrates are unfolded by a AAA+ motor within the complex, while another enzyme called a deubiquitinase cleaves the covalently linked ubiquitin chain from the substrate. The AAA+ ATPase directs the unfolded substrate into a barrel-shaped proteolytic chamber which contains six proteolytic active sites that degrade the substrate.
- NTPases
-
A generic term that encompasses enzymes capable of binding nucleotide triphosphate (NTP) molecules, such as ATP and GTP. AAA+ proteins are defined as a subclass of P-loop NTPases.
- α–β Rossman fold
-
A super-secondary structure composed of alternating β-strand–α-helix–β-strand segments. The β-strands form a β-sheet and the α–helices surround both faces of the sheet, producing a three-layered sandwich.
- Walker A
-
A G-XXXX-GK-[T/S] sequence motif, where X can be any amino acid. This motif (also known as the P-loop) stabilizes the binding of the nucleotide by interacting with the β-phosphate, and is present in many nucleotide-binding proteins.
- Walker B
-
A consensus sequence (hhhhDE) where h represents any bulky, hydrophobic amino acid. The aspartic acid (D) is important for coordination of a magnesium ion, which in turn helps neutralize the negative charges of the phosphate groups present in the nucleotide. The adjacent glutamate (E) residue serves as a catalytic base, activating water for nucleophilic attack on the γ-phosphate during ATP hydrolysis.
- Nucleophilic attack
-
A fundamental reaction class in which a partially or fully positively-charged group (electrophile) is attacked by an electron-rich molecule (nucleophile) that substitutes a leaving group.
- Cation–π interactions
-
Non-covalent interactions between an electron-rich π system (for example, aromatic amino acids — phenylalanine, tryptophan and tyrosine) and an adjacent cation (for example, basic residues — arginine, lysine and histidine).
- van der Waals interactions
-
Distance-dependent interactions between atoms or molecules that are significantly weaker than other kinds of interactions, such as electrostatic ones.
- SNAPs
-
(Soluble N-ethylmaleimide-sensitive factor (NSF) attachment proteins). Adaptor proteins that bind both the N-terminal domains of type II AAA+ protein NSF and a SNARE complex, giving rise to the so-called 20S complex.
- π-stacking interactions
-
Electrostatic interactions that can occur between two π systems. In proteins, aromatic residues that are in close proximity to each other can engage in such interactions.
Rights and permissions
About this article
Cite this article
Puchades, C., Sandate, C.R. & Lander, G.C. The molecular principles governing the activity and functional diversity of AAA+ proteins. Nat Rev Mol Cell Biol 21, 43–58 (2020). https://doi.org/10.1038/s41580-019-0183-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41580-019-0183-6
- Springer Nature Limited
This article is cited by
-
Molecular basis for transposase activation by a dedicated AAA+ ATPase
Nature (2024)
-
Visualization of the Cdc48 AAA+ ATPase protein unfolding pathway
Nature Communications (2024)
-
Mechanism of allosteric inhibition of human p97/VCP ATPase and its disease mutant by triazole inhibitors
Communications Chemistry (2024)
-
Mechanisms of SNARE proteins in membrane fusion
Nature Reviews Molecular Cell Biology (2024)
-
Unwinding of a eukaryotic origin of replication visualized by cryo-EM
Nature Structural & Molecular Biology (2024)