Abstract
A central hurdle in developing small interfering RNAs (siRNAs) as therapeutics is the inefficiency of their delivery across the plasma and endosomal membranes to the cytosol, where they interact with the RNA interference machinery. With the aim of improving endosomal release, a poorly understood and inefficient process, we studied the uptake and cytosolic release of siRNAs, formulated in lipoplexes or lipid nanoparticles, by live-cell imaging and correlated it with knockdown of a target GFP reporter. siRNA release occurred invariably from maturing endosomes within ∼5–15 min of endocytosis. Cytosolic galectins immediately recognized the damaged endosome and targeted it for autophagy. However, inhibiting autophagy did not enhance cytosolic siRNA release. Gene knockdown occurred within a few hours of release and required <2,000 copies of cytosolic siRNAs. The ability to detect cytosolic release of siRNAs and understand how it is regulated will facilitate the development of rational strategies for improving the cytosolic delivery of candidate drugs.




Similar content being viewed by others
References
Felgner, P.L. et al. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. Proc. Natl. Acad. Sci. USA 84, 7413–7417 (1987).
Elouahabi, A. & Ruysschaert, J.-M. Formation and intracellular trafficking of lipoplexes and polyplexes. Mol. Ther. 11, 336–347 (2005).
Kanasty, R., Dorkin, J.R., Vegas, A. & Anderson, D. Delivery materials for siRNA therapeutics. Nat. Mater. 12, 967–977 (2013).
Zimmermann, T.S. et al. RNAi-mediated gene silencing in nonhuman primates. Nature 441, 111–114 (2006).
Semple, S.C. et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 28, 172–176 (2010).
Coelho, T. et al. Safety and efficacy of RNAi therapy for transthyretin amyloidosis. N. Engl. J. Med. 369, 819–829 (2013).
Gilleron, J. et al. Image-based analysis of lipid nanoparticle–mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat. Biotechnol. 31, 638–646 (2013).
Sahay, G. et al. Efficiency of siRNA delivery by lipid nanoparticles is limited by endocytic recycling. Nat. Biotechnol. 31, 653–658 (2013).
Payne, C.K., Jones, S.A., Chen, C. & Zhuang, X. Internalization and trafficking of cell surface proteoglycans and proteoglycan-binding ligands. Traffic 8, 389–401 (2007).
ur Rehman, Z., Hoekstra, D. & Zuhorn, I.S. Mechanism of polyplex- and lipoplex-mediated delivery of nucleic acids: real-time visualization of transient membrane destabilization without endosomal lysis. ACS Nano 7, 3767–3777 (2013).
Li, X. et al. Generation of destabilized green fluorescent protein as a transcription reporter. J. Biol. Chem. 273, 34970–34975 (1998).
Thurston, T.L.M., Wandel, M.P., von Muhlinen, N., Foeglein, Á. & Randow, F. Galectin 8 targets damaged vesicles for autophagy to defend cells against bacterial invasion. Nature 482, 414–418 (2012).
Stalder, L. et al. The rough endoplasmatic reticulum is a central nucleation site of siRNA-mediated RNA silencing. EMBO J. 32, 1115–1127 (2013).
Mu, F.T. et al. EEA1 is a conserved α-helical peripheral membrane protein flanked by cysteine “fingers” and contains a calmodulin-binding IQ motif. J. Biol. Chem. 270, 13503–13511 (1995).
Huotari, J. & Helenius, A. Endosome maturation. EMBO J. 30, 3481–3500 (2011).
Poteryaev, D., Datta, S., Ackema, K., Zerial, M. & Spang, A. Identification of the switch in early-to-late endosome transition. Cell 141, 497–508 (2010).
Nechaev, S. et al. Intracellular processing of immunostimulatory CpG–siRNA: Toll-like receptor 9 facilitates siRNA dicing and endosomal escape. J. Control. Release 170, 307–315 (2013).
