Abstract
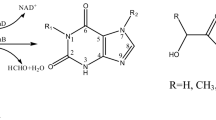
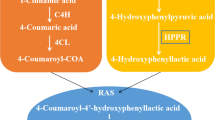
Hydroxyphenylpyruvate reductase (HPPR) is an enzyme involved in the biosynthesis of rosmarinic acid in Lamiaceae reducing hydroxyphenylpyruvates in dependence of NAD(P)H to the corresponding hydroxyphenyllactates. The HPPR protein was purified from suspension cells of Coleus blumei accumulating high levels of rosmarinic acid by ammonium sulfate precipitation, anion exchange chromatography, hydroxylapatite chromatography, chromatography on 2′,5′-ADP-Sepharose 4B and SDS-polyacrylamide gel electrophoresis. The protein was tryptically digested and the peptides sequenced. Sequence information was used to isolate a full-length cDNA-clone for HPPR (EMBL accession number AJ507733) by RT-PCR, screening of a C. blumei cDNA-library and 5′-RACE-PCR. The open reading frame of the HPPR-cDNA consists of 939 nucleotides encoding a protein of 313 amino acid residues. The sequence showed that HPPR belongs to the family of D-isomer-specific 2-hydroxyacid dehydrogenases. The HPPR-cDNA was heterologously expressed in Escherichia coli and the protein was shown to catalyse the NAD(P)H-dependent reduction of 4-hydroxyphenylpyruvate to 4-hydroxyphenyllactate and 3,4-dihydroxyphenylpyruvate to 3,4-dihydroxyphenyllactate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bernard, N., Delcour, J., Alvarez, A., Cortes, A., Willis, C. and Holbrook, J.J. 1995. Dehydrogenases engineering to correct substrate inhibition in a commercial dehydrogenase. Entry in the Protein Data Base.
Blum, H., Beler, H. and Gross, H.J. 1987. Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8: 93–99.
Boland, M.J. and Schubert, K.R. 1983. Phosphoglycerate dehydrogenase from soybean nodules. Partial purification and some kinetic properties. Plant Physiol. 71: 658–661.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Capela, D., Barloy-Hubler, F., Gouzy, J., Bothe, G., Ampe, F., Batut, J., Boistard, P., Becker, A., Boutry, M., Cadieu, E., Dreano, S., Gloux, S., Godrie, T., Goffeau, A., Kahn, D., Kiss, E., Lelaure, V., Masuy, D., Pohl, T., Portetelle, D., Puehler, A., Purnelle, B., Ramsperger, U., Renard, C., Thebault, P., Vandenbol, M., Weidner, S. and Galibert, F. 2001. Analysis of the chromosome sequence of the legume symbiont Sinorhizobium meliloti strain 1021. Proc. Natl. Acad. Sci. USA 98: 9877–9882.
D'Amelio, F.S. 1999. Botanicals: A Phytocosmetic Desk Reference. CRC Press, Boca Raton, FL, p. 361.
De Eknamkul, W. and Ellis, B.E. 1987. Tyrosine aminotransferase: the entrypoint enzyme of the tyrosine-derived pathway in rosmarinic acid biosynthesis. Phytochemistry 26: 1941–1946.
Dengler, U., Niefind, K., Kiess, M. and Schomburg, D. 1997. Crystal structure of a ternary complex of d-2-hydroxyisocaproate dehydrogenase from Lactobacillus casei, NAD+ and 2-oxoisocaproate at 1.9 Å resolution. J. Mol. Biol. 267: 640–660.
Douce, R. and Joyard, J. 1996. Biosynthesis of thylakoid membrane lipids. In: D.R. Ort and C.F. Yocum (Eds.) Advances in Photosynthesis, vol. 4. Oxygenic Photosynthesis: The Light Reactions, Kluwer Academic Publishers, Dordrecht, Netherlands, pp. 69–101.
Ellis, B.E. and Towers, G.H.N. 1970. Biogenesis of rosmarinic acid in Mentha. Biochem. J. 118: 291–297.
Garvie, E.L. 1980. Bacterial lactate dehydrogenases. Microbiol. Rev. 44: 106–139.
Goldberg, J.D., Yoshida, T. and Brick, P. 1994. Crystal structure of a NAD-dependent D-glycerate dehydrogenase at 2.4 Å resolution. J. Mol. Biol. 236: 1123–1140.
