Abstract
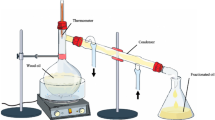
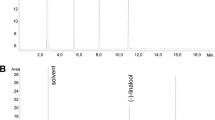
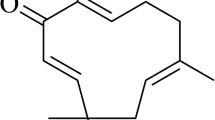
Ageratum fastigiatum (Gardner) R.M. King & H. Rob., a member of the Asteraceae family popularly known in Brazil as “matapasto”, is indicated in folk medicine as anti-inflammatory and analgesic. Despite its popular use, little is known about its potential effect on the parameters involved in an inflammatory response. The objective of this study was to characterize the chemical composition of the essential oil from A. fastigiatum and to evaluate the frequency of tumor necrosis factor alpha and interferon gamma producing cells in peripheral blood lymphocytes stimulated with phorbol myristate acetate in the presence of essential oil from A. fastigiatum. Non-toxic concentrations of essential oil from A. fastigiatum were evaluated in cultures of peripheral blood leucocytes using the trypan blue exclusion assay by flow cytometry. GC–MS analysis revealed that the prevalent compounds identified in the essential oil from A. fastigiatum sample were α-pinene, limonene, trans-caryophyllene, α-humulene, caryophyllene oxide, 1,2-humulene-epoxide, 1,6-humulanodien-3-ol, and α-cadinol. Results showed that exposure to essential oil from A. fastigiatum at concentrations of 0.5 × 10−2 and 1 × 10−2 μl/ml caused no alterations in leukocyte viability as compared to the control group. Both concentrations lowered the percentage of tumor necrosis factor alpha (+)-lymphocytes and neutrophils. There were no changes in the percentage of lymphocytes positive for the interferon gamma cytokine. Our results suggest that part of the anti-inflammatory activity attributed to A. fastigiatum may be due to the effect of some of its components in decreasing the number of cells that produce the pro-inflammatory cytokine tumor necrosis factor alpha.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams, R.P., 1995. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy. Allured Publishing Corporation, Illinois.
Alasandagutti, M.L., Ponnana, M., Sivangala, R., Thada, S., Joshi, L., Hussain, H., Ansari, S.S., Valluri, V., Gaddam, S.L., 2014. Role of IFN-ψ and IL-6 cytokines and their association in determining susceptibility to chronic idiopathic urticaria. Genet. Test Mol. Biomarkers 18, 804–809.
Amaldi, I., Reith, W., Berte, C., Mach, B., 1989. Induction of HLA class II genes by IFNgamma is transcriptional and requires a trans-acting protein. J. Immunol. 142, 999–1004.
Apostolaki, M., Armaka, M., Victoratos, P., Kollias, G., 2010. Cellular mechanisms of TNF function in models of inflammation and autoimmunity. Curr. Dir. Autoimmun. 11, 1–26.
Avelar-Freitas, B.A., Almeida, V.G., Pinto, M.C., Mourão, F.A., Massensini, A.R., Martins-Filho, O.A., Rocha-Vieira, E., Brito-Melo, G.E., 2014. Trypan blue exclusion assay by flow cytometry. Braz. J. Med. Biol. Res. 47, 307–315.
Bae, G.S., Park, K.C., Choi, S.B., Jo, I.J., Choi, M.O., Hong, S.H., Song, K., Song, H.J., Park, S.J., 2012. Protective effects of alpha-pinene in mice with cerulein-induced acute pancreatitis. Life Sci. 17–18, 866–871.
Bibikova, E., Youn, M.Y., Danilova, N., Ono-Uruga, Y., Konto-Ghiorghi, Y., Ochoa, R., Narla, A., Glader, B., Lin, S., Sakamoto, K.M., 2014. TNF-α mediated inflammation represses GATA1 and activates p38 MAP kinase in RPS19-deficient hematopoieticprogenitors. Blood 124, 3791–3798.
Bishayi, B., Samanta, A.K., 2002. Modulation of interleukin-8 receptor expression by lipopolysaccharide (LPS) and phorbol myristate acetate (PMA) in human peripheral monocytes–a preliminary study. Indian J. Physiol. Pharmacol. 46, 407–422.
Braegger, C.P., Nicholls, S., Murch, S.H., Stephens, S., MacDonald, T.T., 1992. Tumor necrosis factor alpha in stool as marker of intestinal inflammation. Lancet 339, 89–91.
Chavan, M.J., Wakte, P.S., Shinde, D.B., 2010. Analgesic and anti inflammatory activity of Caryophyllene oxide from Annona squamosa L. bark. Phytomedicine 17, 149–151.
Del-Vechio-Vieira, G., Sousa, O.V., Miranda, M.A., Senna-Valle, L., Kaplan, M.A.C., 2009. Analgesic and anti-inflammatory properties of essential oil from Ageratum fastigiatum. Braz. Arch. Biol. Technol. 52, 1115–1121.
