Abstract
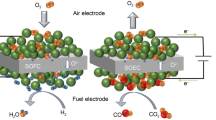
Sr-Co containing perovskite oxides are prospective air electrode candidates for reversible solid oxide cells (RSOCs). However, their efficiencies are limited by Sr segregation and the high thermal expansion coefficient (TEC) of Co-based perovskites. Herein, La0.6Ca0.4Fe08Ni0.2O3−δ (LCaFN) is tailored as an Sr-Co-free perovskite air electrode for highperformance RSOCs. Compared with La0.6Sr0.4Fe0.8Ni0.2O3−δ (LSFN) and La0.6Sr0.4Co0.2Fe0.8O3−δ (LSCoF), LCaFN has a high electrical conductivity (297 S cm−1), TEC compatibility (11.2 × 10−6 K−1) and improved chemical stability. Moreover, LCaFN has high oxygen reduction reaction (ORR) activity with a low polarization resistance (0.06 Ω cm2) at 800°C. A single-cell Ni-YSZ/YSZ/gadolinium-doped ceria (GDC)/LCaFN-GDC operated at 800°C yields a maximum power density of 1.08 W cm−2 using H2 as fuel. In the solid oxide electrolysis cell (SOEC) mode, the cell can achieve a current density of approximately 1.2 A cm−2 at 1.3 V with 70% humidity at 800°C. The cell exhibits good reversibility and remains stable in continuous SOEC and solid oxide fuel cell (SOFC) modes. These findings indicate the potential application of LCaFN as an air electrode material for RSOCs.

摘要
可逆固体氧化物燃料电池 (RSOC) 是一种新型高效的能量存储和转化装置, 具有高效率、 无污染和模块化等优点. 在本文中, La0.6Ca0.4Fe0.8Ni0.2O3−δ (LCaFN) 被用于高性能RSOC的无锶无钴钙钛矿空气电极. 与La0.6Sr0.4Fe0.8Ni0.2O3−δ (LSFN) 和La0.6Sr0.4Co0.2Fe0.8-O3−δ (LSCoF) 相比, LCaFN 具有较高的导电性 (297 S cm−1)、 良好的热膨胀系数兼容性 (11.2 × 10−6 K−1) 和较高的化学稳定性. 此外, LCaFN 在 800°C还具有高催化活性和低极化电阻 (0.06 Ω cm2). 单电池 Ni-YSZ/YSZ/GDC/LCaFN-GDC在800°C下的最大功率密度为1.08 W cm−2. 在固体氧化物电解池模式下, 电池在800°C、70% H2O、1.3 V电压下可实现约 1.2 A cm−2的电流密度. 同时电池还表现出良好的可逆性和运行稳定性. 研究结果表明, LCaFN作为RSOC的空气电极材料具有广阔的应用前景.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Duan C, Kee R, Zhu H, et al. Highly efficient reversible protonic ceramic electrochemical cells for power generation and fuel production. Nat Energy, 2019, 4: 230–240
Zheng Y, Wang J, Yu B, et al. A review of high temperature co-electrolysis of H2O and CO2 to produce sustainable fuels using solid oxide electrolysis cells (SOECs): Advanced materials and technology. Chem Soc Rev, 2017, 46: 1427–1463
Choi S, Davenport TC, Haile SM. Protonic ceramic electrochemical cells for hydrogen production and electricity generation: Exceptional reversibility, stability, and demonstrated faradaic efficiency. Energy Environ Sci, 2019, 12: 206–215
Graves C, Ebbesen SD, Jensen SH, et al. Eliminating degradation in solid oxide electrochemical cells by reversible operation. Nat Mater, 2015, 14: 239–244
Ai N, Li N, He S, et al. Highly active and stable Er0.4Bi1.6O3 decorated La0.76Sr0.19MnO3+δ nanostructured oxygen electrodes for reversible solid oxide cells. J Mater Chem A, 2017, 5: 12149–12157
Shimada H, Fujimaki Y, Fujishiro Y. Highly active and durable La0.4Sr0.6MnO3−δ and Ce0.8Gd0.2O1.9 nanocomposite electrode for high-temperature reversible solid oxide electrochemical cells. Ceramics Int, 2020, 46: 19617–19623
Aguadero A, Pérez-Coll D, Alonso JA, et al. A new family of Modoped SrCoO3−δ perovskites for application in reversible solid state electrochemical cells. Chem Mater, 2012, 24: 2655–2663
Hong WT, Risch M, Stoerzinger KA, et al. Toward the rational design of non-precious transition metal oxides for oxygen electrocatalysis. Energy Environ Sci, 2015, 8: 1404–1427
Guan F, Zhang X, Song Y, et al. Effect of Gd0.2Ce0.8O1.9 nanoparticles on the oxygen evolution reaction of La0.6Sr0.4Co0.2Fe0.8O3−δ anode in solid oxide electrolysis cell. Chin J Catal, 2018, 39: 1484–1492