Chen, X. et al. Autophagy induced by calcium phosphate precipitates targets damaged endosomes. J. Biol. Chem. 289, 11162–11174 (2014).
Roberts, R. et al. Autophagy and formation of tubulovesicular autophagosomes provide a barrier against nonviral gene delivery. Autophagy 9, 667–682 (2013).
Kimura, S., Noda, T. & Yoshimori, T. Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3, 452–460 (2007).
Paz, I. et al. Galectin-3, a marker for vacuole lysis by invasive pathogens. Cell. Microbiol. 12, 530–544 (2010).
Maier, M.A. et al. Biodegradable lipids enabling rapidly eliminated lipid nanoparticles for systemic delivery of RNAi therapeutics. Mol. Ther. 21, 1570–1578 (2013).
Akinc, A. et al. Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol. Ther. 18, 1357–1364 (2010).
Alabi, C.A. et al. Multiparametric approach for the evaluation of lipid nanoparticles for siRNA delivery. Proc. Natl. Acad. Sci. USA 110, 12881–12886 (2013).
Jayaraman, M. et al. Maximizing the potency of siRNA lipid nanoparticles for hepatic gene silencing in vivo. Angew. Chem. Int. Ed. Engl. 51, 8529–8533 (2012).
McNamara, J.O. et al. Cell type–specific delivery of siRNAs with aptamer-siRNA chimeras. Nat. Biotechnol. 24, 1005–1015 (2006).
Stewart, S.A. et al. Lentivirus-delivered stable gene silencing by RNAi in primary cells. RNA 9, 493–501 (2003).
Sharma, D.K. et al. Glycosphingolipids internalized via caveolar-related endocytosis rapidly merge with the clathrin pathway in early endosomes and form microdomains for recycling. J. Biol. Chem. 278, 7564–7572 (2003).
Choudhury, A. et al. Rab proteins mediate Golgi transport of caveola-internalized glycosphingolipids and correct lipid trafficking in Niemann-Pick C cells. J. Clin. Invest. 109, 1541–1550 (2002).
Acknowledgements
The authors thank E. Marino for maintaining the Imaging Resource used in this study and K. Ketman and N. Barteneva for technical assistance. This work was supported by US National Institutes of Health (NIH) grants AI090671 and CA139444 (J.L.), the Swedish Research Council (A.W.), NIH grant GM075252 (T.K.) and New England Regional Center of Excellence in Biodefense and Emerging Infectious Disease, Core Imaging Facility grant AI057159 (T.K.).
Author information
Authors and Affiliations
Contributions
A.W. and J.L. conceived the study, designed the experiments and wrote the manuscript. T.K. provided advice. A.W. performed most of the experiments. A.A., P.H., R.T. and X.L. performed some of the experiments. K.C. and M.M. provided LNPs and advice for their use.
Corresponding authors
Ethics declarations
Competing interests
J.L. is a member of the Scientific Board of Alnylam Pharmaceuticals. K.C. and M.M. are employees of Alnylam Pharmaceuticals.
Integrated supplementary information
Supplementary Figure 1 High dynamic range (HDR) imaging of live cells with spinning disk confocal microscopy.
During image acquisition, z-sections, encompassing most of the cellular volume, are captured with short exposure times adjusted to the brightest structures in the cell. After all short exposures are captured, a single plane with approximately 10 times longer exposure time, is captured from the lower third of the cell. Only a single plane with long exposure time is cap- tured in order to limit phototoxicity.
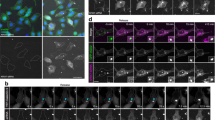
Supplementary Figure 2 Cytosolic siRNA release coincides with decrease in vesicular siRNA fluorescence.
HeLa cells were incubated with siRNA-AF647 lipoplexes and imaged every 3 s. Blue line: fluorescence intensity traces of releasing vesicle (from maximum projection of short exposure images). Red line: fluorescence intensity traces from cytosolic Region of Interest (ROI) adjacent to the releasing vesicle (from single plane long exposure images). This representative tracing suggests release occurs as sudden discrete events without prior leakage.