Graeve, K., von Schaewen, A. and Scheibe, R. 1994. Purification, characterization and cDNA sequence of glucose-6-phosphate dehydrogenase from potato (Solanum tuberosum L.). Plant J. 5: 353–361.
Grant, G.A. 1989. A new family of 2-hydroxyacid dehydrogenases. Biochem. Biophys. Res. Comm. 165: 1371–1374.
Greenler, J.M., Sloan, J.S., Schwartz, B.W. and Becker, W.M. 1989. Isolation, characterization and sequence analysis of a full-length cDNA clone encoding NADH-dependent hydroxypyruvate reductase from cucumber. Plant Mol. Biol. 13: 139–150.
Haslam, E. 1993. Shikimic Acid: Metabolism and Metabolites. Wiley, Chichester, UK.
Haösler, E., Petersen, M. and Alfermann, A.W. 1991. Hydroxyphenylpyruvate reductase from cell suspension cultures of Coleus blumei Benth. Z. Naturforsch. 46c: 371–376.
Hess, J.L. 1993. Vitamin E, a-tocopherol. In: R.G. Alscher and J.L. Hess (Eds.) Antioxidants in Higher Plants, CRC Press, Boca Raton, FL, pp. 111–134.
Ho, C.T., Ferraro, T., Chen, Q., Rosen, R.T. and Huang, M.T. 1994. Phytochemicals in teas and rosemary and their cancerpreventive properties. In: C.T. Ho, T. Osawa, M.T. Huang and R.T. Rosen (Eds.) Food Phytochemicals II: Teas, Spices, and Herbs, American Chemical Society, Washington, pp. 2–19.
Kochhar, S., Hunziker, P.E., Leong-Morgenthaler, P. and Hottinger, H. 1992. Evolutionary relationship of NAD+-dependent D-lactate dehydrogenase: comparison of primary structure of 2-hydroxy acid dehydrogenases. Biochem. Biophys. Res. Comm. 184: 60–66.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Meinhard, E.J. 1991. DeVersuche zur Reinigung der Hydroxyphenylpyruvatreduktase aus Zellkulturen von Coleus blumei. Diploma Thesis, University of Düsseldorf, Germany.
Parnham, M.J. and Kesselring, K. 1985. Rosmarinic acid. Drugs of the Future 10: 756–757.
Petersen, M. 1997. Cytochrome P450-dependent hydroxylation in the biosynthesis of rosmarinic acid in Coleus. Phytochemistry 45: 1165–1172.
Petersen, M.S. 1991. Characterization of rosmarinic acid synthase from cell cultures of Coleus blumei. Phytochemistry 30: 2877–2881.
Petersen, M. and Alfermann, A.W. 1988. Two new enzymes of rosmarinic acid biosynthesis from cell cultures of Coleus blumei: hydroxyphenylpyruvate reductase and rosmarinic acid synthase. Z. Naturforsch. 43c: 501–504.
Petersen, M., Haüsler, E., Karwatzki, B. and Meinhard, J. 1993. Proposed biosynthetic pathway for rosmarinic acid in cell cultures of Coleus blumei. Planta 189: 10–14.
Petersen, M., Haüsler, E., Meinhard, J., Karwatzki, B. and Gertlowski, C. 1994. The biosynthesis of rosmarinic acid in suspension cultures of Coleus blumei. Plant Cell Tiss. Org. Cult. 38: 171–179.
Petersen, M. and Simmonds, M.S.J. 2003. Rosmarinic acid. Phytochemistry 62: 121–125.
Petersen, M., Szabo, E., Meinhard, J., Karwatzki, B., Gertlowski, C., Kempin, B. and Fuss, E. 1995a. Biosynthesis and accumulation of rosmarinic acid in suspension cultures of Coleus blumei. Plant Cell Tiss. Org. Cult. 43: 89–92.
Petersen, M., Van der Straeten, D. and Bauw, G. 1995b. Fulllength cDNA clone from Coleus blumei (Z49150) with high similarity to cobalamine-independent methionine-synthase (PGR 95-049). Plant Physiol. 109: 338.
Razzaque, A. and Ellis, B.E. 1977. Rosmarinic acid production in Coleus cell cultures. Planta 137: 287–291.
Rosenblum, I.Y. and Sallach, H.J. 1975. D-3-phosphoglycerate dehydrogenase from wheat germ. Meth. Enzymol. 41: 285–289.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory Press, Plainview, NY.