Tan Y, Duan N, Wang A, et al. Performance enhancement of solution impregnated nanostructured La0.8Sr0.2Co0.8Ni0.2O3−δ oxygen electrode for intermediate temperature solid oxide electrolysis cells. J Power Sources, 2016, 305: 168–174
López-Robledo MJ, Laguna-Bercero MA, Larrea A, et al. Reversible operation of microtubular solid oxide cells using La0.6Sr0.4Co0.2-Fe0.8O3−δ-Ce0.9Gd0.1O2−δ oxygen electrodes. J Power Sources, 2018, 378: 184–189
Fu Y-P, Subardi A, Hsieh M-Y, et al. Electrochemical properties of La0.5Sr0.5Co0.8M0.2O3−δ (M = Mn, Fe, Ni, Cu) perovskite cathodes for IT-SOFCs. J Am Ceram Soc, 2016, 99: 1345–1352
Majnoni d’Intignano X, Cademartori D, Clematis D, et al. Infiltrated Ba0.5Sr0.5Co0.8Fe0.2O3−δ-based electrodes as anodes in solid oxide electrolysis cells. Energies, 2020, 13: 3659
Zhu L, Wei B, Wang Z, et al. Electrochemically driven deactivation and recovery in PrBaCo2O5+δ oxygen electrodes for reversible solid oxide fuel cells. ChemSusChem, 2016, 9: 2443–2450
Ebbesen SD, Jensen SH, Hauch A, et al. High temperature electrolysis in alkaline cells, solid proton conducting cells, and solid oxide cells. Chem Rev, 2014, 114: 10697–10734
Laguna-Bercero MA. Recent advances in high temperature electrolysis using solid oxide fuel cells: A review. J Power Sources, 2012, 203: 4–16
Zhou Q, Cheng Y, Li W, et al. Investigation of cobalt-free perovskite Sr2FeTi0.75Mo0.25O6−δ as new cathode for solid oxide fuel cells. Mater Res Bull, 2016, 74: 129–133
Tian Y, Zhang L, Jia L, et al. Novel quasi-symmetrical solid oxide electrolysis cells with in-situ exsolved cathode for CO2 electrolysis. J CO2 Util, 2019, 31: 43–50
Bian LZ, Wang LJ, Chen N, et al. Enhanced performance of La0.7Sr0.3Fe0.9Ni0.1O3 cathode by partial substitution with Ce. Ceramics Int, 2017, 43: 982–987
Tian Y, Zhang L, Liu Y, et al. A self-recovering robust electrode for highly efficient CO2 electrolysis in symmetrical solid oxide electrolysis cells J Mater Chem A, 2019, 7: 6395–6400
Tian Y, Zheng H, Zhang L, et al. Direct electrolysis of CO2 in symmetrical solid oxide electrolysis cell based on La0.6Sr0.4Fe0.8-Ni0.2O3−δ electrode. J Electrochem Soc, 2018, 165: F17–F23
Meng L, Wang F, Wang A, et al. High performance La0.8Sr0.2MnO3-coated Ba0.5Sr0.5Co0.8Fe0.2O3 cathode prepared by a novel solid-solution method for intermediate temperature solid oxide fuel cells Chin J Catal, 2014, 35: 38–42
Sumi H, Ohshiro T, Nakayama M, et al. Prevention of reaction between (Ba,Sr)(Co,Fe)O3 cathodes and yttria-stabilized zirconica electrolytes for intermediate-temperature solid oxide fuel cells Electrochim Acta, 2015, 184: 403–409
Konysheva E, Penkalla H, Wessel E, et al. Chromium poisoning of perovskite cathodes by the ODS alloy Cr5Fe1Y2O3 and the high chromium ferritic steel Crofer22APU. J Electrochem Soc, 2006, 153: A765
Kim YM, Chen X, Jiang SP, et al. Effect of strontium content on chromium deposition and poisoning in Ba1−xSrxCo0.8Fe0.2O3−δ (0.3 ≤ x ≤ 0.7) cathodes of solid oxide fuel cells. J Electrochem Soc, 2011, 159: B185–B194
Tian Y, Wang W, Liu Y, et al. Cobalt-free perovskite oxide La0.6Sr0.4Fe0.8Ni0.2O3−δ as active and robust oxygen electrode for reversible solid oxide cells. ACS Appl Energy Mater, 2019, 2: 3297–3305
Muhammed Ali SA, Anwar M, Somalu MR, et al. Enhancement of the interfacial polarization resistance of La0.6Sr0.4Co0.2Fe0.8O3−δ cathode by microwave-assisted combustion method Ceramics Int, 2017, 43: 4647–4654
