Abstract
Background
AMPA receptors are highly expressed throughout the central nervous system and are suggested to be involved in mood regulation. Studies found changes in glutamate, its metabolites and receptors in patients with bipolar disorder (BPD) or major depression (MD) and in animal models of stress. Additional data suggest that the glutamatergic system and AMPA receptors specifically, have an important role in modulating the therapeutic effects of mood stabilizers. Further research on the role of AMPA receptors in mood regulation can be done using AMPAkines, positive modulators of AMPA receptors. AMPAkines have been studied for cognitive enhancement in neurodegenerative disorders and some were also examined in preclinical studies of mood disorders. In that context, the present study was designed to test the effects of the AMPAkine CX717 in a strain specific battery of tests for mania-like behaviors.
Methods
Black Swiss male mice were sub-chronically treated with 5 different doses of CX717 or vehicle and tested in a battery of behavioral tests including spontaneous activity, sweet solution preference, resident-intruder, forced swim and amphetamine-induced hyperactivity.
Results
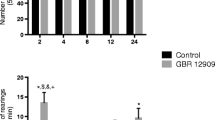
Data show that CX717 doses of 30 mg/kg and above, but not lower, reduce activity levels. Moreover, 45 mg/kg and above reduce interactions in the resident-intruder test and ameliorate amphetamine-induced hyperactivity.
Conclusions
The results therefore show a partial effect of CX717 on manic-like behavior, somewhat similar to previously demonstrated effects of atypical antipsychotic drugs in this strain. It is therefore suggested that further work related to AMPAkines in the treatment of affective disorders might be warranted.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Carlson PJ, Singh JB, Zarate CAJJr, Drevets WC, Manji HK. Neural circuitry and neuroplasticity in mood disorders: insights for novel therapeutic targets. NeuroRx 2006;3(1):22–41.
Machado-Vieira R, Ibrahim L, Henter ID, Zarate CAJ Jr. Novel glutamatergic agents for major depressive disorder and bipolar disorder. Pharmacol Biochem Behav 2012;100(4):678–87. Epub 2011 Sep 25.
Du J, Gray NA, Falke CA, Chen W, Yuan P, Szabo ST, et al. Modulation of synaptic plasticity by antimanic agents: the role of AMPA glutamate receptor subunit 1 synaptic expression. J Neurosci 2004;24(29):6578–89.
Snyder GL, Allen PB, Fienberg AA, Valle CG, Huganir RL, Nairn AC, et al. Regulation of phosphorylation of the GluR1 AMPA receptor in the neostriatum by dopamine and psychostimulants in vivo. J Neurosci 2000;20(12):4480–8.
Du J, Creson TK, Wu LJ, Ren M, Gray NA, Falke C, et al. The role of hippocampal GluR1 and GluR2 receptors in manic-like behavior. J Neurosci 2008;28(1):68–79.
Beneyto M, Kristiansen LV, Oni-Orisan A, McCullumsmith RE, Meador-Woodruff JH. Abnormal glutamate receptor expression in the medial temporal lobe in schizophrenia and mood disorders. Neuropsychopharmacology 2007;32(9): 1888–902.
Ng WX, Lau IY, Graham S, Sim K. Neurobiological evidence for thalamic, hippocampal and related glutamatergic abnormalities in bipolar disorder: a review and synthesis. Neurosci Biobehav Rev 2009;33(3):336–54.
Bleakman D,Alt A, Witkin JM. AMPA receptors in the therapeutic management of depression. CNS Neurol Disord Drug Targets 2007;6(2):117–26.
Kishimoto H, Yamada K, Iseki E, Kosaka K, Okoshi T. Brain imaging of affective disorders and schizophrenia. Psychiatry Clin Neurosci 1998;52(Suppl.):S212–4.
Dickerson F, Stallings C, Vaughan C, Origoni A, Khushalani S, Yolken R. Antibodies to the glutamate receptor in mania. Bipolar Disord 2012;14(5):547–53. https://doi.org/10.1111/j.399-5618.2012.01028.x. Epub 2012 Jun 6.