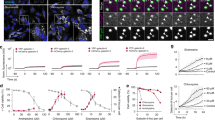
Supplementary Figure 3 Cytosolic release of siRNA is associated with cytosolic calcium transients.
HeLa cells pre-loaded with the fluores- cent Ca++ indicator Fluo-4 were incubated with siRNA-AF647 lipoplexes and imaged every 15 s. a, Representative cell exhibiting a small localized calcium release coincident with siRNA release. Scale bar, 10 μm. b, Quantitation of whole-cell cytosolic siRNA fluorescence (red curve, left axis) and Ca++/Fluo-4 fluorescence (blue curve, right axis) in four typical cells (1-4). siRNA release events are indicated with black triangles. Spontaneous Ca++ spikes are seen in cell 4, for comparison. Ca++ transients during release are of similar or smaller magnitude than spontaneous Ca++ fluctuations.
Supplementary Figure 4 Cytosolic release of siRNA in both non-illuminated and continously imaged cells.
HeLa cells were incubated with 100 nM siRNA-AF647 lipoplexes for 1 h and either continously imaged every 10 s (right panels) or left non-illuminated until the end of the incuba- tion (left panels). Red outlines indicate cells that have experienced a release event and white dashed outlines indicate cells without release events. Cells are counterstained to facilitate localization (Fluo-4). Scale bar, 20 μm. Shown is representative fields of >10 imaged. The same observation, i.e., no difference in release probability between illuminated and adjacent fields has been observed throughout the imaging experiments reported herein. In addition, the extent of knockdown measured with flow cytometry in end-point analyses recapitulates the extent of knockdown seen during continous imaging (not shown).
Supplementary Figure 5 Endosomes releasing lipoplex siRNA do not associate with the ER.
HeLa cells expressing an ER-GFP marker were incubated with siRNA-AF647 lipoplexes and imaged every 1.5 min. Whole cell (upper row, scale bar 10 μm) and detail time-lapse images of cell exhibiting a release event. Scale bar, 2 μm. White triangles indicate a representative releasing endosome.
Supplementary Figure 6 LNPs merge with pre-existing EEA1+ struc- tures and rapidly lose EEA1 association around the time of release.
HeLa cells, expressing mCherry-galectin-8 and GFP-EEA1, were incubat- ed with LNP-siRNA-AF647 and imaged every 10 s. Detail time-lapse images of an LNP containing vesicle (indicated with filled white triangles) associating with mCherry-galectin-8, are shown. Open triangles indicate pre-existing EEA1+ vesicle. Scale bar, 2 μm.
Supplementary Figure 7 LC3 is recruited to LNP containing vesicles.
HeLa cells expressing tfLC3 (GFP signal: green) were incubated with LNP-siRNA-AF647 and imaged every 30 s. Detail time-lapse images of an LNP-containing vesicle (white triangles) associating with LC3, are shown. Scale bar, 2 μm.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and legends (PDF 3865 kb)
Rights and permissions
About this article
Cite this article
Wittrup, A., Ai, A., Liu, X. et al. Visualizing lipid-formulated siRNA release from endosomes and target gene knockdown. Nat Biotechnol 33, 870–876 (2015). https://doi.org/10.1038/nbt.3298
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3298
- Springer Nature America, Inc.
This article is cited by
-
Engineered EVs with pathogen proteins: promising vaccine alternatives to LNP-mRNA vaccines
Journal of Biomedical Science (2024)
-
Emerging non-viral vectors for gene delivery
Journal of Nanobiotechnology (2023)
-
Aberrant Cyclin D1 splicing in cancer: from molecular mechanism to therapeutic modulation
Cell Death & Disease (2023)
-
Single-cell quantification and dose-response of cytosolic siRNA delivery
Nature Communications (2023)
-
Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs
Nature Reviews Materials (2023)