Abstract
Recent Findings
Evidences suggest the pathophysiological role of GABA, glutamate, inflammatory cascade, hypoxia, etc. in the development of status epilepticus (SE). But the exact understanding of SE seizure is still unknown.
Purpose of Review
SE is a lethal neurological disorder that is associated with elevated rates of mortality and morbidity. SE is demarcated by recurring seizures caused by a failure of mechanisms responsible for the termination of seizures or from the initiation of mechanisms, which lead to abnormally, persistent seizures after time point (t1). Long-term consequences are spotted in this condition after time point (t2), together with the death of neurons, neuronal injury, and alteration of neuronal networks, depending on the type and duration of seizures. Clinically first-line treatment with benzodiazepines is given for seizure inhibition. But in severe cases, these drugs are not efficient for treatment. The unavailability of viable medications to halt the onset and progression of the condition could be due to a lack of understanding of the pathophysiological mechanism. Due to the complexity of the brain, it is very difficult to understand exact pathways; thorough understanding of pathophysiology would be beneficial in designing new drug regimen for patients.
Summary
This review paper emphasizes on pathophysiology, etiology, diagnostic parameters, and drugs used in the secession of seizures promptly, which would be helpful for the development of effective therapeutics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
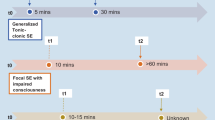
Status epilepticus (SE) is a serious medical condition where the seizures are prolonged and need medical intervention, resulting in long-lasting consequences and fatalities if not treated at the time. The International League Against Epilepsy (ILAE) has changed the concepts, definition, and classification of SE. The newly emerged definition of SE is as follows: SE is known to be tenacious or recurring seizures caused by a failure of mechanisms responsible for the termination of seizures or from the initiation of mechanisms, which lead to abnormally prolonged seizures (after first time point which is considered t1). Long-term consequences are seen in this condition (after second time point which is considered t2), including the death of neurons, neuronal injury, and alteration of neuronal networks, depending on the type and duration of seizures [1]. In the case of CSE (tonic-clonic), both time points (t1 at 5 min and t2 at 30 min) are based on animal experiments and clinical research. The occurrences of SE in the elderly population and the neonates are frequently observed. SE annually covers approximately 200,000 cases in the USA and affects almost every age group [2]. The time varies for a different state of seizure as in the case of CSE (5 min), FSE (10 min), and absent SE, predominantly recognized as NCSE (10–15 min) [3]. CSE has disastrous results as it permanently causes damage to neurons after 30 min of its commencement. In the case of bilateral tonic-clonic seizures, 5 min of continuous seizure activity constitutes status, and prolonged impacts of disorder commence at 30 min of status [4]. If seizure activity continues after giving first-line treatment with benzodiazepines, it comes under ESE, which can be treated using second-line medications. The type of SE that lasts after first-line and second-line drugs, demarcated as RSE and SRSE, is a seizure activity that continues despite anesthetic therapy or the repetition of seizure activity with the secession of anesthetic therapy [5, 6]. The prevalence of SE in the population has been reported, and the data ranges from nearly 5 to 40 per 100,000 obtained from various studies done in population throughout the US, Asian, and European countries through a current meta-analysis reporting incidence of 12.6 per 100,000 annually. There is no vital distinction in the incidence of SE in males and females, yet the incidence in aged persons is 27.1 per 100,000 annually that is around four times that of non-geriatric adults. The patients who survive from SE have allied mental and neurological deficits, and significantly it is a risk factor in those without epilepsy, as they have chances of progression of epilepsy for a prolonged duration [7, 8]. The prevalence of SE is more in the population having structural brain damage, and the patients already having epilepsy have chances of triggering epilepsy with the withdrawal of certain drugs. The SRSE and RSE are highly severe, importunate forms of SE; besides, the percentage of RSE in SE cases is 29 to 43%; correspondingly the percentage of SRSE in cases of SE is 12 to 26% [9,10,11]. In both developed and developing nations, there is certainly not any distinction in the incidence of SE.
Etiology of Status Epilepticus
Status epilepticus is a highly vigorous and deadly neurological entity, and its pathophysiology is still not very clear. SE is generally an advent of epilepsy that is already present in patients, and significant causative factors include chronic epilepsy in patients with low blood concentrations of AEDs (34%), a multitude of cerebral attacks such as remote symptomatic causes (24%), injury to the brain (22%) [12], anoxia or hypoxia (10%), metabolic disorders (10%), and due to withdrawal of alcohol or because of drug withdrawal (10%) (Fig. 1). Some drugs are reported in the study that can cause SE, and there may be chances of SE occurrence from certain drugs, which are responsible for the excitation of the CNS. For instance, glutamate agonists or those having a property of reducing inhibition such as GABA antagonists are seen in clinical practice. SE can also be caused by AEDs, psychotropic medications. An example of an AED responsible for causing SE is the drug levetiracetam-induced SE, which is mainly associated with high or rapidly intensifying doses, with possible causation. Several antibiotics can induce SE; also, 50% of the reports having cases of neurotoxicity involve seizures, which are associated with cephalosporins, beta-lactams, isoniazid, quinolones, and theophylline [13, 14].