Auer DP, Putz B, Kraft E, Lipinski B, Schill J, Holsboer F. Reduced glutamate in the anterior cingulate cortex in depression: an in vivo proton magnetic resonance spectroscopy study. Biol Psychiatry 2000;47(4):305–13.
Lapidus KA, Soleimani L, Murrough JW. Novel glutamatergic drugs for the treatment of mood disorders. Neuropsychiatr Dis Treat 2013;9:1101–12. https://doi.org/10.2147/NDT.S36689. Epub 2013 Aug 7.
Meador-Woodruff JH, Hogg AJJJr, Smith RE. Striatal ionotropic glutamate receptor expression in schizophrenia, bipolar disorder, and major depressive disorder. Brain Res Bull 2001;55(5):631–40.
Paul IA, Skolnick P. Glutamate and depression: clinical and preclinical studies. Ann N Y Acad Sci 2003;1003:250–72.
Reznikov LR, Grillo CA, Piroli GG, Pasumarthi RK, Reagan LP, Fadel J. Acute stress-mediated increases in extracellular glutamate levels in the rat amygdala: differential effects of antidepressant treatment. Eur J Neurosci 2007;25(10):3109–14.
Zhang XH, Jia N, Zhao XY, Tang GK, Guan LX, Wang D, et al. Involvement of pGluR1, EAAT2 and EAAT3 in offspring depression induced by prenatal stress. Neuroscience 2013;250:333–41. https://doi.org/10.1016/j.neuroscience.2013.04.031. Epub May 18.
McCullumsmith RE, Meador-Woodruff JH. Striatal excitatory amino acid transporter transcript expression in schizophrenia, bipolar disorder, and major depressive disorder. Neuropsychopharmacology 2002;26(3):368–75.
Gray NA,Du J, Falke CS, Yuan P, Manji HK. Lithium regulates total and synaptic expression of the AMPA glutamate receptor GluR2 in vitro and in vivo. Ann NY Acad Sci 2003;1003:402–4.
Gould TD, O’Donnell KC, Dow ER, Du J, Chen G, Manji HK. Involvement of AMPA receptors in the antidepressant-like effects of lithium in the mouse tail suspension test and forced swim test. Neuropharmacology 2008;54(3):577–87.
Coyle JT, Duman RS. Finding the intracellular signaling pathways affected by mood disorder treatments. Neuron 2003;38(2):157–60.
Du J, Gray NA, Falke C, Yuan P, Szabo S, Manji HK. Structurally dissimilar antimanic agents modulate synaptic plasticity by regulating AMPA glutamate receptor subunit GluR1 synaptic expression. Ann N Y Acad Sci 2003;1003: 378–80.
Kubota-Sakashita M, Iwamoto K, Bundo M, Kato T. A role of ADAR2 and RNA editing of glutamate receptors in mood disorders and schizophrenia. Mol Brain 2014;7:5.
Lynch G. Glutamate-based therapeutic approaches: ampakines. Curr Opin Pharmacol 2006;6(1):82–8.
Arai AC, Kessler M. Pharmacology of ampakine modulators: from AMPA receptors to synapses and behavior. Curr Drug Targets 2007;8(5):583–602.
Arai A, Guidotti A, Costa E, Lynch G. Effect of the AMPA receptor modulator IDRA 21 on LTP in hippocampal slices. Neuroreport 1996;7(13):2211–5.
Hess US, Whalen SP, Sandoval LM, Lynch G, Gall CM. Ampakines reduce methamphetamine-driven rotation and activate neocortex in a regionally selective fashion. Neuroscience 2003;121(2):509–21.
Davis CM, Moskovitz B, Nguyen MA, Tran BB, Arai A, Lynch G, et al. A profile of the behavioral changes produced by facilitation of AMPA-type glutamate receptors. Psychopharmacology (Berl) 1997;133(2):161–7.
