Abstract
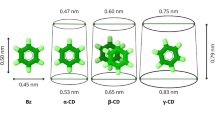
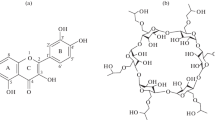
Inclusion complex formation betweensixteen para-substituted phenols andβ-cyclodextrin have been investigated in orderto establish Quantitative Structure AffinityRelationships. An analytical methodology is proposed,in order to obtain reliable evaluation of bindingaffinities. Potentiometry and circular dichroism havebeen applied to define experimental conditions and toconfirm postulated equilibriums. In addition, the useof algorithmic treatments and concentrationoptimisation to determine formation constants leads tocoherent values between 1H NMR, direct UVSpectroscopy and the spectral displacement method. Theresults emphasise the contribution of van der Waalsinteractions, provided that no significant differencein the dipole of the molecule arises from thepara-substituent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Szejtli: Chem. Rev. 98, 1743 (1998).
W. J. Shieh and A. R. Hedges: Pure Appl. Chem. A33, 673 (1996).
J. Szejtli: J. Mater Chem. 7, 575 (1997).
J. S. Pagington: Chemistry in Britain, 455 (1987).
W. Saenger: Angew. Chem. Int. Ed. Engl. 19, 344 (1980).
F. Fava and F. Grassi: Biotech. Tech. 10, 291 (1996).
G. Crini, S. Bertini, G. Torri, A. Naggi, D. Sforzini, C. Vecchi, L. Janus, Y. Lekchiri, and M. Morcellet: J. Appl. Polym., Sci. 68, 1973 (1998).
I. Uemasu and S. Kushiyama: J. Incl. Phenom. 17, 177 (1994).
S. Tanada, T. Nakamura, N. Kawasaki, S. Kitayama, and Y. Takebe: J. Colloid Interface Sci. 186, 180 (1997).
D. A. Dantz, C. Meschke, H.-J. Buschmannand, and E. Schollmeyer: Supramol. Chem. 9, 79 (1998).
E. Fenyvesy, J. Szeman and J. Szejtli: J. Incl. Phenom. 25, 229 (1996).
X. Wang and M. L. Brusseau: Environ. Sci. Technol. 19, 2632 (1995).
M. T. Butterfield, R. A. Agbaria, and I. M. Warner: Anal. Chem. 68, 1187 (1996).
L. Liu and Q.-X. Guo: J. Phys. Chem. B 103, 3461 (1999).
K. A. Connors: Chem. Rev. 97, 1325 (1997).
K. A. Connors and D. D. Pendergast: J. Am. Chem. Soc. 106, 7607 (1984).
R. Carpignano, M. Marzona, E. Cattaneo, and S. Quaranta: Anal. Chim. Acta 348, 489 (1997).
M. V. Rekharsky and Y. Inoue: Chem. Rev. 98, 1875 (1998).
S. Hamai and N. Satoh: Carbohydr. Res. 304, 229 (1997).
L. Szente, in J. L. Atwood, J. E. D. Davies, D. D. MacNicol, and F. Vögtle (eds.), Comprehensive Supramolecular Chemistry, Vol. 3, Pergamon, Oxford, 1996, p. 253.
S. M. Hoenigman and C. E. Evans: Anal Chem. 68, 3274 (1996).
Y. Matsui, M. Fujie, and H. Sakate: Bull. Chem. Soc. Jpn. 61, 3409 (1988).
S.-F. Lin and K. A. Connors: J. Pharm. Sci. 72, 1333 (1983).
Y. Liu, B.-H. Han, A.-D. Qi, and R.-T. Chen: Bioorg. Chem. 25, 155 (1997).
S. Li and W. C. Purdy: Anal. Chem. 64, 1405 (1992).
A. Buvari-Barcza, E. Rak, A. Meszaros, and L. Barcza: J. Incl. Phenom. 32, 453 (1998).
C. Hansch and A. Leo: Substituent Constants for Correlation Analysis in Chemistry and Biology, John Wiley & Sons, New York (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Landy, D., Fourmentin, S., Salome, M. et al. Analytical Improvement in Measuring Formation Constants of Inclusion Complexes between β-Cyclodextrin and Phenolic Compounds. Journal of Inclusion Phenomena 38, 187–198 (2000). https://doi.org/10.1023/A:1008156110999
Issue Date:
DOI: https://doi.org/10.1023/A:1008156110999