Pathophysiology of Status Epilepticus
In normal conditions, the possibility of seizure occurrence is less, as long as neuronal stability is maintained and inhibits synchronous discharges of neurons that instigate a seizure. Various significant mechanisms that occur typically as a part of the regular synaptic information transfer permit only a single action potential to pass in a time interval from one neuron to the next neuron. If certain circumstances disrupt the stability of a healthy brain, it leads to incitement of seizure activity; for instance, abnormal levels of ions as in the case of hyponatremia result in a loss of the standard electrochemical gradients across cell membranes required for maintaining the stability of the brain. Withdrawal of certain drugs like benzodiazepines and barbiturates bring about sensitization of inhibitory GABA-A receptors so that neuronal activity that is generally considered innocuous can trigger the seizure activity. A thorough understanding of the actions of the GABA and NMDA receptors provides improvements in pharmacologic methods for the management of SE. Variations in the location and subunit composition of these neurotransmitter receptors occur in SE, which is clear from the pathophysiology of SE, and by these variations, self-enduring seizure activity is promoted. As SE persists, the efficiency of drugs that are used for the treatment of the initial phases of SE eventually decreases. Phasic inhibitory currents are generated in response to the vesicular release of GABA through synaptic GABA-A receptors. There is a wide distribution of these receptors in the brain, and benzodiazepines target these receptors and get internalized in the course of SE decreasing inhibition [15, 16]. Excitation increases during SE due to the accretion of glutamate receptors (mainly NMDA) in the synaptic membrane [17]. In comparison to the extensive expression of synaptic GABAA receptors, extrasynaptic GABAA receptors only exist in limited parts of the brain like the hippocampus, thalamus, amygdala, cerebellum, and hypothalamus. Non-desensitizing tonic inhibitory currents are produced in retort to extracellular GABA by the activation of these receptors. However, benzodiazepines do not target these receptors; besides, they are assumed to denote a favorable, novel medicinal target. The binding properties of GABA, in addition to glutamate receptors, are regulated by the subunit composition of these receptors [18]. During the development of the brain, as a retort to seizures, there may be changes in the composition of these receptors. In the immature brain or the undeveloped brain exposed to continuous seizure activity, the composition of AMPA, NMDA, and GABAA receptors (increased ratio of non-α1/α1 in GABAA receptors, GluN2B/GluN2A in NMDA receptors, and GluA1/GluA2 in AMPA receptors) leads to further excitability besides promoting self-sustaining seizures [19,20,21,22]. Furthermore, in the immature brain GABAergic neurotransmission is excitatory in nature stated that overexpression of the chlorine NKCC1 transporter helps in promoting aggregation of chlorine intracellularly [23]. Altogether, the immature brain becomes vulnerable to seizures and is susceptible to the progression of abnormal neuronal networks in the course of cortical formation and synaptogenesis due to the enhanced excitability of neurons [24, 25]. The mechanisms of epileptogenesis are affected by a disturbance in the blood-brain barrier [26]. The invasion of cells and molecules, such as leukocytes and albumin, perhaps happens due to severe neurological attacks or by extended epileptic activity, or by a compromise of the blood-brain barrier. Astrocytes activated by albumin, together with the induction of calcium signaling [27], enhance the generation of the pro-convulsant cytokine IL-1β [28]. The excitability of astrocytes, increased by the subsequent uptake of albumin, has been proposed as a novel mechanism of epileptogenesis and is related to TGF β-signaling [29]. Attacking cells such as leukocytes and other cells present in the brain (for instance, activated microglia and astrocytes, neurons, endothelial cells of the blood-brain barrier, and blood-borne macrophages) have the capability of synthesizing and releasing substances that have inflammatory properties. This property results in an inflammatory cascade, which can reduce the threshold for seizures in response to several factors such as infections, autoimmune diseases, and seizures [30]. These inflammatory substances are responsible for actuating neuromodulators, generating seizures, epileptogenesis, and resistance caused by drugs [31, 32]. An additional reason for AED resistance is an altered expression of specific transport proteins or drug-metabolizing enzymes in the blood-brain barrier [33, 34]. A blood-brain barrier that does not function properly can boost or lessen the distribution of the AED into the brain. The changes in the composition and location of specific neurotransmitter receptors, neuroinflammation, and the blood-brain barrier are considered the targets of immense potential for new-fangled drugs used in epilepsy, along with processes responsible for pharmacy resistance to current drugs used in the treatment [35].
Diagnostic Parameters of SE
Finding out the etiology is an integral part of the SE treatment, and the most challenging aspect is the management of SE. In maximum cases, there is a known etiology of SE, and the most common causes are intracranial lesions, infections, and previous seizures. Diagnosis of SE can be done by various parameters like head CT scan, fingerstick glucose test, monitoring of vital signs followed by laboratory tests such as total blood count, and fundamental metabolic panel by determining levels of AEDs, calcium, and magnesium. Monitoring of cEEG is mandatory for all patients who are incapable of returning to a normal phase. Other examining parameters include brain MRI and comprehensive toxicology which includes various symptoms like movement disorders, ischemic attacks, shaking of limbs, infections of the NSS, psychiatric disorders, sleep disorders, and hypoglycemia [36]. When a patient arrives in the hospital, within minutes, he/she is initially investigated for venous blood, not only for determining electrolytes but also for the glucose level, liver function tests, AED levels, complete blood count, and toxicological screens or levels of other drugs. Then, followed with the head CT as soon as the patient gets stabilized to observe structural lesion(s), or presence of intracranial lesions in acute form like hematomas and hemorrhages that may require emergent intervention. For patients suffering from fever and abrupt onset of the altered status of mental health, the lower threshold is required for performing the lumbar puncture to eliminate the common infections exclusively for herpes encephalitis. An abrupt EEG must be contemplated in the prolonged seizure cases and also an examination for NCSE when a patient takes longer than expected to get back to the normal stage [37]. SE is detected and subsequently managed using EEG. The SE diagnosis by EEG criteria includes electrographic seizures on a frequent repetitive basis and repetitive focal or generalized discharges of epileptiform of more than 3Hz. Periodic or repetitive discharges of epileptiform lower than 3 Hz may be contemplated as ictal if it is accompanied by a better clinical response of benzodiazepines with repeated short treatment. EEG patterns that are deprived of apparent response fall in the category of an ictal-interictal continuum and do not have explicit consensus and indications to continue the treatment [38]. After the initiation of IVAD administration, the achievement of seizure freedom treatment goals versus the pattern of the burst suppression also requires the help of cEEG [39]. In specific examples, EEG reactivity to the administration of drug-like frontal alpha development after administering ketamine can be suggested as a feasible indicator of desired or required results [40]. Software used for qEEG and advanced EEG integrated with amplitude and assessment of ratios of burst suppression using the color density exclusive array can be used to support seizure detection. When EEG is used on its own, the rate of false-positive can be average, high approximately being one per hour [41]. SE and seizures can occur due to lesions of structural neuroimaging, which in turn can reveal many abnormalities. Abnormalities in acute form and old lesions in chronic epilepsy are revealed with the help of the CT scan technology, e.g., easy identification of lesions can be made by cranial CT scan comprising of vascular malformation, intracranial hemorrhage, stroke, abscess, brain tumor, or other processes of infection or even malformation of the brain [42].
Difficulties Associated With Status Epilepticus
Central Nervous System
The activity of convulsions causes sympathetic stimulation which leads to an increased flow of blood in the cerebral region of the brain and an increased level of glucose in the blood. The primary effects of this activity complete the requirements of enhanced metabolic neuronal demand, but if the seizures continue, then these mechanisms which are compensatory raise to the level of exhaustion, an occurrence of anaerobic metabolism which results in the damage of neurons and cerebral hypoxia. Homeostasis safeguarding is necessary to prevent the injury of neurons and to maximize the brain’s supply of oxygen and glucose by keeping blood gases and blood flow in the cerebral region of the brain. This homeostasis is equally essential in reducing the metabolic needs of the cerebral region by restriction of hyperthermia and seizures [43].
