Abstract
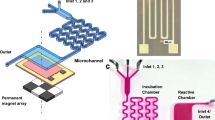
This paper addresses an immunoreaction period-independent microfluidic diagnostic method for the detection of prostate-specific antigen. Immunoreaction-based biosensors generally use sandwich binding of a capture antibody, followed by an antigen, and then a detection antibody with a fluorescent label. Fluorescent intensity is proportional not only to the antigen density in a sample but also to the immunoreaction period. Generally, immunoreaction-based sensors are dependent on the immunoreaction period because the amount of antigen bound to fluorescent labels increases as the immunoreaction period increases. A wash-out step is suggested to eliminate dependence on a specific reaction period. Further, the optimal time to start the wash-out step was investigated. For the conventional detection process, fluorescent intensity increases constantly as the immunoreaction period increases, even though the antigen concentration in the sample has not changed. After applying the wash-out process, however, the fluorescent intensity was maintained over a 15-min period. Consequently, even though a user may miss the optimal reading time for the assay, an accurate result can be reported because the wash-out step enables microfluidic diagnostic devices to maintain a constant fluorescent intensity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wu, J., Fu, Z., Yan, F. & Ju, H. Biomedical and clinical applications of immunoassays and immunosensors for tumor markers. Trends Analyt. Chem. 26, 679–688 (2007).
Okada, H., Hosokawa, K. & Maeda, M. Power-free microchip immunoassay of PSA in human serum for point-of-care testing. Anal. Sci. 27, 237 (2011).
Saad, F. The treatment continuum in the management of prostate cancer patients: What’s new? Eur. Urol. 5, 539–542 (2006).
Grönberg, H. Prostate cancer epidemiology. The Lancet 361, 859–864 (2003).
Ertürk, G., Hedström, M., Tümer, M.A., Denizli, A. & Mattiasson, B. Real-time prostate-specific antigen detection with prostate-specific antigen imprinted capacitive biosensors. Anal. Chim. Acta 891, 120–129 (2015).
D’Orazio, P. Biosensors in clinical chemistry -2011 update. Clin. Chim. Acta 412, 1749–1761 (2011).
Lee, T.M.-H. Over-the-counter biosensors: Past, present, and future. Sensors 8, 5535–5559 (2008).
Robinson, G. The commercial development of planar optical biosensors. Sens. Actuators B Chem. 29, 31–36 (1995).
Barbosa, A.I., Castanheira, A.P., Edwards, A.D. & Reis, N.M. A lab-in-a-briefcase for rapid prostate specific antigen (PSA) screening from whole blood. Lab Chip 14, 2918–2928 (2014).
Lee, S. et al. Improved porous silicon microarray based prostate specific antigen immunoassay by optimized surface density of the capture antibody. Anal. Chim. Acta 796, 108–114 (2013).
Gembitsky, D.S., Lawlor, K., Jacovina, A., Yaneva, M. & Tempst, P. A prototype antibody microarray platform to monitor changes in protein tyrosine phosphorylation. Mol. Cell. Proteomics 3, 1102–1118 (2004).
Saviranta, P. et al. Evaluating sandwich immunoassays in microarray format in terms of the ambient analyte regime. Clin. Chem. 50, 1907–1920 (2004).
Yan, M. et al. Fluorescence immunosensor based on p-acid-encapsulated silica nanoparticles for tumor marker detection. The Analyst 137, 2834–2839 (2012).
Lin, P., Wang, Y.L., Feng, J.J., Zou, Z.H. & Yang, Q. Competitive enzymatic fluorescence immunoassay for human IgG by using a temperature-sensitive phase separating polymer with regulated phase transition temperature. Chinese J. Chem. 26, 794–798 (2008).
Hosokawa, K., Omata, M. & Maeda, M. Immunoassay on a power-free microchip with laminar flow-assisted dendritic amplification. Anal. Chem. 79, 6000–6004 (2007).
Schweitzer, B. et al. Immunoassays with rolling circle DNA amplification: a versatile platform for ultrasensitive antigen detection. Proc. Natl. Acad. Sci. U.S.A. 97, 10113–10119 (2000).
Healy, D.A., Hayes, C.J., Leonard, P., McKenna, L. & O’Kennedy, R. Biosensor developments: application to prostate-specific antigen detection. Trends Biotechnol. 25, 125–131 (2007).
Kim, Y.C., Kim, S.-H., Kim, D., Park, S.-J. & Park, J.-K. Plasma extraction in a capillary-driven microfluidic device using surfactant-added poly (dimethylsiloxane). Sens. Actuators B Chem. 145, 861–868 (2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H.J., Kim, B.H. & Seo, Y.H. Immunoreaction-based Microfluidic Diagnostic Device for the Detection of Prostate-Specific Antigen. BioChip J 12, 154–162 (2018). https://doi.org/10.1007/s13206-017-2208-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-017-2208-6