Abstract
Purpose of Review
The goal of this paper is to review the surgical techniques for the correction of sternal cleft in children.
Recent Findings
Various surgical techniques have been developed for the correction of sternal cleft since 1949, when the first successful repair was reported.
Summary
Sternal cleft is an uncommon congenital chest wall deformity. The optimal correction can be achieved by primary repair of the defect in patients younger than 1 year of age, especially during the neonatal period, mostly with an easy technique. In older children, more extensive techniques are applied, using either autologous tissues or synthetic prosthetic materials, with satisfactory functional and aesthetic results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
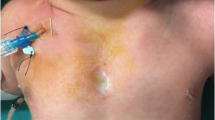
A variety of malformations of the chest wall may be identified in childhood. Sternal cleft—sternal fissure, bifid sternum—is rare when compared to pectus deformities. It is the least severe form of sternal defects which also include thoracic ectopia cordis, cervical ectopia cordis and thoracoabdominal ectopia cordis. An infant with sternal cleft has a partial or total separation of the sternum, and a normally positioned intrathoracic heart [1,2,3,4,5] (Fig. 1).
The sternum is a mesodermal structure and has a common origin with the pectoral muscles [6]. Lateral mesodermal plates move ventrally during the sixth week of gestation, creating two parallel mesenchymal strips. These strips begin to unite craniocaudally, creating the sternal corpus and partially the manubrium during the seventh week. This union is completed by the end of the tenth week of gestation. The sternum originates as cartilage, then multiple ossification centres develop in a craniocaudal sequence. It is believed that the isolated sternal defect results from the failure of the mesenchymal plate fusion process during the eighth week of gestation [7,8,9].
Sternal cleft can be classified as complete and incomplete [10]. A complete cleft is the rarest form. Incomplete clefts are either superior of inferior. Inferior clefts may occur as an isolated entity, although it is generally associated with anomalies like ectopia cordis, or thoracoabdominal ectopia cordis–Cantrell’s pentalogy. Superior clefts are often an isolated entity [11].
When the sternal cleft is associated with defects of the pericardium, anterior diaphragm and abdominal wall, early surgical repair through a midline thoracoabdominal incision is obligatory [1, 2]. The pericardium is repaired, the diaphragm is attached anteriorly, the sternal bars are approximated and the diastasis recti is corrected. Early repair, when the chest wall is more flexible, is preferred if the infant is healthy enough to tolerate the operation. Although ectopia cordis is generally incompatible with life, thoracoabdominal ectopia cordis has a better prognosis [2].
When the defect is confined to the sternum and is covered with skin, surgical repair may often be elective [2]. Various techniques to correct sternal defects have been described since 1888 when Lannelongue reported the first limited repair [12,13,14,15,16,17,18,19], yet primary repair has become the treatment of choice in infants [20,21,22]. Burton described the first true surgical repair of sternal cleft achieved by placing a costal cartilage graft over the defect in 2 cases in 1949 [14]. Again in 1949, Maier performed the first primary closure of sternal cleft on a 6-week-old child, stressing the need for early correction [15]. In 1958, Sabiston described sliding chondrotomies, increasing the length of costal cartilages by oblique incisions [16]. In 1964, Meissner described his technique where he divided the costal cartilages laterally and rotated them medially to cover the sternal defect [17]. Repair with different autologous grafts like costal cartilage or ribs [20,21,22], and with prosthetic materials like acrylic, silicone, Marlex mesh, or titanium plate have all been reported [19,20,21, 23].
Surgical Treatment of Sternal Cleft—Various Techniques
Simple Primary Repair
Primary repair of the defect by direct approximation of the sternal bars by simple sutures is the treatment of choice whenever it is possible as it reduces the problems with the prosthetic materials and the troubles in the osteocartilaginous cicatrization that may develop in the chondrocostal division procedures [9, 20, 24]. The age of the patient is the key factor for primary repair as in the neonatal period the chest wall is more flexible and has a lower resistance, allowing an easy approximation of the sternal bars without any compression on the mediastinal structures, especially on the heart [9, 21, 25]. Primary repair becomes very difficult after the neonatal period and almost impossible after the first year of life, when more extensive techniques should be applied [9].
For a primary repair, a midline vertical incision is made overlying the sternal cleft. Subcutaneous flaps are created, followed by the mobilization of the pectoralis major muscles, then the endothoracic fascia is dissected off the sternal bars and the pericardium is released. The “U” defect at the base of the cleft is turned into a “V” by excising a wedge of cartilage to facilitate the approximation of the sternal bars without any tension (Fig. 2a). Nonabsorbable sutures are placed in interrupted fashion around the bars to reapproximate them [9, 21] (Fig. 2b).