Systemic Effects
Systemic effects show a relationship with the massive release of catecholamines and a hyperactive adrenergic state, which can result in pulmonary, neuro-cardiogenic, and, in few cases, renal or musculoskeletal injury [44].
Drug Therapy for Stage of Disorder
The treatment of SE begins with SE as a prime objective and aggressively managing an acute symptomatic etiology. If the SE is not treated timely and adequately, then it may turn into SRSE and RSE. In a general way, SE becomes more severe, depending on the period for which it is left untreated [45, 46]. A thorough critical SE study, the study of Veterans Cooperative Affairs, demonstrated that the treatment of SE becomes ineffective day by day with an increase in a period of SE. Remarkably, the treatment of a nonconvulsive type of SE is severe, and about 15% of all cases can be controlled by giving AEDs as compared to convulsive type SE, among which, the response to first AED is seen in around 55% of cases [47]. Likewise, the rate of mortality of RSE on a short-term basis ranges from 16 to 39%, which is around three times greater in comparison to that of NRSE. Presently, the guidelines for management are not specific for SE related to the age of the patient, the reason being that the pathophysiology of SE and the effects of a drug on receptors of neurons are the same in adults, children, and infants. However, neonates might be excluded, e.g., follow the consecutive i.v. administration of the benzodiazepines intended for brisk SE control; conventional AEDs directed at forms of early resistance (Fig. 2) and long-term coverage; and general anesthetic drugs for SRSE, immunoglobulin. Drugs according to the stage of epilepsy and some other drugs exclusively used in the treatment of SE (Fig. 3) are as follows: Lacosamide, employed as a medication for the cure of focal epilepsies, has been existent in the European Union since the year 2008. Bioequivalence studies conducted in healthy adults with a report of epilepsy demonstrated better tolerability of the solution given by the i.v. route of administration. Lacosamide showed a good efficiency on animal models of SE [48]. Another drug is paraldehyde with established anticonvulsant characteristics in both animal and human models. Its mechanism is not yet explicitly recognized, but its cardiovascular tolerability is deemed to be harmless. The rectal route of administration is a replacement of the conventional IM injection that is pain-free and unsolicited with the sterile abscess hazard [49]. Lidocaine is a drug of the local anesthetic category that acts by inhibiting ionic currents throughout membrane depolarization with the help of voltage-gated sodium channels. It is exciting to know that while not being explicitly approved for the management of this particular condition, i.v. lidocaine is extensively used for the treatment of CSE in Japan [50]. Carbamazepine is a drug that possesses ILAE level A facts for the treatment of focal epilepsy in adults. This drug has poor solubility in water, which is a primary obstruction in the preparation of its parenteral dosage form. The testing of i.v. carbamazepine in animal models as well as in humans has shown quick commencement of anticonvulsant action and absence of side effects on the respiratory or cardiovascular system. Novel formulations, with better pharmacokinetic profiles, have been developed for the i.v. route. Nevertheless, their application in clinical practice needs to be further evaluated [51]. Chlormethiazole (clomethiazole) is a drug derived from a thiazole that acts by boosting the activity of GABA, followed by an increase in inhibitory neurotransmissions. However, no data is available for controlled studies, as there were not any of the latter conducted for estimating its tolerability and its efficacy in SE. To date, there are a limited number of reports available of patients with SE, mostly children, getting therapeutic benefit from drug i.v. chlormethiazole [52], and numerous side effects have been reported related to the use of this drug comprising of respiratory depression and thrombophlebitis (Table 1).
Newer Therapies for the Treatment of Status Epilepticus
A type of steroid synthesized inside the brain through glia, having neurological effects, is neurosteroid; neuroactive is another type possessing a neurological activity and is synthesized in the area outside the CNS by conversion of the gonadal, placental, or adrenal steroids. Neuroactive steroids derived from progesterone, testosterone, or deoxycorticosterone can cross the blood-brain barrier and alter the structure and function of the brain [68]. Neurosteroids and neuroactive steroids are essential for the differences—depending on the sex of patients—in the structure, function, and development of the brain. In adults, the metabolic levels, neuronal receptors, and networks affected by levels of neurosteroids or neuroactive steroids differ according to the gender of the patient. These properties can explain gender-based differences seen in epilepsy regarding prevalence, etiology, progress, and response to the treatment. For instance, men are generally more susceptible to seizures, whereas women have more fluctuations in susceptibility to seizures because of fluctuations in hormones; they have a higher susceptibility to some syndromes of epilepsy that are very much complex and generally intractable [69,70,71,72]. Neurosteroids like tetrahydro deoxycorticosterone, androstane diol, and allopregnanolone show interaction with GABAA receptors, where they can directly stimulate the receptors in high concentrations and behave as potent definite allosteric agonists in low concentrations. Depending upon the location, GABAA receptors are of two types, synaptic and the extrasynaptic receptors of GABAA, as previously explained in the section of pathophysiology. Neurosteroids act on both the receptors but show a higher affinity for extrasynaptic GABAA receptors [73, 74]. Neurosteroids can opt for therapeutic action intended for SE, providing the targeting of those extrasynaptic receptors of GABAA that are not subjected to inhibition throughout the SE, as synaptic GABAA receptors [33].
GABAA Receptors
Allopregnanolone is studied for its potential therapeutic efficacy in SE treatment. A lot of preclinical studies done on the SE animal models show beneficial results associated with the usage of different types of neuroactive steroids like Allopregnanolone and others [75, 76]. Different types of clinical studies conducted on humans also show promising results. Broomall et al., in 2014, reported the use of allopregnanolone for the first time in two pediatric patients suffering from SRSE. From the study, it was found that both the patients responded to allopregnanolone after the futile use of various drugs having antiepileptic action [77]. Likewise, Vaitkevicius et al. demonstrated the use of allopregnanolone effectively in two adult patients of SRSE [78]. Some other neuroactive steroids similar to allopregnanolone, like ganaxolone, are also tested for refractory catamenial epilepsy, focal seizures in RSE, neonatal seizures, or infantile spasms [79,80,81]. Drugs that target the NMDA receptors by causing functional changes in their glutamate receptor, like ketamine and other drugs, show involvement in SE pathophysiology. Hence, in the SE treatment, the antagonists of NMDA receptors can be a better approach theoretically [82, 83]. Currently, the only NMDA receptor antagonist is ketamine, which is intravenously administered and commonly available in most nations. Currently, immunotherapy of epilepsy depends on immunosuppressants (monoclonal antibodies, plasmapheresis, immunoglobulins, adrenocorticotropic hormone, and corticotropic steroid hormones) and has been used in a particular set of conditions, some types of epilepsy like Lennox-Gastaut, autoimmune conditions or severe encephalitis, and infantile spasms [84]. The modern, novel approaches are being emphasized on the targeting of principal pro-inflammatory mediators that show their involvement in the inflammation of neurons associated with epilepsy with great potential.