Stevenson JW, Armstrong TR, Carneim RD, et al. Electrochemical properties of mixed conducting perovskites La1−xMxCo1−yFeyO3−δ (M = Sr, Ba, Ca). J Electrochem Soc, 1996, 143: 2722–2729
Huheey JE, Keiter EA, Keiter RL. Inorganic Chemistry: Principles of Structure and Reactivity. New York: Harper Collins, 1993
Whangbo MH, Koo HJ, Villesuzanne A, et al. Effect of metal-oxygen covalent bonding on the competition between jahn-teller distortion and charge disproportionation in the perovskites of high-spin d4 metal ions LaMnO3 and CaFeO3. Inorg Chem, 2002, 41: 1920–1929
Swierczek K Thermoanalysis, nonstoichiometry and thermal expansion of La0.4Sr0.6Co0.2Fe0.8O3−δ, La0.2Sr0.8Co0.2Fe0.8O3−δ, La0.9Sr0.1-Co1/3Fe1/3Ni1/3O3−δ and La0.6Sr0.4Co0.2Fe0.6Ni0.2O3−δ perovskites. Solid State Ion, 2008, 179: 126–130
Hayashi H, Kanoh M, Quan CJ, et al. Thermal expansion of Gddoped ceria and reduced ceria Solid State Ion, 2000, 132: 227–233
Yang E, Noh Y, Ramesh S, et al. The effect of promoters in La0.9M0.1Ni0.5Fe0.5O3 (M = Sr, Ca) perovskite catalysts on dry reforming of methane. Fuel Process Technol, 2015, 134: 404–413
Sun YF, Li JH, Cui L, et al. A-site-deficiency facilitated in situ growth of bimetallic Ni-Fe nano-alloys: A novel coking-tolerant fuel cell anode catalyst. Nanoscale, 2015, 7: 11173–11181
Natile MM, Poletto F, Galenda A, et al. La0.6Sr0.4Co1−yFeyO3−δ perovskites: Influence of the Co/Fe atomic ratio on properties and catalytic activity toward alcohol steam-reforming. Chem Mater, 2008, 20: 2314–2327
Huang TJ, Shen XD, Chou CL. Characterization of Cu, Ag and Pt added La0.6Sr0.4Co0.2Fe0.8O3−δ and gadolinia-doped ceria as solid oxide fuel cell electrodes by temperature-programmed techniques. J Power Sources, 2009, 187: 348–355
Zhou F, Zhou L, Hu M, et al. Pd-doped La0.6Sr0.4Co0.2Fe0.8O3−δ perovskite oxides as cathodes for intermediate temperature solid oxide fuel cells Solid State Ion, 2018, 319: 22–27
Liu J, Barnett SA. Thin yttrium-stabilized zirconia electrolyte solid oxide fuel cells by centrifugal casting. J Am Ceramic Soc, 2004, 85: 3096–3098
Abdalla AM, Hossain S, Zhou J, et al. NdBaMn2O5+δ layered perovskite as an active cathode material for solid oxide fuel cells Ceramics Int, 2017, 43: 15932–15938
Zhang L, Li D, Zhang S. One-step synthesized Co1.5Mn1.5O4-Ce0.8Sm0.2O1.9 composite cathode material for intermediate temperature solid oxide fuel cells Ceramics Int, 2017, 43: 2859–2863
Wang JX, Sun JL, He CR, et al. Mass synthesis of high performance (La0.75Sr0.25)0.95MnO3±δ nano-powder prepared via a low-carbon chemical solution method J Power Sources, 2014, 253: 424–430
Wang W, Zhang X, Zhang D, et al. Highly promoted performance of triple-conducting cathode for YSZ-based SOFC via fluorine anion doping. Ceramics Int, 2020, 46: 23964–23971
Salehzadeh D, Torabi M, Sadeghian Z, et al. A multiscale-architecture solid oxide fuel cell fabricated by electrophoretic deposition technique. J Alloys Compd, 2020, 830: 154654
Li P, Duan N, Ma J, et al. Exploring MnCr2O4-Gd0.1Ce0.9O2−δ as a composite electrode material for solid oxide fuel cell Int J Hydrogen Energy, 2019, 44: 31333–31341
Guan C, Wang Y, Chen K, et al. Molten salt synthesis of Nb-doped (La, Sr)FeO3 as the oxygen electrode for reversible solid oxide cells Mater Lett, 2019, 245: 114–117
Fan H, Keane M, Singh P, et al. Electrochemical performance and stability of lanthanum strontium cobalt ferrite oxygen electrode with gadolinia doped ceria barrier layer for reversible solid oxide fuel cell J Power Sources, 2014, 268: 634–639