Primary Repair with Chondrotomies
The use of multiple bilateral oblique—or sliding—chondrotomies reduces the internal organ compression in older patients [16]. These chondrotomies increase the chest wall dimensions and flexibility. The technique is useful in older infants and children with a less flexible chest and a widening defect [1]. As a modification, the cartilages can also be divided laterally and swung medially to cover the sternal defect [1, 17].
For a repair with chondrotomies, the sternal bars are prepared as in the simple primary repair. A sliding oblique chondrotomy is performed bilaterally on the first three to five costal cartilages, then the sternal bars are advanced from both sides to meet in the midline [26] (Fig. 3). If the attempts to approximate the sternal bars are unsuccessful due to the unyielding sternoclavicular joints, the clavicles can be fractured at the junction of the medial and lateral two-thirds and then the sternal bars are approximated [27]. As an alternative, a partial resection of the first three costal cartilages can be performed bilaterally, followed by the disruption of the sternoclavicular junction on both sides [9]. In these patients, the repair is performed with transsternal steel wires. The detached major pectoral muscles are sutured back on the area of costal slide to close the dead space—if any—on the costal cartilages on both side. A mediastinal and a subcutaneous drain are placed, followed by wound closure [26, 27].
Autologous Repair
Autologous repair techniques are suitable for older children who are not suitable for primary repair. It should be preferred as it avoids problems related to the implanting of prosthetic materials.
de Campos described his autologous repair technique in 1998 [11]. In this technique, a posterior sternal wall is created with periosteal flaps from the sternal bars. The periosteum of each bar is incised on its lateral border and elevated from its anterior and medial surfaces (Fig. 4a). Both flaps are then turned inside out and sutured together in the midline with absorbable sutures, providing a stable posterior sternal wall. Two or three chondral grafts are resected subperichondrially, and the gap between the two sternal bars is filled with these grafts. Then the cartilaginous bridge at the distal end of the cleft is resected to facilitate the approximation of the sternal bars (Fig. 4b). Approximation, also the stabilization of the chondral grafts is provided by steel wires [11, 28]. Servical muscle-sparing is mostly not needed with this technique. Pectoral muscles are sutured in the median plane.
a The periosteum of each bar is incised on its lateral border and elevated from its anterior and medial surfaces. Then the cartilaginous bridge at the distal end of the cleft is resected to facilitate the approximation of the sternal bars. b Both flaps are then turned inside out and sutured together in the midline with absorbable sutures, providing a stable posterior sternal wall. Two or three chondral grafts are resected subperichondrially, and the gap between the two sternal bars is filled with these grafts. Approximation, also the stabilization of the chondral grafts is provided by steel wires
Primary approximation and the use of autogenous tissue rather than synthetic materials are the two main advantages of this technique. Cardiac compression is less likely to develop with this procedure as periosteal flaps enlarge the thoracic perimeter. Moreover, periosteal flaps bridge confers an anatomical substract for osteogenesis, allowing a neosternum remodelling. This technique allows normal growth of the chest wall and provides sufficient strength and rigidity in the neosternum [28].
Similar to this periosteal flaps procedure, other autologous repair techniques such as “bilateral sternal bar turnover flaps,” “double osteochondroplasty flap,” or “reversed sternal plate flaps” techniques can be performed for the correction of sternal clefts [29,30,31•]. Reversed sternal flaps are a good option for older patients. Autologous split rib grafts can also be an alternative for this purpose [32].
Repair Using Synthetic Materials
Although the use of synthetic materials such as Marlex, stainless steel mesh, silicone, acrylic, Teflon, Prolene mesh, absorbable polyglactin plate, and porous polyethylene implant are reported to be associated with tissue reactions and increased risk of infection, they appear to provide a much more rigid frame when compared to autologous tissues [33,34,35,36]. Besides, the therapy can be affected by the limited availability of autologous tissues in infants.
Advantages of titanium plates include higher strength and stiffness, light weight, plasticity, resistance to infection, good histocompatibility, and low interference with magnetic resonance imaging and computed tomography [34•]. Displacement, migration, erosion into vital structures or infection are the potential complications as with other prosthetic materials.
In some older children and teenagers, a rib homograft can be harvested to place into the defect, and the defect can then be covered by a prosthetic mesh. The cervical muscles inserting into the separated sternal bars are mobilized and then reinserted in the midline [9].