Prognosis
Different types of studies have been conducted, which are overall focused on SE prognostic factors, but there is minuscule availability of data regarding SRSE or RSE. However, there is an admixture of comparatively improved SE diagnosis, poor SRSE prognosis, and stroke-induced RSE accompanied by high mortality and poor prognosis [85]. Based on another study, brain tumors and post anoxic encephalopathy were autonomously accompanied by a high death rate [86]. The recent epilepsy history was a result of deprived outcomes in a single study, not in other studies [87]. The high mortality rate resulted when the SE occurred with the onset of low consciousness levels (stupor or coma), and also, the NCSE and GCSE individually resulted in the death of patients. The period of RSE and coma for more than 10 days also results in a negative outcome [88]. However, there are specific reports in which patients survived, even those suffering from severe, persistent SE. The discharges of periodic epileptiform are highly accompanied by the RSE that can be seen in EEG findings. Oppositely, isoelectric EEG and burst suppression unavailability are allied with excellent outcomes, which may be due to less burden on the medications of anesthesia and reduced duration of the state of coma and hospitalization in this line [89]. Low levels of albumin, in the beginning, are autonomously allied to RSE and associated with death according to a study [90]. Reduction or withdrawal of AEDs is likely not going to result in RSE [91].
Patent and Clinical Trials for Status Epilepticus
Patent data plays a vital role as a fundamental information platform in the detection of developments in technological growth [92]. Patent data for status epilepticus is included in Table 2. Clinical trials are investigational studies conducted on humans for discovering the therapeutic agent or for determining any adverse reaction [103]; therefore, the therapeutic agents on which clinical trials are conducted are discussed in Table 3.
Conclusion and Future Prospects
Status epilepticus is a lethal disorder affecting the brain and a state of medical emergency that is associated with electrical discharges of cortical neurons. It requires prompt treatment and diagnosis for management; hence, the pathophysiological aspects are covered to fully understand the disease and associated complications. EEG is a useful technique used for detecting different characteristics of SE and for the monitoring of the patient in ICU. SE is a critical condition, and its stages, the RSE and the SRSE, are more severe, making their management all the more challenging. Patients and clinicians would conjointly have the benefit of a wide-ranging meta-analysis of extrapolative factors as at this point; disparate studies demonstrate varied results. Correspondingly, investigations devoted to management and outcome of the investigations in populations comprising primarily of elderly conceived females and those who are suffering from neurodegenerative disorders are also deficient. In the future, studies must be conducted to tackle these issues effectively. Continual advancements in fundamental research can influence the treatment of SE in emergencies and seizures. It is clear from the pathophysiology of SE that prompt cessation of SE can have a positive effect on patients. Shortly, there should be the development of new delivery systems of the prevailing drugs to terminate seizure activity, and this will lead to a change in strategies of treatment. New drugs will be available, those having neuroprotective action, that will help in preventing injury to neurons, although they may not be of aid in seizure termination, for instance, the availability of NMDA antagonists. Advancement in EEG will enable to detection of subtle SE, and this may impact treatment in emergency conditions.
Abbreviations
- SE:
-
Status epilepticus
- CSE:
-
Convulsive SE
- ESE:
-
Established SE
- RSE:
-
Refractory SE
- FSE:
-
Focal SE
- SRSE:
-
Super RSE
- GCSE:
-
Generalized CSE
- GABA:
-
Gamma-aminobutyric acid
- NMDA:
-
N-methyl-D-aspartate
- AMPA:
-
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid
- NKCC:
-
Na-K-Cl cotransporter
- CNS:
-
Central nervous system
- AEDs:
-
Antiepileptic drugs
- CT:
-
Computed tomography
- cEEG:
-
Continuous electroencephalogram
- NCSE:
-
Non-CSE
- IVAD:
-
Implantable ventricular assist device
- qEEG:
-
Quantitative EEG
- NRSE:
-
Non-RSE
- ICU:
-
Intensive care unit
- MRI:
-
Magnetic resonance imaging
References
Trinka E, Cock H, Hesdorffer D, Rossetti AO, Scheffer IE, Shinnar S, Shorvon S, Lowenstein DH. A definition and classification of status epilepticus–report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia. 2015;56(10):1515–23. https://doi.org/10.1111/epi.13121.
Guterman EL, Burke JF, Sporer KA. Prehospital treatment of status epilepticus in the United States. JAMA. 2021;326(19):1970–1.
Lee SK. Diagnosis and treatment of status epilepticus. J Epilepsy Res. 2020;10(2):45–54.
Johnson EL, Kaplan PW. Status epilepticus: definition, classification, pathophysiology, and epidemiology. Semin Neurol. 2020;40:647–51. https://doi.org/10.1055/s-0040-1718722.
Rossetti AO, Alvarez V. Update on the management of status epilepticus. Curr Opin Neurol. 2021;34(2):172–81. https://doi.org/10.1097/WCO.0000000000000899.
Claassen J, Goldstein JN. Emergency neurological life support: status epilepticus. Neurocrit Care. 2017;27(1):152–8. https://doi.org/10.1007/s12028-017-0460-1.
Glauser T, Shinnar S, Gloss D, Alldredge B, Arya R, Bainbridge J, Bare M, Bleck T, Dodson WE, Garrity L, Jagoda A. Evidence-based guideline: treatment of convulsive status epilepticus in children and adults: report of the Guideline Committee of the American Epilepsy Society. Epilepsy Curr. 2016;16(1):48–61. https://doi.org/10.5698/1535-7597-16.1.48.
Hirsch LJ, Gaspard N, van Baalen A, Nabbout R, Demeret S, Loddenkemper T, Navarro V, Specchio N, Lagae L, Rossetti AO, Hocker S. Proposed consensus definitions for new-onset refractory status epilepticus (NORSE), febrile infection-related epilepsy syndrome (FIRES), and related conditions. Epilepsia. 2018;59(4):739–44. https://doi.org/10.1111/epi.14016.
