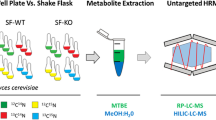
Abstract
Our understanding of metabolism is limited by a lack of knowledge about the functions of many enzymes. Here, we develop a high-throughput mass spectrometry approach to comprehensively profile proteins for in vitro enzymatic activity. Overexpressed or purified proteins are incubated in a supplemented metabolome extract containing hundreds of biologically relevant candidate substrates, and accumulating and depleting metabolites are determined by nontargeted mass spectrometry. By combining chemometrics and database approaches, we established an automated pipeline for unbiased annotation of the functions of novel enzymes. In screening all 1,275 functionally uncharacterized Escherichia coli proteins, we discovered 241 potential novel enzymes, 12 of which we experimentally validated. Our high-throughput in vitro metabolomics method is generally applicable to any purified protein or crude cell lysate of its overexpression host and enables performing up to 1,200 nontargeted enzyme assays per working day.



Similar content being viewed by others
References
Hanson, A.D., Pribat, A., Waller, J.C. & de Crécy-Lagard, V. 'Unknown' proteins and 'orphan' enzymes: the missing half of the engineering parts list--and how to find it. Biochem. J. 425, 1–11 (2009).
Galperin, M.Y. & Koonin, E.V. 'Conserved hypothetical' proteins: prioritization of targets for experimental study. Nucleic Acids Res. 32, 5452–5463 (2004).
Jaroszewski, L. et al. Exploration of uncharted regions of the protein universe. PLoS Biol. 7, e1000205 (2009).
Sorokina, M., Stam, M., Médigue, C., Lespinet, O. & Vallenet, D. Profiling the orphan enzymes. Biol. Direct 9, 10 (2014).
Chen, L. & Vitkup, D. Distribution of orphan metabolic activities. Trends Biotechnol. 25, 343–348 (2007).
Tian, W. & Skolnick, J. How well is enzyme function conserved as a function of pairwise sequence identity? J. Mol. Biol. 333, 863–882 (2003).
Bork, P. Powers and pitfalls in sequence analysis: the 70% hurdle. Genome Res. 10, 398–400 (2000).
Blaby-Haas, C.E. & de Crécy-Lagard, V. Mining high-throughput experimental data to link gene and function. Trends Biotechnol. 29, 174–182 (2011).
Galperin, M.Y. Conserved 'hypothetical' proteins: new hints and new puzzles. Comp. Funct. Genomics 2, 14–18 (2001).
Tipton, K. & Boyce, S. Nomenclature committee of the international union of biochemistry and molecular biology (NC-IUBMB), Enzyme Supplement 5 (1999). Eur. J. Biochem. 264, 610–650 (1999).
Pouliot, Y. & Karp, P.D. A survey of orphan enzyme activities. BMC Bioinformatics 8, 244 (2007).
Shearer, A.G., Altman, T. & Rhee, C.D. Finding sequences for over 270 orphan enzymes. PLoS One 9, e97250 (2014).
Lespinet, O. Orphan enzymes? Science 307, 42 (2005).
Sévin, D.C., Kuehne, A., Zamboni, N. & Sauer, U. Biological insights through nontargeted metabolomics. Curr. Opin. Biotechnol. 34, 1–8 (2015).
Feist, A.M., Herrgård, M.J., Thiele, I., Reed, J.L. & Palsson, B.Ø. Reconstruction of biochemical networks in microorganisms. Nat. Rev. Microbiol. 7, 129–143 (2009).
Nam, H. et al. Network context and selection in the evolution to enzyme specificity. Science 337, 1101–1104 (2012).
Kuznetsova, E. et al. Enzyme genomics: application of general enzymatic screens to discover new enzymes. FEMS Microbiol. Rev. 29, 263–279 (2005).
Prosser, G.A., Larrouy-Maumus, G. & de Carvalho, L.P. Metabolomic strategies for the identification of new enzyme functions and metabolic pathways. EMBO Rep. 15, 657–669 (2014).
Lee, D., Redfern, O. & Orengo, C. Predicting protein function from sequence and structure. Nat. Rev. Mol. Cell Biol. 8, 995–1005 (2007).
Plata, G., Fuhrer, T., Hsiao, T.-L., Sauer, U. & Vitkup, D. Global probabilistic annotation of metabolic networks enables enzyme discovery. Nat. Chem. Biol. 8, 848–854 (2012).
Zhao, S. et al. Discovery of new enzymes and metabolic pathways by using structure and genome context. Nature 502, 698–702 (2013).