Since the synthetic prosthetic materials will not grow with the child, another surgical intervention may be necessary in the future. Depending on the child’s age and availability of autologous tissues, either another synthetic material can be inserted or an attempt can be made to perform a primary closure using autologous tissues [34•]. To overcome this problem, an alternative solution can be using absorbable mesh plates as a prosthetic material [35•]. The use of an absorbable polyglactin plate allows to maintain any osseous or cartilage autografts, to leave a sufficient volume for the rib cage, and to avoid synthetic materials that may hamper growth.
Repair Using Muscle Flaps
Muscle or musculocutaneous flaps, such as pectoralis major muscle or rectus abdominis musculocutaneous flaps, can also be used for the repair of sternal cleft, either on their own or together with autologous or synthetic materials [36,37,38].
For a repair with pectoralis major muscle flaps, the parasternal origins of both pectoralis major muscles are released and the muscles are elevated on their thoracoacromial circulation. Each pectoralis major muscle is advanced to the midline and approximated over the pericardium. In case of a complete sternal cleft, the upper abdominal closure can be performed by direct approximation of the rectus abdominis muscles. Drains should be placed above and below the pectoralis muscle flaps and the overlying fasciocutaneous flaps are advanced to achieve skin closure. For inferior or total clefts bilateral contiguous pectoralis major and rectus abdominis muscle flaps can also be used [38].
Conclusions
Sternal cleft is an uncommon congenital chest wall deformity. The optimal correction can be achieved by primary repair of the defect in patients younger than 1 year of age, especially during the neonatal period, mostly with an easy technique. In older children, more extensive techniques are applied, using either autologous tissues or synthetic prosthetic materials, with satisfactory functional and aesthetic results.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Shamberger RC. Chest wall deformities. In: Shields TW, editor. General thoracic surgery. Malvern: Williams & Wilkins; 1994. p. 529–57.
Fonkalsrud EW. Chest wall abnormalities. In: Baue AE, Geha AS, Hammond GL, Laks H, Naunheim KS, editors. Glenn’s thoracic and cardiovascular surgery. Stamford: Appleton & Lange; 1996. p. 581–92.
Shamberger RC, Hendren WH III. Congenital deformities. In: Pearson FG, Deslauriers J, Ginsberg RJ, Hiebert CA, McKneally MF, Urschel HC, editors. Thoracic surgery. New York: Churchill Livingstone; 1995. p. 1189–209.
Sabiston DC Jr. Disorders of the sternum and the thoracic wall congenital deformities of the chest wall. In: Sabiston Jr DC, Spencer FC, editors. Gibbon’s surgery of the chest. Philadelphia: WB Saunders Company; 1990. p. 418–37.
Welch KJ. Chest wall deformities. In: Holder TH, Ashcraft K, editors. Pediatric surgery. Philadelphia: WB Saunders Company; 1980. p. 162–82.
Heron D, Lyonnet S, Iserin L, et al. Sternal cleft: case report and review of a series of nine patients. Am J Med Genet. 1995;59:154–6.
O’Neal M, Dwornik J, Ganey T, et al. Postnatal development of the human sternum. J Pediatric Orthop. 1998;18:398–405.
Kotzot D, Huk W, Pfeiffer R. Genetic counseling of cleft sternum. Genet Couns. 1994;5:147–50.
Acastello E, Majluf R, Garrido P, Barbosa LM, Peredo A. Sternal cleft: a surgical opportunity. J Pediatr Surg. 2003;38:178–83.
Samarrai AR, Charmolockly AM, Attra AA. Complete cleft sternum: classification and surgical repair. Int Surg. 1985;70:71–3.
de Campos JRM, Filomeno LT, Fernandez A, Ruiz RL, Minamoto H, de Campos Werebe E, Jatene FB. Repair of congenital sternal cleft in infants and adolescents. Ann Thorac Surg. 1998;66:1151–4.
Ravitch MM. Congenital deformities of the chest wall and their operative correction. Philadelphia: Saunders; 1977. p. 23–5.
Lannelongue M. De le´ctocardie et de sa cure par autoplasie. Ann Med Chir. 1888;4:101.
Burton J. Method of correction of ectopia cordis. Arch Surg. 1947;54:79–84.
Maier HC, Bortone F. Complete failure of sternal fusion with herniation of perdicardium. Report of a case corrected surgically in infancy. J Thorac Surg. 1949;18:851–9.
Sabiston DC. The surgical management of congenital bifid sternum with partial ectopia cordis. J Thorac Surg. 1958;35:118–22.
Meissner F. Fissura sterni congenita. Zentralbl Chir. 1964;89:1832–9.
Kontiris A, Tsironis A. Bifid sternal-successful repair by use of and acrylic plaque: report of a case. J Int Col Surg. 1964;41:301–7.