DeLorenzo RJ, Hauser WA, Towne AR, Boggs JG, Pellock JM, Penberthy L, Garnett L, Fortner CA, Ko D. A prospective, population-based epidemiologic study of status epilepticus in Richmond,Virginia. Neurology. 1996;46(4):1029–35. https://doi.org/10.1212/WNL.46.4.1029.
Lv RJ, Wang Q, Cui T, Zhu F, Shao XQ. Status epilepticus-related etiology, incidence and mortality: a meta-analysis. Epilepsy Res. 2017;136:12–7. https://doi.org/10.1016/j.eplepsyres.2017.07.006.
Tiamkao S, Pranbul S, Sawanyawisuth K, Thepsuthammarat K. Integrated Epilepsy Research Group. A national database of incidence and treatment outcomes of status epilepticus in Thailand. Int J Neurosci. 2014;124(6):416–20. https://doi.org/10.3109/00207454.2013.850084.
Al-Sofyani KA. An insight into the current understanding of status epilepticus: from concept to management. Neurol Res Int. 2021. https://doi.org/10.1155/2021/9976754.
Paris M, Reddy U. Update on the management of status epilepticus. Anaesth Intensive Care Med. 2018;19(3):83–6. https://doi.org/10.1016/j.mpaic.2017.12.008.
Cock HR. Drug-induced status epilepticus. Epilepsy Behav. 2015;49:76–82. https://doi.org/10.1016/j.yebeh.2015.04.034.
Goodkin HP, Yeh JL, Kapur J. Status epilepticus increases the intracellular accumulation of GABAA receptors. J Neurosci. 2005;25(23):5511–20. https://doi.org/10.1523/JNEUROSCI.0900-05.2005.
Naylor DE, Liu H, Wasterlain CG. Trafficking of GABAA receptors, loss of inhibition, and a mechanism for pharmacoresistance in status epilepticus. J Neurosci. 2005;25(34):7724–33. https://doi.org/10.1523/JNEUROSCI.4944-04.2005.
Naylor DE, Liu H, Niquet J, Wasterlain CG. Rapid surface accumulation of NMDA receptors increases glutamatergic excitation during status epilepticus. Neurobiol Dis. 2013;54:225–38. https://doi.org/10.1016/j.nbd.2012.12.015.
Naylor DE. Glutamate and GABA in the balance: convergent pathways sustain seizures during status epilepticus. Epilepsia. 2010;51:106–9. https://doi.org/10.1111/j.1528-1167.2010.02622.x.
Brooks-Kayal AR, Pritchett DB. Developmental changes in human γ-aminobutyric acida receptor subunit composition. Ann Neurol. 1993;34(5):687–93. https://doi.org/10.1002/ana.410340511.
Kumar SS, Bacci A, Kharazia V, Huguenard JR. A developmental switch of AMPA receptor subunits in neocortical pyramidal neurons. J Neurosci. 2002;22(8):3005–15. https://doi.org/10.1523/JNEUROSCI.22-08-03005.2002.
Crino PB, Duhaime AC, Baltuch G, White R. Differential expression of glutamate and GABA-A receptor subunit mRNA in cortical dysplasia. Neurology. 2001;56(7):906–13. https://doi.org/10.1212/WNL.56.7.906.
Loddenkemper T, Talos DM, Cleary RT, Joseph A, Fernández IS, Alexopoulos A, Kotagal P, Najm I, Jensen FE. Subunit composition of glutamate and gamma-aminobutyric acid receptors in status epilepticus. Epilepsy Res. 2014;108(4):605–15. https://doi.org/10.1016/j.eplepsyres.2014.01.015.
Dzhala VI, Talos DM, Sdrulla DA, Brumback AC, Mathews GC, Benke TA, Delpire E, Jensen FE, Staley KJ. NKCC1 transporter facilitates seizures in the developing brain. Nat Med. 2005;11(11):1205–13. https://doi.org/10.1038/nm1301.
Rakhade SN, Jensen FE. Epileptogenesis in the immature brain: emerging mechanisms. Nat Rev Neurol. 2009;5(7):380. https://doi.org/10.1038/nrneurol.2009.80.
Rakhade SN, Zhou C, Aujla PK, Fishman R, Sucher NJ, Jensen FE. Early alterations of AMPA receptors mediate synaptic potentiation induced by neonatal seizures. J Neurosci. 2008;28(32):7979–90. https://doi.org/10.1523/JNEUROSCI.1734-08.2008.
Van Vliet EA, da Costa AS, Redeker S, Van Schaik R, Aronica E, Gorter JA. Blood–brain barrier leakage may lead to progression of temporal lobe epilepsy. Brain. 2007;130(2):521–34. https://doi.org/10.1093/brain/awl318.
Han H, Mann A, Ekstein D, Eyal S. Breaking bad: the structure and function of the blood-brain barrier in epilepsy. AAPS J. 2017;19(4):973–88. https://doi.org/10.1208/s12248-017-0096-2.
Nadal A, Fuentes E, McNaughton PA. Glial cell responses to lipids bound to albumin in serum and plasma. Prog Brain Res. 2001;132:367–74. https://doi.org/10.1016/S0079-6123(01)32088-5.
Ranaivo HR, Wainwright MS. Albumin activates astrocytes and microglia through mitogen-activated protein kinase pathways. Brain Res. 2010;1313:222–31. https://doi.org/10.1016/j.brainres.2009.11.063.
Cacheaux LP, Ivens S, David Y, Lakhter AJ, Bar-Klein G, Shapira M, Heinemann U, Friedman A, Kaufer D. Transcriptome profiling reveals TGF-β signaling involvement in epileptogenesis. J Neurosci. 2009;29(28):8927–35. https://doi.org/10.1523/JNEUROSCI.0430-09.2009.
Vezzani A, Friedman A, Dingledine RJ. The role of inflammation in epileptogenesis. Neuropharmacology. 2013;69:16–24. https://doi.org/10.1016/j.neuropharm.2012.04.004.
Van Vliet EA, Aronica E, Vezzani A, Ravizza T. Neuroinflammatory pathways as treatment targets and biomarker candidates in epilepsy: emerging evidence from preclinical and clinical studies. Neuropathol Appl Neurobiol. 2018;44(1):91–111. https://doi.org/10.1111/nan.12444.