Del-Vechio-Vieira, G., Sousa, O.V., Yamamoto, C.H., Kaplan, M.A.C., 2009. Chemical composition and antimicrobial activity of the essential oils of Ageratum fastigiatum (Asteraceae). Rec. Nat. Prod. 3, 52–57.
Fernandes, E.S., Passos, G.F., Medeiros, R., da Cunha, F.M., Ferreira, J., Campos, M.M., Pianowski, L.F., Calixto, J.B., 2007. Anti-inflammatory effects of compounds alpha-humulene and (−)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur. J. Pharmacol. 569, 228–236.
Gobbo-Neto, L., Lopes, N.P., 2007. Plantas medicinais: fatores de influência no conteúdo de metabólitos secundários. Quim. Nova 30, 374–381.
Gonçalves, L.D., Almeida, H.R., Oliveira, P.M., Lopes, N.P., Turatti, I.C.C., Archanjo, F.C., Grael, C.F.F., 2011. Contribution for the phytochemical studies of Ageratum fastigiatum. Rev. Bras. Farmacogn. 21, 936–942.
Hirota, R., Roger, N.N., Nakamura, H., Song, H.S., Sawamura, M., Suganuma, N., 2010. Anti-inflammatory effects of limonene from yuzu (Citrus junos Tanaka) essential oil on eosinophils. J. Food Sci. 75, H87–H92.
Issekutz, A.C., 1981. Vascular responses during acute neutrophilic inflammation. Their relationship to in vivo neutrophil emigration. Lab. Invest. 45, 435–441.
Kees, U., Kaltmann, B., Marcucci, F., Hültner, L., Staber, F., Krammer, P.H., 1984. Frequency and activity of immune interferon (IFN-gamma) and colony-stimulating factor-producing human peripheral blood T lymphocytes. Eur. J. Immunol. 14, 368–373.
Lima, H.R.P., Kaplan, M.A.C., Cruz, A.V.M., 2003. Influência dos fatores abióticos na produção e variabilidade de terpenóides em plantas. Floresta Ambient. 10, 71–77.
Medeiros, R., Passos, G.F., Vitor, C.E., Koepp, J., Mazzuco, T.L., Pianowski, L.F., Campos, M.M., Calixto, J.B., 2007. Effect of two active compounds obtained from the essential oil of Cordia verbenacea on the acute inflammatory responses elicited by LPS in the rat paw. Br. J. Pharmacol. 151, 618–627.
Sedger, L.M., McDermott, M.F., 2014. TNF and TNF-receptors: from mediators of cell death and inflammation to therapeutic giants–past, present and future. Cytokine Growth Factor Rev. 25, 453–472.
Taylor, A., Tang, W., Bruscia, E.M., Zhang, P.X., Lin, A., Gaines, P., Wu, D., Halene, S., 2014. SRF is required for neutrophil migration in response to inflammation. Blood 123, 3027–3036.
Van Den Dool, H., Kratz, P.D., 1963. A generalization of the retention index system including linear temperature programmed gas–liquid partition chromatography. J. Chromatogr. 11, 463–471.
Wajant, H., 2009. The role of TNF in cancer. Results Probl. Cell Differ. 49, 1–15.
Wilderman, M.J., Kim, S., Gillespie, C.T., Sun, J., Kapoor, V., Vachani, A., Sterman, D.H., Kaiser, L.R., Albelda, S.M., 2006. Blockade of TNF alpha decreases both inflammation and efficacy of intrapulmonary Ad.IFNbeta immunotherapy in an orthotopic model of bronchogenic lung cancer. Mol. Ther. 13, 910–917.
Yoon, W.J., Lee, N.H., Hyun, C.G., 2010. Limonene suppresses lipopolysaccharideinduced production of nitric oxide, prostaglandin E2, and pro-inflammatory cytokines in RAW 264.7 macrophages. J. Oleo Sci. 59, 415–421.
Acknowledgments
We are grateful to the Brazilian funding agencies FAPEMIG (APQ-01626-13) and CNPq (Processo n. 476367/2011-5) and CAPES for financial support, as well as to the Multicentric Post-graduate Program in Physiological Science and Center for Research in Natural and Synthetic Products, Faculty of Pharmaceutical Sciences of Ribeirão Preto, University of São Paulo for providing infrastructure to perform the analysis in GC-MS.
Author information
Authors and Affiliations
Contributions
BAA (PhD student) contributed in collecting plant sample and identification, confection of herbarium, running the laboratory work, analyzing the data and drafted the paper and contributed to biological studies. VGA, MGS and JATS contributed to biological studies. PRB, CFFG and LEG contributed in plant identification and herbarium confection and to chromatographic analysis. ERV and GEBAM designed the study, supervised the laboratory work and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Avelar-Freitas, B.A., Almeida, V.G., Santos, M.G. et al. Essential oil from Ageratum fastigiatum reduces expression of the pro-inflammatory cytokine tumor necrosis factor-alpha in peripheral blood leukocytes subjected to in vitro stimulation with phorbol myristate acetate. Rev. Bras. Farmacogn. 25, 129–133 (2015). https://doi.org/10.1016/j.bjp.2015.03.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.03.002