Saito, N. et al. Metabolite profiling reveals YihU as a novel hydroxybutyrate dehydrogenase for alternative succinic semialdehyde metabolism in Escherichia coli. J. Biol. Chem. 284, 16442–16451 (2009).
Notebaart, R.A. et al. Network-level architecture and the evolutionary potential of underground metabolism. Proc. Natl. Acad. Sci. USA 111, 11762–11767 (2014).
Guzmán, G.I. et al. Model-driven discovery of underground metabolic functions in Escherichia coli. Proc. Natl. Acad. Sci. USA 112, 929–934 (2014).
Coelho, P.S. et al. A serine-substituted P450 catalyzes highly efficient carbene transfer to olefins in vivo. Nat. Chem. Biol. 9, 485–487 (2013).
Saito, N. et al. Metabolomics approach for enzyme discovery. J. Proteome Res. 5, 1979–1987 (2006).
Kuznetsova, E. et al. Genome-wide analysis of substrate specificities of the Escherichia coli haloacid dehalogenase-like phosphatase family. J. Biol. Chem. 281, 36149–36161 (2006).
Bennett, B.D. et al. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nat. Chem. Biol. 5, 593–599 (2009).
Fuhrer, T., Heer, D., Begemann, B. & Zamboni, N. High-throughput, accurate mass metabolome profiling of cellular extracts by flow injection-time-of-flight mass spectrometry. Anal. Chem. 83, 7074–7080 (2011).
Kanehisa, M. et al. Data, information, knowledge and principle: back to metabolism in KEGG. Nucleic Acids Res. 42, D199–D205 (2014).
Xu, Y.F., Lu, W. & Rabinowitz, J.D. Avoiding misannotation of in-source fragmentation products as cellular metabolites in liquid chromatography-mass spectrometry-based metabolomics. Anal. Chem. 87, 2273–2281 (2015).
Kitagawa, M. et al. Complete set of ORF clones of Escherichia coli ASKA library (a complete set of E. coli K-12 ORF archive): unique resources for biological research. DNA Res. 12, 291–299 (2005).
Hattori, M., Okuno, Y., Goto, S. & Kanehisa, M. Development of a chemical structure comparison method for integrated analysis of chemical and genomic information in the metabolic pathways. J. Am. Chem. Soc. 125, 11853–11865 (2003).
Hattori, M., Tanaka, N., Kanehisa, M. & Goto, S. SIMCOMP/SUBCOMP: chemical structure search servers for network analyses. Nucleic Acids Res. 38, W652–W656 (2010).
da Silva, R.R., Dorrestein, P.C. & Quinn, R.A. Illuminating the dark matter in metabolomics. Proc. Natl. Acad. Sci. USA 112, 12549–12550 (2015).
Yamamoto, N. et al. Update on the Keio collection of Escherichia coli single-gene deletion mutants. Mol. Syst. Biol. 5, 335 (2009).
Struys, E.A. et al. Mutations in the D-2-hydroxyglutarate dehydrogenase gene cause D-2-hydroxyglutaric aciduria. Am. J. Hum. Genet. 76, 358–360 (2005).
Dang, L. et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462, 739–744 (2009).
Linster, C.L., Van Schaftingen, E. & Hanson, A.D. Metabolite damage and its repair or pre-emption. Nat. Chem. Biol. 9, 72–80 (2013).
Amend, J.P. & Shock, E.L. Energetics of overall metabolic reactions of thermophilic and hyperthermophilic Archaea and bacteria. FEMS Microbiol. Rev. 25, 175–243 (2001).
Khersonsky, O. & Tawfik, D.S. Enzyme promiscuity: a mechanistic and evolutionary perspective. Annu. Rev. Biochem. 79, 471–505 (2010).
Nobeli, I., Favia, A.D. & Thornton, J.M. Protein promiscuity and its implications for biotechnology. Nat. Biotechnol. 27, 157–167 (2009).
Fuhrer, T. & Zamboni, N. High-throughput discovery metabolomics. Curr. Opin. Biotechnol. 31, 73–78 (2015).
Dunn, W.B. et al. Mass appeal: metabolite identification in mass spectrometry-focused untargeted metabolomics. Metabolomics 9, 44–66 (2013).
Li, L. et al. MyCompoundID: using an evidence-based metabolome library for metabolite identification. Anal. Chem. 85, 3401–3408 (2013).
Nichols, R.J. et al. Phenotypic landscape of a bacterial cell. Cell 144, 143–156 (2011).
Lorenz, P. & Eck, J. Metagenomics and industrial applications. Nat. Rev. Microbiol. 3, 510–516 (2005).