Dey A, Kang X, Qiu J, Du Y, Jiang J. Anti-inflammatory small molecules to treat seizures and epilepsy: from bench to bedside. Trends Pharmacol Sci. 2016;37(6):463–84. https://doi.org/10.1016/j.tips.2016.03.001.
Bankstahl JP, Löscher W. Resistance to antiepileptic drugs and expression of P-glycoprotein in two rat models of status epilepticus. Epilepsy Res. 2008;82(1):70–85. https://doi.org/10.1016/j.eplepsyres.2008.07.007.
Amengual-Gual M, Fernández IS, Wainwright MS. Novel drugs and early polypharmacotherapy in status epilepticus. Seizure. 2019;68:79–88. https://doi.org/10.1016/j.seizure.2018.08.004.
Al-Mufti F, Claassen J. Neurocritical care: status epilepticus review. Crit Care Clin. 2014;30(4):751–64. https://doi.org/10.1016/j.ccc.2014.06.006.
Marawar R, Basha M, Mahulikar A, Desai A, Suchdev K, Shah A. Updates in refractory status epilepticus. Crit Care Res Pract. 2018;2018:9768949. https://doi.org/10.1155/2018/9768949.
Brophy GM, Bell R, Claassen J, Alldredge B, Bleck TP, Glauser T, LaRoche SM, Riviello JJ, Shutter L, Sperling MR, Treiman DM. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care. 2012;17(1):3–23. https://doi.org/10.1007/s12028-012-9695-z.
Basha MM, Alqallaf A, Shah AK. Drug-induced EEG pattern predicts effectiveness of ketamine in treating refractory status epilepticus. Epilepsia. 2015;56(4):e44–8. https://doi.org/10.1111/epi.1294.
Haider HA, Esteller R, Hahn CD, Westover MB, Halford JJ, Lee JW, Shafi MM, Gaspard N, Herman ST, Gerard EE, Hirsch LJ. Sensitivity of quantitative EEG for seizure identification in the intensive care unit. Neurology. 2016;87(9):935–44. https://doi.org/10.1212/WNL.0000000000003034.
Stewart CP, Otsubo H, Ochi A, Sharma R, Hutchison JS, Hahn CD. Seizure identification in the ICU using quantitative EEG displays. Neurology. 2010;75(17):1501–8. https://doi.org/10.1212/WNL.0b013e3181f9619e.
Nair PP, Kalita J, Misra UK. Role of cranial imaging in epileptic status. Eur J Radiol. 2009;70(3):475–80. https://doi.org/10.1016/j.ejrad.2008.02.007.
Legriel S, Bruneel F, Dalle L, Appere-de-Vecchi C, Georges JL, Abbosh N, Henry-Lagarrigue M, D’Allonnes LR, Mokhtar HB, Audibert J, Guezennec P. Recurrent takotsubo cardiomyopathy triggered by convulsive status epilepticus. Neurocrit Care. 2008;9(1):118–21. https://doi.org/10.1007/s12028-008-9107-6.
Bleck TP. Convulsive disorders: status epilepticus. Clin Neuropharmacol. 1991;14(3):191–8. https://doi.org/10.1097/00002826-199106000-00001.
Legriel S, Mourvillier B, Bele N, Amaro J, Fouet P, Manet P, Hilpert F. Outcomes in 140 critically ill patients with status epilepticus. Intensive Care Med. 2008;34(3):476–80. https://doi.org/10.1007/s00134-007-0915-5.
Towne AR, Pellock JM, Ko D, DeLorenzo RJ. Determinants of mortality in status epilepticus. Epilepsia. 1994;35(1):27–34. https://doi.org/10.1111/j.1528-1157.1994.tb02908.x.
Giovannini G, Monti G, Polisi MM, Mirandola L, Marudi A, Pinelli G, Valzania F, Girardis M, Nichelli PF, Meletti S. A one-year prospective study of refractory status epilepticus in Modena. Italy Epilepsy Behav. 2015;49:141–5. https://doi.org/10.1016/j.yebeh.2015.05.022.
Biton V, Rosenfeld WE, Whitesides J, Fountain NB, Vaiciene N, Rudd GD. Intravenous lacosamide as replacement for oral lacosamide in patients with partial-onset seizures. Epilepsia. 2008;49(3):418–24. https://doi.org/10.1111/j.1528-1167.2007.01317.x.
Rowland AG, Gill AM, Stewart AB, Appleton RE, Al Kharusi A, Cramp C, Yeung LK. Review of the efficacy of rectal paraldehyde in the management of acute and prolonged tonic–clonic convulsions. Arch Dis Child. 2009;94(9):720–3. https://doi.org/10.1136/adc.2009.157636.
Kobayashi O, Iwasaki Y, Yamanouchi H, Sugai K. A case of refractory status epilepticus associated with aberrant intracranial shunt tube: efficacy of lidocaine in the determination of the epileptic focus. Brain Dev. 1994;26(4):329–34.
Franco V, Iudice A, Grillo E, Citraro R, De Sarro G, Russo E. Perspective on the use of perampanel and intravenous carbamazepine for generalized seizures. Expert Opin Pharmacother. 2014;15(5):637–44. https://doi.org/10.1517/14656566.2014.879572.
Miller P, Kovar I. Chlormethiazole in the treatment of neonatal status epilepticus. Postgrad Med. 1983;59(698):801–2. https://doi.org/10.1136/pgmj.59.698.801.
Derivan AT, Homan RW, Walker J, Ramsay RE, Patrick B. Double-blind study of lorazepam and diazepam in status epilepticus. Jama. 1983;249(11):1452–4. https://doi.org/10.1001/jama.1983.03330350028021.
Trinka E Benzodiazepines used primarily for emergency treatment (diazepam, lorazepam and midazolam). The treatment of epilepsy. 2009 431-46. https://doi.org/10.1002/9781444316667.ch34.
Congdon PJ, Forsythe WI. Intravenous clonazepam in the treatment of status epilepticus in children. Epilepsia. 1980;21(1):97–102. https://doi.org/10.1111/j.1528-1157.1980.tb04049.x.
Burneo JG, Anandan JV, Barkley GL. A prospective study of the incidence of the purple glove syndrome. Epilepsia. 2001;42(9):1156–9. https://doi.org/10.1046/j.1528-1157.2001.12901.x.
Perucca E. Pharmacological and therapeutic properties of valproate. CNS Drugs. 2002;16(10):695–714. https://doi.org/10.2165/00023210-200216100-00004.
