Abstract
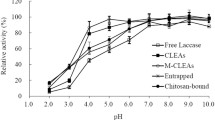
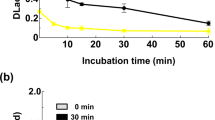
Laccase (E.C. 1.10.3.2) from Trametes versicolor was immobilized (adsorbed) by drying on various supports (glass, glass powder, silica gel, and Nylon 66 membrane). The enzyme activity and stability were determined in diethyl ether, ethyl acetate, and methylene chloride. The initial rate for the oxidation of syringaldazine varied up to 245-fold depending on the solvent and support, the best results being obtained with Nylon 66 membrane. No inactivation of immobilized laccase over 72 h was observed in diethyl ether and ethyl acetate, while exposure to methylene chloride resulted in significant activity decreases regardless of the support material.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chaudhary AK, Kamat SV, Beckman EJ, Nurok D, Kleyle RM, Hajdu P, Russell AJ (1996) Control of subtilisin substrate speci-ficity by solvent engineering in organic solvents and supercritical fluoroform. J. Am. Chem. Soc. 118: 12891–12901.
Chin JT, Wheeler SL, Klibanov AM (1994) On protein solubility in organic solvents. Biotechnol. Bioeng. 44: 140–145.
Clonfero E, Nardini B, Marchioro M, Bordin A, Gabbani G (1996) Mutagenecy and contents of polycyclic aromatic hydrocarbons in used and recycled motor oils. Mutat. Res. 368: 283–291.
Field JA, de Jong E, Costa GF, Bont JAM (1992) Biodegradation of polycyclic aromatic hydrocarbons by new isolates of white rot fungi. Appl. Environ. Microbiol. 58: 2219–2216.
Gianfreda L, Sannino F, Filazzola MT, Leonowicz A (1998) Catalytic behavior and detoxifying ability of a laccase from the fungal strain Cerrena unicolor. J. Mol. Catal. B Enzym. 4: 13–23.
Kilburn KH, Warshaw RH (1995) Neurotoxic effects from residential exposure to chemicals from an oil reprocessing facility and superfund site. Neurotoxicol. Teratol. 17: 89–102.
Kirk TK, Farrell RL (1987) Enzymatic 'combustion': the microbial degradation of lignin. Annu. Rev. Microbiol. 41: 465–505.
Klibanov AM (1989) Enzymatic catalysis in anhydrous organic solvents. Trends Biochem. Sci. 14: 141–144.
Klibanov AM (1997) Why are enzymes less active in organic solvents than in water? Trends Biotechnol. 15: 97–101.
Laane C, Boeren S, Kees V, Veeger C (1987) Rules for optimization of biocatalysis in organic solvents. Biotechnol. Bioeng. 30: 81–87.
Milstein O, Nicklas B, Hüttermann A (1989) Oxidation of aromatic compounds in organic solvents with laccase from Trametes versicolor. Appl. Microbiol. Biotechnol. 31: 70–74.
Orsat B, Drtina GJ, Williams MG, Klibanov AM (1994) Effect of support material and enzyme pretreatment on enantioselectivity of immobilized subtilisin in organic solvents. Biotechnol. Bioeng. 44: 1265–1269.
Rama R, Mougin C, Boyer F-D, Kollman A, Malosse C, Sigoillet, J-C (1998) Biotransformation of benzo[a]pyrene in bench scale reactor using laccase of Pycnoporus cinnabarinus. Biotechnol. Lett. 20: 1101–1104.
van Erp C, Kamenskaya EO, Khmelnitsky YL (1991) The effect of water content and nature of organic solvent on enzyme activity in low-water media. A quantitative description. Eur. J. Biochem. 202: 379–384.
Yadav JS, Quensen JF III, Tiedje JM, Reddy CA (1995) Degradation of polychlorinated biphenyl mixtures (Acroclors 1242, 1254, and 1260) by the white rot fungus Phanerochaete chrysosporium as evidenced by congener-specific analysis. Appl. Environ. Microbiol 61: 2560–2565.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ruiz, A.I., Malavé, A.J., Felby, C. et al. Improved activity and stability of an immobilized recombinant laccase in organic solvents. Biotechnology Letters 22, 229–233 (2000). https://doi.org/10.1023/A:1005698301681
Issue Date:
DOI: https://doi.org/10.1023/A:1005698301681