Fan H, Keane M, Li N, et al. Electrochemical stability of La0.6Sr0.4Co0.2Fe0.8O3−δ-infiltrated YSZ oxygen electrode for reversible solid oxide fuel cells Int J Hydrogen Energ, 2014, 39: 14071–14078
Tian Y, Liu Y, Wang W, et al. High performance and stability of double perovskite-type oxide NdBa0.5Ca0.5Co1.5Fe0.5O5+δ as an oxygen electrode for reversible solid oxide electrochemical cell J Energy Chem, 2020, 43: 108–115
Kobayashi K, Suzuki TS. Distribution of relaxation time analysis for non-ideal immittance spectrum: Discussion and progress. J Phys Soc Jpn, 2018, 87: 094002
Yan J, Chen H, Dogdibegovic E, et al. High-efficiency intermediate temperature solid oxide electrolyzer cells for the conversion of carbon dioxide to fuels J Power Sources, 2014, 252: 79–84
Acknowledgements
This work was supported by the National Key Research & Development Program of China (2020YFB1506304, 2017YFE0129300 and 2016YFE0126900), the National Natural Science Foundation of China (52072135, 51672095 and U1910209), and the Major Science and Technology Innovation Project of Hubei Province (2018AAA057). Analytical and Testing Centre of Huazhong University of Science and Technology is appreciated for sample characterization assistance.
Author information
Authors and Affiliations
Contributions
Wang W conducted the experiments and wrote this paper; Tian Y gave the overall concept and revised the draft; Abhishek N conducted the data analysis; Liu Y and Li Y contributed to the theoretical analysis; Chi B was responsible for review and editing; Pu J contributed to resource support. All authors participated in the general discussion.
Corresponding authors
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Wenjie Wang received her Bachelor degree in engineering from China University of Geosciences (Wuhan). She is currently studying for a Master’s degree under the supervision of Professor Bo Chi at Huazhong University of Science and Technology (HUST). Her research interests include electrode materials for electrochemical energy storage devices, such as fuel cells.
Yunfeng Tian is currently a lecturer in the School of Materials Science and Physics at China University of Mining and Technology. He received his PhD degree from HUST, under the supervision of Professor Bo Chi. His research focuses on the development and characterization of novel electrode materials for solid oxide fuel cells and electrolysis cells.
Bo Chi is a professor of the Center for Fuel Cell Innovation, School of Materials Science and Technology at HUST, China. He received his BSc and Master degrees in materials science from China University of Geosciences (Wuhan) in 1998 and 2001, respectively, and his PhD degree in materials science from Tsinghua University in 2005. He worked as JSPS (The Japan Society for the Promotion of Science) postdoc at the National Institute of Advanced Industrial Science and Technology (AIST), Japan from 2005 to 2007 before he joined HUST. His current research interests are solid oxide fuel cells/electrolysis cells, metal-air battery and solar cells.
Rights and permissions
About this article
Cite this article
Wang, W., Tian, Y., Liu, Y. et al. Tailored Sr-Co-free perovskite oxide as an air electrode for high-performance reversible solid oxide cells. Sci. China Mater. 64, 1621–1631 (2021). https://doi.org/10.1007/s40843-020-1567-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-020-1567-2