Larson J, Quach CN, LeDuc BQ, Nguyen A, Rogers GA, Lynch G. Effects of an AMPA receptor modulator on methamphetamine-induced hyperactivity in rats. Brain Res 1996;738(2):353–6.
Johnson SA, Luu NT, Herbst TA, Knapp R, Lutz D, Arai A, et al. Synergistic interactions between ampakines and antipsychotic drugs. J Pharmacol Exp Ther 1999;289(1):392–7.
Li X, Tizzano JP, Griffey K, Clay M, Lindstrom T, Skolnick P. Antidepressant-like actions of an AMPA receptor potentiator (LY392098). Neuropharmacology 2001;40(8):1028–33.
Li X, Witkin JM, Need AB, Skolnick P. Enhancement of antidepressant potency by a potentiator of AMPA receptors. Cell Mol Neurobiol 2003;23(3):419–30.
Silverman JL, Oliver CF, Karras MN, Gastrell PT, Crawley JN. AMPAKINE enhancement of social interaction in the BTBR mouse model of autism. Neuropharmacology 2013;64:268–82.
Lauterborn JC, Truong GS, Baudry M, Bi X, Lynch G, Gall CM. Chronic elevation of brain-derived neurotrophic factor by ampakines. J Pharmacol Exp Ther 2003;307(1):297–305.
Hiscock KM, Linde JA, Einat H. Black Swiss mice as a new animal model for mania: a preliminary study. J Med Biol Sci 2007;1(2):1–6.
Flaisher-Grinberg S, Overgaard S, Einat H. Attenuation of high sweet solution preference by mood stabilizers: a possible mouse model for the increased reward-seeking domain of mania. J Neurosci Methods 2009;177(1):44–50.
Flaisher-Grinberg S, Einat H. A possible utilization of the mice forced swim test for modeling manic-like increase in vigor and goal-directed behavior. J Pharmacol Toxicol Methods 2009;59(3):141–5.
Hannah-Poquette C, Anderson GW, Flaisher-Grinberg S, Wang J, Meinerding TM, Einat H. Modeling mania: further validation for black swiss mice as model animals. Behav Brain Res 2011;223(1):222–6.
Flaisher-Grinberg S, Einat H. Strain specific battery of tests for separate behavioral domains of mania. Front Psychiatry 2010;1(Article 10):1–10.
Kalinichev M, Dawson LA. Evidence for antimanic efficacy of glycogen synthase kinase-3 (GSK-3) inhibitors in a strain specific model of acute mania. Int J Neuropsychopharmacol 2011;6:1–17.
Ene HM, Kara NZ, Einat H. The effects of the atypical antipsychotic asenapine in a strain-specific battery of tests for mania-like behaviors. Behav Pharmacol 2015 [e-pub ahead of print].
Einat H. Partial effects of the protein kinase C inhibitor chelerythrine in a battery of tests for manic-like behavior in black Swiss mice. Pharmacol Rep 2014;66(4):722–5.
Gould TD, Picchini AM, Einat H,Manji HK. Targeting glycogen synthase kinase-3 in the CNS: implications for the development of new treatments for mood disorders. Curr Drug Targets 2006;7(11):1399–409.
Zheng Y, Balabhadrapatruni S, Masumura C, Darlington CL, Smith PF. Effects of the putative cognitive-enhancing ampakine, CX717, on attention and object recognition memory. Curr Alzheimer Res 2011;8(8):876–82.
Staubli U, Rogers G, Lynch G. Facilitation of glutamate receptors enhances memory. Proc Natl Acad Sci U S A 1994;91(2):777–81.
Porrino LJ, Daunais JB, Rogers GA, Hampson RE, Deadwyler SA. Facilitation of task performance and removal of the effects of sleep deprivation by an ampakine (CX717) in nonhuman primates. PLoS Biol 2005;3(9):e299.