Treiman DM, Meyers PD, Walton NY, Collins JF, Colling C, Rowan AJ, Handforth A, Faught E, Calabrese VP, Uthman BM, Ramsay RE. A comparison of four treatments for generalized convulsive status epilepticus. N Engl J Med. 1998;339(12):792–8. https://doi.org/10.1056/NEJM199809173391202.
Singhi S, Murthy A, Singhi P, Jayashree M. Continuous midazolam versus diazepam infusion for refractory convulsive status epilepticus. J Child Neurol. 2002;17(2):106–10. https://doi.org/10.1177/088307380201700203.
Marik PE. Propofol: therapeutic indications and side-effects. Curr Pharm Des. 2004;10(29):3639–49. https://doi.org/10.2174/1381612043382846.
Rossetti AO, Milligan TA, Vulliémoz S, Michaelides C, Bertschi M, Lee JW. A randomized trial for the treatment of refractory status epilepticus. Neurocrit Care. 2011;14(1):4–10. https://doi.org/10.1007/s12028-010-9445-z.
Rossetti AO. Novel anesthetics and other treatment strategies for refractory status epilepticus. Epilepsia. 2009;50(Suppl 12):51–3. https://doi.org/10.1111/j.1528-1167.2009.02369.x.
Mirsattari SM, Sharpe MD, Young GB. Treatment of refractory status epilepticus with inhalational anesthetic agents isoflurane and desflurane. Arch Neurol. 2004;61(8):1254–9. https://doi.org/10.1001/archneur.61.8.1254.
Shorvon S, Ferlisi M. The treatment of super-refractory status epilepticus: a critical review of available therapies and a clinical treatment protocol. Brain. 2011;134(10):2802–18. https://doi.org/10.1093/brain/awr215.
Borris DJ, Bertram EH, Kapur J. Ketamine controls prolonged status epilepticus. Epilepsy Res. 2000;42(2-3):117–22. https://doi.org/10.1016/S0920-1211(00)00175-3.
Shank RP, Gardocki JF, Streeter AJ, Maryanoff BE. An overview of the preclinical aspects of topiramate: pharmacology, pharmacokinetics, and mechanism of action. Epilepsia. 2000;41:3–9. https://doi.org/10.1111/j.1528-1157.2000.tb02163.x.
Gaspard N, Foreman B, Judd LM, Brenton JN, Nathan BR, McCoy BM, Al-Otaibi A, Kilbride R, Fernández IS, Mendoza L, Samuel S. Intravenous ketamine for the treatment of refractory status epilepticus: a retrospective multicenter study. Epilepsia. 2013;54(8):1498–503. https://doi.org/10.1111/epi.12247.
Reddy DS. Neurosteroids: endogenous role in the human brain and therapeutic potentials. Prog Brain Res. 2010;186:113–37. https://doi.org/10.1016/B978-0-444-53630-3.00008-7.
Hauser WA, Annegers JF, Kurland LT. Incidence of epilepsy and unprovoked seizures in Rochester, Minnesota: 1935–1984. Epilepsia. 1993;34(3):453–8. https://doi.org/10.1111/j.1528-1157.1993.tb02586.x.
McHugh JC, Delanty N. Epidemiology and classification of epilepsy: gender comparisons. Int Rev Neurobiol. 2008;83:11–26. https://doi.org/10.1016/S0074-7742(08)00002-0.
Frye CA. Hormonal influences on seizures: basic neurobiology. Int Rev Neurobiol. 2008;83:27–77. https://doi.org/10.1016/S0074-7742(08)00003-2.
Herzog AG. Catamenial epilepsy: update on prevalence, pathophysiology and treatment from the findings of the NIH Progesterone Treatment Trial. Seizure. 2015;28:18–25. https://doi.org/10.1016/j.seizure.2015.02.024.
Reddy DS, Estes WA. Clinical potential of neurosteroids for CNS disorders. Trends Pharmacol Sci. 2016;37(7):543–61. https://doi.org/10.1016/j.tips.2016.04.003.
Reddy D. Role of hormones and neurosteroids in epileptogenesis. Front Cell Neurosci. 2013;7:115. https://doi.org/10.3389/fncel.2013.00115.
Lévesque M, Herrington R, Leclerc L, Rogawski MA, Avoli M. Allopregnanolone decreases interictal spiking and fast ripples in an animal model of mesial temporal lobe epilepsy. Neuropharmacology. 2017;121:12–9. https://doi.org/10.1016/j.neuropharm.2017.04.020.
Dhir A, Chopra K. On the anticonvulsant effect of allopregnanolone (a neurosteroid) in neonatal rats. Life Sci. 2015;143:202–8. https://doi.org/10.1016/j.lfs.2015.09.008.
Lonsdale D, Burnham WM. The anticonvulsant effects of allopregnanolone against amygdala-kindled seizures in female rats. Neurosci Lett. 2007;411(2):147–51. https://doi.org/10.1016/j.neulet.2006.10.023.
Broomall E, Natale JE, Grimason M, Goldstein J, Smith CM, Chang C, Kanes S, Rogawski MA, Wainwright MS. Pediatric super-refractory status epilepticus treated with allopregnanolone. Ann Neurol. 2014;76(6):911–5. https://doi.org/10.1002/ana.24295.
Reddy K, Reife R, Cole AJ. SGE-102: a novel therapy for refractory status epilepticus. Epilepsia. 2013;54:81–3. https://doi.org/10.1111/epi.12286.
Meletti S, Lucchi C, Monti G, Giovannini G, Bedin R, Trenti T, Rustichelli C, Biagini G. Decreased allopregnanolone levels in cerebrospinal fluid obtained during status epilepticus. Epilepsia. 2017;58(2):e16–20. https://doi.org/10.1111/epi.13625.
Rosenthal ES, Claassen J, Wainwright MS, Husain AM, Vaitkevicius H, Raines S, Hoffmann E, Colquhoun H, Doherty JJ, Kanes SJ. Brexanolone as adjunctive therapy in super-refractory status epilepticus. Ann Neurol. 2017;82(3):342–52. https://doi.org/10.1002/ana.25008.
Dorandeu F. Ketamine for the treatment of (super) refractory status epilepticus? Not quite yet. Expert Rev Neurother. 2017;17:419–21. https://doi.org/10.1080/14737175.2017.1288099.
Dorandeu F, Dhote F, Barbier L, Baccus B, Testylier G. Treatment of status epilepticus with ketamine, are we there yet? CNS Neurosci Ther. 2013;19(6):411–27. https://doi.org/10.1111/cns.12096.