Daoud S, Jacob Y, Lefort G, et al. Correction de la bifide sternale par une prothese en silastic. Chir Pediat. 1964;21:415–7.
Greenberg BM, Becker JM, Pletcher BA. Congenital bifid sternum: repair in early infancy and literature review. Plast Reconstr Surg. 1991;88:886–9.
Shamberger R, Welch K. Sternal defects. Pediatr Surg Int. 1990;5:154–64.
Knox L, Tuggle D, Knoxtt-Craig CJ. Repair of congenital sternal cleft in adolescence and infancy. J Pediatr Surg. 1994;29:1513–6.
Hebra A, Davidoff A, O’Neill J. Neonatal sternal cleft associated with an extrathoracic cystic mass. J Pediatric Surg. 1997;32:627–30.
Yavuzer S, Kara M. Primary repair of a sternal cleft in an infant with autogenous tissues. Interact CardioVasc Thorac Surg. 2003;2:541–3.
Hazari A, Mercer N, Pawade A, et al. Superior sternal cleft: construction with a titanium plate. Plast Reconstruct Surg. 1998;101:167–70.
Muthialu N. Primary repair of sternal cleft in infancy using combined periosteal flap and sliding osteochondroplasty. Interact Cardiovasc Thorac Surg. 2013;16(6):923–5.
Mathai J, Cherian VK, Chacko J, Sen S, Karl S, Pandyan MS, Mathew AK. Bridging the cleft over the throbbing heart. Ann Thorac Surg. 2006;82(6):2310–1.
de Campos JR, Das-Neves-Pereira JC, Velhote MC, Jatene FB. Twenty seven-year experience with sternal cleft repair. Eur J Cardiothorac Surg. 2009;35(3):539–41.
Chia HL, Yeow VK. Repair of inferior sternal cleft using bilateral sternal bar turnover flaps in a patient with pentalogy of cantrell. Arch Plast Surg. 2014;41(1):77–80.
Ballouhey Q, Armendariz M, Vacquerie V, Marcheix PS, Berenguer D, Mouliès D, Fourcade L. Primary repair of sternal cleft with a double osteochondroplasty flap. Interact Cardiovasc Thorac Surg. 2013;17(6):1036–7.
• Elsayed H, Soliman S; Reversed autogenous sternal plate flaps for treatment of sternal clefts: a novel technique. J Pediatr Surg 2015;50(11):1991–4. This article showed that reversed sternal flaps were a good option for older patients.
Hill CA, Argenta LC, Hines M. Superior sternal cleft repair using autologous rib grafts in an infant with complex congenital heart disease. Ann Thorac Surg. 2007;84(2):673–4.
Baqain EB, Lataifeh IM, Khriesat WM, Fraiwan NM, Armooti MA. Primary repair of a large incomplete sternal cleft in an asymptomatic infant with Prolene mesh. J Pediatr Surg. 2008;43(10):e39–41.
• Qin T, Liu Y, An Y; Repair of inferior sternal cleft using a titanium plate in an infant with pentalogy of cantrell. J Card Surg. 2016;31(11):700–2. This article showed the advantages of titanium plates when compared to other synthetic materials used for the repair of sternal clefts.
• Al-Yamani M, Lavrand F, Thambo JB, Roubertie F; Upper sternal cleft with a complex congenital heart defect: repair in a single stage. Ann Thorac Sur. 2016;101(2):760–2. This article revealed the advantages of using an absorbable polyglactin plate for the repair of sternal clefts.
Shen W, Cui J, Chen J, Zou J, Ji Y, Chen H. Reconstruction of sternal cleft and aplasia cutis with a medpor and a rectus abdominis musculocutaneous flap. J Craniofac Surg. 2012;23(1):169–71.
Snyder BJ, Robbins RC, Ramos D. Primary repair of complete sternal cleft with pectoralis major muscle flaps. Ann Thorac Surg. 1996;61:983–4.
Hochberg J, Ardenghy MF, Gustafson RA, Murray GF. Repair of thoracoabdominal ectopia cordis with myocutaneous flaps and intraoperative tissue expansion. Plast Reconstr Surg. 1995;95:148–51.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Korkut Bostanci and Davor Stamenovic declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical collection on Thoracic Surgery.
Rights and permissions
About this article
Cite this article
Bostanci, K., Stamenovic, D. Repair of Sternal Cleft in Children. Curr Surg Rep 6, 8 (2018). https://doi.org/10.1007/s40137-018-0205-x
Published:
DOI: https://doi.org/10.1007/s40137-018-0205-x