Abstract
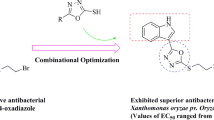
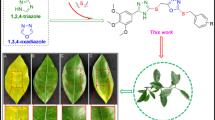
Through a facile structural modification on the natural bioactive ingredient 18β-glycyrrhetinic acid(GA), a series of novel GA hydrazide or amide derivatives was obtained, and their final molecular frameworks were characterized by NMR and HRMS analysis. Antibacterial bioassays revealed that some of the GA hydrazide or amide derivatives were able to suppress the growth of three tested plant pathogens. Particularly, compound 3c exhibited excellent in vitro activity against Xanthomonas oryzae pv. Oryzae (Xoo), Pseudomonas syringae pv. actinidiae(Psa), and Xanthomonas axonopodis pv. citri(Xac), providing the EC50 values of 5.89, 16.1, and 3.64 µg/mL, respectively. The data were better than those of the positive controls thiodiazole copper(92.7, 77.8, and 89.9 µg/mL, respectively) and bismerthiazol(31.1, 125.6, and 77.4 µg/mL, respectively). In addition, in vivo experiments suggested that, compared with thiodiazole copper(41.93% and 39.73%, respectively), compound 3c exerted prominently curative and protective activities against rice bacterial leaf blight at 200 µg/mL with the control effects of 52.36% and 51.40%, respectively. Given these obtained results, GA hydrazide or amide derivatives could serve as the feasible leads for exploring highly bioactive substrates.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Verma G., Sharma M., Mondal K. K., Funct. Plant Biol., 2018, 45, 561
Nozue K., Park C. J., Ronald P. C., J. Plant Biol., 2011, 54, 269
Chakraborty S., Newton A. C., Plant Pathol., 2011, 60, 2
Wang P. Y., Wang M. W., Zeng D., Xiang M., Rao J. R., Liu Q. Q., Liu L. W., Wu Z. B., Li Z., Song B. A., Yang S., J. Agric. Food Chem., 2019, 67, 3535
Huang N., Angeles E. R., Domingo J., Magpantay G., Singh S., Zhang G., Kumaravadivel N., Bennett J., Khush G. S., Theor. Appl. Genet., 1997, 95, 313
Liu W., Liu J., Triplett L., Leach J., Wang G., Annu. Rev. Phytopathol., 2014, 52, 213
Lin Y., He Z., Rosskopf E. N., Conn K. L., Powell C. A., Lazarovits G., Plant Dis., 2010, 94, 201
Chen J. X., Yi C. F., Wang S. B., Wu S. K., Li S. Y., Hu D. Y., Song B. A., Bioorg. Med. Chem. Lett., 2019, 29, 1203
Donati I., Cellini A., Sangiorgio D., Vanneste J. L., Scortichini M., Balestra G. M., Spinelli F., Microb. Ecol., 2020, 80, 81
Scortichini M., Eur. J. Plant Pathol., 2014, 140, 887
Abderrahim L. A., Taibi K., Abderrahim N. A., Boussaid M., Rios-Navarro C., Ruiz-Sauri A., Burns, 2019, 45, 1695
Saeed A., Eur. J. Med. Chem., 2016, 116, 290
Graebin C. S., Verli H., Guimarães J. A., J. Brazil. Chem. Soc., 2010, 21, 1595
Jin L., Huang R. Z., Huang X. C., Zhang B., Ji M., Wang, H. S., Bioorgan. Med. Chem., 2018, 26, 1759
Zhou F., Wu G. R., Cai D. S., Xu B., Yan M. M., Ma T., Guo W. B., Zhang W. X., Huang X. M., Yang Y. Q., Gao F., Wang P. L., Jia X. H., Lei H., Eur. J. Med. Chem., 2019, 178, 623
Huang L. R., Hao X. J., Li Q. J., Wang D. P., Zhang J. X., Luo H., Yang X. S., J. Nat. Prod., 2016, 79, 721
Xiang M., Zhou X., Luo T. R., Wang P. Y., Liu L. W., Li Z., Wu Z. B., Yang S., J. Agric. Food Chem., 2019, 67, 13212
Xiang M., Song Y. L., Ji J., Zhou X., Liu L. W., Wang P. Y., Wu Z. B., Li Z., Yang S., Pest Manag. Sci., 2020, 76, 2959
Zigolo M. A., Salinas M., Alche L., Baldessari A., Linares G. G., Bioorg. Chem., 2018, 78, 210
Radwan M. O., Ismail M. A. H., El-Mekkawy S., Ismail N. S. M., Hanna A. G., Arab. J. Chem., 2016, 9, 390
Wu S. Y., Cui S. C., Wang L., Zhang Y. T., Yan X. X., Lu H. L., Xing G. Z., Ren J., Gong L. K., Acta Pharmacol. Sin., 2018, 39, 1865
Chen H. J., Kang S. P., Lee I. J., Lin Y. L., J. Agric. Food Chem., 2014, 62, 618
Dai L. H., Li J., Yang J. G., Men Y., Zeng Y., Cai Y., Sun Y. X., Catalysts, 2018, 8, 615
Jin L., Dai L. M., Ji M., Wang H. S., Bioorg. Chem., 2019, 85, 179
Hussain H., Green I. R., Shamraiz U., Saleem M., Badshah A., Abbas G., Rehman N. U., Irshad M., Expert Opin. Ther. Pat., 2018, 28, 383
Cai D., Zhang Z. H., Meng Y. F., Zhu K. L., Chen L. Y., Yu C. X., Yu C. W., Fu Z. Y., Yang D. S., Gong Y. X., Beilstein J. Org. Chem., 2020, 16, 798
Chen C. X., Wei M. X., Li X. Q., Li T. C., Zhou X. Z., Chinese J. Org. Chem., 2015, 35, 835
Cai D., Zhang Z. H., Chen Y., Ruan C., Li S. Q., Chen S. Q., Chen L. S., RSC Adv., 2020, 10, 11694
Liu C. M., Huang J. Y., Sheng L. X., Wen X. A., Cheng K. G., MedChemCommun, 2019, 10, 1370
Beseda I., Czollner L., Shah P. S., Khunt R., Gaware R., Kosma P., Stanetty C., del Ruiz-Ruiz M. C., Amer H., Mereiter K., Da Cunha T., Odermatt A., Classen-Houben D., Jordis U., Bioorgan. Med. Chem., 2010, 18, 437
Tao Q. Q., Liu L. W., Wang P. Y., Long Q. S., Zhao Y. L., Jin L. H., Xu W. M., Chen Y., Li Z., Yang S., J. Agric. Food Chem., 2019, 67, 7626
Wang P. Y., Xiang M., Luo M., Liu H. W., Zhou X., Wu Z. B., Liu L. W., Li Z., Yang S., Pest Manag. Sci., 2020, 76, 2744
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos.21662009, 21702037, 31860516) and the Guizhou Provincial S&T Program, China(Nos.[2017]5788, 2018[1051]).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflicts of interest.
Supporting Information
Rights and permissions
About this article
Cite this article
Zhang, L., Fu, Y., Ding, Y. et al. Antibacterial Activity of Novel 18β-Glycyrrhetinic Hydrazide or Amide Derivatives. Chem. Res. Chin. Univ. 37, 662–667 (2021). https://doi.org/10.1007/s40242-021-0370-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-021-0370-9