Knake S, Rochon J, Fleischer S, Katsarou N, Back T, Vescovi M, Oertel WH, Reis J, Hamer HM, Rosenow F. Status epilepticus after stroke is associated with increased long-term case fatality. Epilepsia. 2006;47(12):2020–6. https://doi.org/10.1111/j.1528-1167.2006.00845.x.
Sutter R, Marsch S, Fuhr P, Rüegg S. Mortality and recovery from refractory status epilepticus in the intensive care unit: a 7-year observational study. Epilepsia. 2013;54(3):502–11. https://doi.org/10.1111/epi.12064.
Hocker SE, Britton JW, Mandrekar JN, Wijdicks EF, Rabinstein AA. Predictors of outcome in refractory status epilepticus. JAMA Neurol. 2013;70(1):72–7. https://doi.org/10.1001/jamaneurol.2013.578.
Bausell R, Svoronos A, Lennihan L, Hirsch LJ. Recovery after severe refractory status epilepticus and 4 months of coma. Neurology. 2011;77(15):1494–5. https://doi.org/10.1212/WNL.0b013e318232abc0.
Mayer SA, Claassen J, Lokin J, Mendelsohn F, Dennis LJ, Fitzsimmons BF. Refractory status epilepticus: frequency, risk factors, and impact on outcome. Arch Neurol. 2002;59(2):205–10. https://doi.org/10.1001/archneur.59.2.205.
Sutter R, Grize L, Fuhr P, Rüegg S, Marsch S. Acute-phase proteins and mortality in status epilepticus: a 5-year observational cohort study. Crit Care Med. 2013;41(6):1526–33. https://doi.org/10.1097/CCM.0b013e318287f2ac.
Holtkamp M, Othman J, Buchheim K, Meierkord H. Predictors and prognosis of refractory status epilepticus treated in a neurological intensive care unit. J Neurol Neurosur Ps. 2005;76(4):534–9. https://doi.org/10.1136/jnnp.2004.041947.
Neligan A, Shorvon SD. Frequency and prognosis of convulsive status epilepticus of different causes: a systematic review. Arch Neurol. 2010;67(8):931–40. https://doi.org/10.1001/archneurol.2010.169.
Yoon J, Kim K. Detecting signals of new technological opportunities using semantic patent analysis and outlier detection. Scientometrics. 2012;90(2):445–61. https://doi.org/10.1007/s11192-011-0543-2.
Baraban SC. Compounds and methods for treating an epileptic disorder. University of California. US10695325B2. 2020-06-30.
McNamara JO, Liu G, Gu B, He XP, Krishnamurthy K, Huang Y, Mook R. Methods for the prevention or treatment of epilepsy. Duke University. US20190269752A1. 2019-09-05.
von Moltke LL, Weiden PJ, Hard ML. Aripiprazole dosing strategy. Alkermes Pharma Ireland Ltd. US20190298716A1. 2019-10-03.
Irazoqui P, Budde RB, Pederson D. Prevention of reflux induced laryngospasm. Purdue Research Foundation. US20190336758A1. 2019-11-07.
Galer BS, Faingold CL, Martin P. Compositions and methods for treating respiratory depression with fenfluramine. Zogenix International Ltd. US20190380979A1. 2019-12-31.
Bekenstein U, Soreq H, Greenberg D. Methods for controlling seizures by manipulating the levels of microRNA-211 (miR-211) in the brain. Uriah Bekenstein. US20200102559A1. 2020-04-02.
Osorio I. Contingent cardio-protection for epilepsy patients. Flint Hills Scientific LLC. US20200206508A1. 2020-07-02.
Rogawski MA, Pessah IS, Cao Z, Lein PJ. Mitigation of epileptic seizures by combination therapy using benzodiazepines and neurosteroids. University of California. US20200215078A1. 2020-07-09.
Salituro FG, Blanc-Pillado MJ, Morningstar M, Harrison BL. 9(11)-unsaturated neuroactive steroids and their methods of use. Sage Therapeutics, Inc. WO2020082065A1. 2020-04-23.
Blanc-Pillado MJ, Salituro FG. 3.alpha.-hydroxy-17.beta.-amide neuroactive steroids and compositions thereof. Sage Therapeutics, Inc. WO2020132504A1. 2020-06-25.
Umscheid CA, Margolis DJ, Grossman CE. Key concepts of clinical trials: a narrative review. Postgrad Med. 2011;123(5):194–204.
Seizure Rescue Medication: Caregiver Education in a Simulation Setting. (ClinicalTrials.gov Identifier: NCT03562351). https://clinicaltrials.gov/ct2/ show/NCT03562351?cond=NCT03562351. BostonChildren'sHospital. 2020.
EEG Cap for Identification of Non-Convulsive Status Epilepticus. (ClinicalTrials.gov Identifier: NCT03138876). https://clinicaltrials.gov/ct2/show/NCT03138876 ?cond=NCT03138876. MayoClinic. 2019.
Efficacy of Ketamine Infusion Compared With Traditional Anti-epileptic Agents in Refractory Status Epilepticus. (ClinicalTrials.gov Identifier: NCT03115489). https://clinicaltrials.gov/ct2/show/NCT03115489?cond=NCT03115489.UniversityofAlabamaatBirmingham. 2020.
Established Status Epilepticus Treatment Trial (ESETT). (ClinicalTrials.gov Identifier: NCT01960075). https://clinicaltrials.gov/ct2/show/NCT01960075?cond=NCT01960075.UniversityofVirginia. 2020.
Study to Evaluate SAGE-547 Injection as Adjunctive Therapy for the Treatment of Super-Refractory Status Epilepticus (SRSE). (ClinicalTrials.gov Identifier: NCT02052739). https://clinicaltrials.gov/ct2/show/NCT02052739?cond=NCT020 52739. SageTherapeutics. 2020.
A Study With SAGE-547 for Super-Refractory Status Epilepticus. (ClinicalTrials.gov Identifier: NCT02477618). https://clinicaltrials.gov/ct2/show/NCT02477618 ?cond=NCT02477618. ] SageTherapeutics. 2019.
Acknowledgements
The authors are very thankful to Radhika Pandita for proofreading and editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors revealed that there is no conflict of interest regarding the publication of this paper.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuropharmacology.
Rights and permissions
About this article
Cite this article
Raina, N., Yadav, M., Rani, R. et al. Status Epilepticus: an Overview for Neuroscientists. Curr Pharmacol Rep 8, 36–47 (2022). https://doi.org/10.1007/s40495-021-00272-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40495-021-00272-7