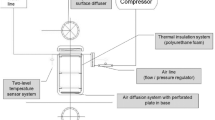
Abstract
Non-isothermal kinetic parameters regarding to the thermal decomposition of the ligninocellulosic fraction present in compost from urban solid residues (USR) obtained through stack covered (SC) with composted material, comes from the usine in composing of Araraquara city, Săo Paulo state, Brazil, and from stack containing academic restaurant organic solid residues (SAR). The samples were periodically revolved round 132 days of composting. Results from TG, DTG and DSC curves obtained on inert atmosphere indicated that the lignocellulosic fraction present, despite the slow degradation during the composting process, is thermally less stable than other substances originated during that process. The lignocellulosic fraction decomposition, between 200 and 400°C, were kinetically evaluated through non-isothermal methods of analysis. By using the Flynn-Wall and Ozawa isoconversional method, the medium activation energy, Ea, and pre-exponential factor, lgA, were 283.0±14.6, 257.6±1.3 kJ mol-1 and 25.4±0.8, 23.2±0.2 min-1, to the SC and SAR, respectively, at 95% confidence level. From E a and lgA values and DSC curves, Malek procedure could be applied, suggesting that the SB (Sesták-Berggren) kinetic model is suitable for the first thermal decomposition step.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. G. Grossi, Tese de Doutorado, Instituto de Química, Universidade de São Paulo 1993, p. 222.
X. T. He, S. J. Traina and T. J. Logan, J. Environ. Qual., 21 (1992) 318.
B. Chefetz, P. G. Hatcher, Y. Hadar and Y. Chen, J. Environ. Qual., 25 (1996) 776.
S. C. Oliveira, C. A. Ribeiro, M. R. Santiago and M. S. Crespi, J. Compost and Utilization, 2002, submitted.
P. Jandura, B. Riedel and B. Kokta, Polym. Degrad. Stab., 70 (2000) 387.
M. E. Brown, D. Dollimore and A. K. Galwey, Reaction in the Solid State, Comprehensive Chemical Kinetics, Vol. 22, Elsevier, Amsterdam 1980.
D. Dollimore and P. Phang, Anal. Chem., 72 (2000) 27.
S. Vyazovkin and C. A. Wight, Thermochim. Acta, 340–341 (1999) 53.
T. Vlase, G. Vlase, M. Doca and N. Doca, J. Therm. Anal. Cal., 72 (2003) 579.
T. Vlase, G. Vlase, A. Chiriac and N. Doca, J. Therm. Anal. Cal., 72 (2003) 839.
T. Vlase, G. Vlase, A. Chiriac and N. Doca, J. Therm. Anal. Cal., 72 (2003) 847.
P. Budrugeac and E. Segal, J. Therm. Anal. Cal., 72 (2003) 831.
J. H. Flynn and J. Wall, Nat. Bur. Stand., 70A (1966) 487.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
T. Ozawa, J. Thermal Anal., 2 (1970) 301.
C. Doyle, J. Appl. Polym. Sci., 6 (1962) 639.
H. E. Kissinger, Anal. Chem., 29 (1957) 1702.
J. Malek, J. Šesták, F. Rouquerol, J. Rouquerol, J. M. Criado and A. Ortega, J. Thermal Anal., 38 (1992) 71.
J. Malek, J. Therm. Anal. Cal., 56 (1999) 763.
J. Malek, Thermochim. Acta, 355 (2000) 239.
J. Malek, J. Mat. Res., 16 (2001) 1862.
S. C. Oliveira, Dissertação de Mestrado, Instituto de Química, Universidade Estadual Paulista, 1997.
J. Šesták and G. Berggren, Thermochim. Acta, 3 (1971) 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, A.R., Crespi, M.S., Ribeiro, C.A. et al. Kinetic of thermal decomposition of residues from different kinds of composting. Journal of Thermal Analysis and Calorimetry 75, 401–409 (2004). https://doi.org/10.1023/B:JTAN.0000027126.92699.ba
Issue Date:
DOI: https://doi.org/10.1023/B:JTAN.0000027126.92699.ba