Schuller, D.J., Grant, G.A. and Banaszak, L.J. 1995. The allosteric ligand site in the Vmax-type cooperative enzyme phosphoglycerate dehydrogenase. Nature Struct. Biol. 2: 69–76.
Schwartz, B.W., Sloan, J.S. and Becker, W.M. 1991. Characterization of genes encoding hydroxypyruvate reductase in cucumber. Plant Mol. Biol. 17: 941–947.
Slaughter, J.C. and Davies, D.D. 1975. 3-Phosphoglycerate dehydrogenase from seedlings of Pisum sativum. Meth. Enzymol. 41: 278–281.
Szabo, E., Thelen, A. and Petersen, M. 1999. Fungal elicitor preparations and methyl jasmonate enhance rosmarinic acid accumulation in in vitro-cultures of Coleus blumei. Plant Cell Rep. 18: 485–489.
Theologis, A., Ecker, J.R., Palm, C.J., Federspiel, N.A., Kaul, S., White, O., Alonso, J., Altafi, H., Araujo, R., Bowman, C.L., Brooks, S.Y., Buehler, E., Chan, A., Chao, Q., Chen, H., Cheuk, R.F., Chin, C.W., Chung, M.K., Conn, L., Conway, A.B., Conway, A.R., Creasy, T.H., Dewar, K., Dunn, P., Etgu, P., Feldblyum, T.V., Feng, J.-D., Fong, B., Fujii, C.Y., Gill, J.E., Goldsmith, A.D., Haas, B., Hansen, N.F., Hughes, B., Huizar, L., Hunter, J.L., Jenkins, J., Johnson-Hopson, C., Khan, S., Khaykin, E., Kim, C.J., Koo, H.L., Kremenetskaia, I., Kurtz, D.B., Kwan, A., Lam, B., Langin-Hooper, S., Lee, A., Lee, J.M., Lenz, C.A., Li, J.H., Li, Y.-P., Lin, X., Liu, S.X., Liu, Z.A., Luros, J.S., Maiti, R., Marziali, A., Militscher, J., Miranda, M., Nguyen, M., Nierman, W.C., Osborne, B.I., Pai, G., Peterson, J., Pham, P.K., Rizzo, M., Rooney, T., Rowley, D., Sakano, H., Salzerg, S.L., Schwartz, J.R., Shinn, P., Southwick, A.M., Sun, H., Tallon, L.J., Tambunga, G., Toriumi, M.J., Town, C.D., Utterback, T., Van Aken, S., Vaysberg, M., Vysotskaia, V.S., Walker, M., Wu, D., Yu, G., Fraser, C.M., Venter, J.C. and Davis, R.W. 2000. Sequence and analysis of chromosome 1 of the plant Arabidopsis thaliana. Nature 408: 816–820.
Wölbling, R.H. and Leonhardt, K. 1994. Local therapy of herpes simplex with dried extracts from Melissa officinalis. Phytomedicine 1: 25–31.
Wood, D.W., Setubal, J.C., Kaul, R., Monks, D.E., Kitajima, J.P., Okura, V.K., Zhou, Y., Chen, L., Wood, G.E., Almeida, N.F. Jr., Woo, L., Chen, Y., Paulsen, I.T., Eisen, J.A., Karp, P.D., Bovee, D. Sr., Chapman, P., Clendenning, J., Deatherage, G., Gillet, W., Grant, C., Kutyavin, T., Levy, R., Li, M.-J., McClelland, E., Palmieri, A., Raymond, C., Rouse, G., Saenphimmachak, C., Wu, Z., Romero, P., Gordon, D., Zhang, S., Yoo, H., Tao, Y., Biddle, P., Jung, M., Krespan, W., Perry, M., Gordon-Kamm, B., Liao, L., Kim, S., Hendrick, C., Zhao, Z.-Y., Dolan, M., Chumley, F., Tingey, S.V., Tomb, J.-F., Gordon, M.P., Olson, M.V. and Nester, E.W. 2001. The genome of the natural genetic engineer Agrobacterium tumefaciens C58. Science 294: 2317–2323.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, K.H., Janiak, V. & Petersen, M. Purification, Cloning and Functional Expression of hydroxyphenylpyruvate Reductase Involved in Rosmarinic Acid Biosynthesis in Cell Cultures of Coleus Blumei . Plant Mol Biol 54, 311–323 (2004). https://doi.org/10.1023/B:PLAN.0000036367.03056.b2
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000036367.03056.b2