Knapp RJ, Goldenberg R, Shuck C, Cecil A, Watkins J, Miller C, et al. Antidepressant activity of memory-enhancing drugs in the reduction of submissive behavior model. Eur J Pharmacol 2002;440(1):27–35.
Simmons DA, Rex CS, Palmer L, Pandyarajan V, Fedulov V, Gall CM, et al. Upregulating BDNF with an ampakine rescues synaptic plasticity and memory in Huntington’s disease knockin mice. Proc Natl Acad Sci U S A 2009;106(12): 4906–11.
Bailey KR, Rustay NR, Crawley JN. Behavioral phenotyping of transgenic and knockout mice: practical concerns and potential pitfalls. ILAR J 2006; 47(2):124–31.
Crawley JN. Behavioral neuroscience. In: Crawley JN, McKay J, editors. Current Protocols in Neuroscience. New York: Wiley Interscience; 2007.
Einat H. Establishment of a battery of simple models for facets of bipolar disorder: a practical approach to achieve increased validity, better screening and possible insights into endophenotypes of disease. Behav Genet 2007;37(1):244–55.
Cryan JF, Holmes A. The ascent of mouse: advances in modelling human depression and anxiety. Nat Rev Drug Discov 2005;4(9):775–90.
Schmidt HD, Duman RS. The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol 2007;18(5–6):391–418.
Sanacora G, Zarate CA, Krystal JH, Manji HK. Targeting the glutamatergic system to develop novel, improved therapeutics for mood disorders. Nat Rev Drug Discov 2008;7(5):426–37.
Bachmann RF, Schloesser RJ, Gould TD, Manji HK. Mood stabilizers target cellular plasticity and resilience cascades: implications for the development of novel therapeutics. Mol Neurobiol 2005;32(2):173–202.
Juetten J, Einat H. Behavioral differences in black Swiss mice from separate colonies: implications for modeling domains of mania. Behav Pharmacol 2012;23(2):211–4.
Scotti MA, Lee G, Stevenson SA, Ostromecki AM, Wied TJ, Kula DJ, et al. Behavioral and pharmacological assessment of a potential new mouse model for mania. Physiol Behav 2011;103(3–4):376–83.
Fountoulakis KN, Kasper S, Andreassen O, Blier P, Okasha A, Severus E, et al. Efficacy of pharmacotherapy in bipolar disorder: a report by the WPA section on pharmacopsychiatry. Eur Arch Psychiatry Clin Neurosci 2012;262(Suppl. 1):1–48.
Gleason SD, Shannon HE. Blockade of phencyclidine-induced hyperlocomotion by olanzapine, clozapine and serotonin receptor subtype selective antagonists in mice. Psychopharmacology (Berl) 1997;129(1):79–84.
Granger R, Staubli U, Davis M, Perez Y, Nilsson L, Rogers GA, et al. A drug that facilitates glutamatergic transmission reduces exploratory activity and improves performance in a learning-dependent task. Synapse 1993;15(4): 326–9.
Boyle J, Stanley N, James LM, Wright N, Johnsen S, Arbon EL, et al. Acute sleep deprivation: the effects of the AMPAKINE compound CX717 on human cognitive performance, alertness and recovery sleep. J Psychopharmacol 2012; 26(8):1047–57.
Sadock JB, Sadock VA. Kaplan and Sadock’s Synopsis of Psychiatry. 10th ed. Philadelphia: Lippincott & Williams; 2007.
Porsolt RD, Bertin A, Jalfre M. ‘‘Behavioural despair’’ in rats and mice: strain differences and the effects of imipramine. Eur J Pharmacol 1978;51(3): 291–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kara, N.Z., Flaisher-Grinberg, S. & Einat, H. Partial effects of the AMPAkine CX717 in a strain specific battery of tests for manic-like behavior in black Swiss mice. Pharmacol. Rep 67, 928–933 (2015). https://doi.org/10.1016/j.pharep.2015.02.008
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.02.008