Figge, R., Barbier, G. & Bestel-Corre, G. Production of N-acylated sulphur-containing amino acids with microorganisms having enhanced N-acyltransferase enzymatic activity. US patent US20100047880 A1 (2010).
Zor, T. & Selinger, Z. Linearization of the Bradford protein assay increases its sensitivity: theoretical and experimental studies. Anal. Biochem. 236, 302–308 (1996).
Riesenberg, D. et al. High cell density cultivation of Escherichia coli at controlled specific growth rate. J. Biotechnol. 20, 17–27 (1991).
Kashket, E.R. Effects of aerobiosis and nitrogen source on the proton motive force in growing Escherichia coli and Klebsiella pneumoniae cells. J. Bacteriol. 146, 377–384 (1981).
Acknowledgements
We thank S. Suter and J. Schmitz for technical assistance with validation enzyme assays. Funding was provided by the MetaNetX project of the Swiss Initiative for Systems Biology (SystemsX.ch; http://metanetx.org; evaluated by the Swiss National Science Foundation) and the Swiss Federal Government through the Federal Office of Education and Science.
Author information
Authors and Affiliations
Contributions
D.C.S. and T.F. performed the experiments and analyzed the data. D.C.S., T.F. and N.Z. developed data analysis software and algorithms. D.C.S., N.Z. and U.S. designed the research and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–21 (PDF 6949 kb)
Supplementary Table 1
Annotation of ions as metabolites based on matching their accurate mass with compounds in the KEGG eco database. (XLSX 899 kb)
Supplementary Table 2
Annotation of ions as metabolites based on correlation with ions annotated by accurate mass. (XLSX 35 kb)
Supplementary Table 3
All strains of the ASKA library used in this work for His-tagged protein expression and purification. (XLSX 100 kb)
Supplementary Table 4
Known metabolic enzymes with ions passing the Z-score cutoff of 5.0. (XLSX 30 kb)
Supplementary Table 5
Functionally uncharacterized proteins with ions passing the Z-score cutoff of 5.0. (XLSX 53 kb)
Supplementary Table 6
Enzymatic reactions predicted to be catalyzed by uncharacterized proteins, based on matching differential metabolites of opposing change direction with the KEGG main reactant pair database. (XLSX 16 kb)
Supplementary Table 7
Predicted reactant pairs based on molecular similarity among two differential metabolites of opposing change direction. (XLSX 17 kb)
Supplementary Table 8
Predicted reactant pairs based on mass difference among two ions of opposing change direction. (XLSX 16 kb)
Supplementary Table 9
List of experimentally tested reaction predictions and outcomes. (XLSX 14 kb)
Supplementary Table 10
Reactant ions that show consistent changes in respective deletion mutants of predicted enzymes. (XLSX 15 kb)
Supplementary Table 11
Growth phenotyping of E. coli single-gene deletion mutants each lacking one of the 12 experimentally validated enzymes. (XLSX 13 kb)
Supplementary Data Set 1
Z-scores of ions in assays of purified functionally uncharacterized proteins. Proteins with concentrations < 50 mg/L were not analyzed. (CSV 26690 kb)
Supplementary Data Set 2
Z-scores of ions in cell lysate assays of strains overexpressing functionally uncharacterized proteins. (CSV 19446 kb)
Supplementary Data Set 3
Metabolomics of 223 viable single-gene deletion mutants each lacking a predicted enzyme discovered in this study. (CSV 8592 kb)
Rights and permissions
About this article
Cite this article
Sévin, D., Fuhrer, T., Zamboni, N. et al. Nontargeted in vitro metabolomics for high-throughput identification of novel enzymes in Escherichia coli. Nat Methods 14, 187–194 (2017). https://doi.org/10.1038/nmeth.4103
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.4103
- Springer Nature America, Inc.
This article is cited by
-
Emerging tools and best practices for studying gut microbial community metabolism
Nature Metabolism (2024)
-
The Y-ome Conundrum: Insights into Uncharacterized Genes and Approaches for Functional Annotation
Molecular and Cellular Biochemistry (2024)
-
Pickaxe: a Python library for the prediction of novel metabolic reactions
BMC Bioinformatics (2023)
-
Discovery of a non-canonical prototype long-chain monoacylglycerol lipase through a structure-based endogenous reaction intermediate complex
Nature Communications (2023)
-
Stable isotope-assisted metabolite profiling reveals new insights into L-tryptophan chemotrophic metabolism of Rubrivivax benzoatilyticus
World Journal of Microbiology and Biotechnology (2023)