Abstract
Rewetting is the most common restoration approach for milled peatlands in Europe, with the aim of creating suitable conditions for the development of peatland specific plant cover and carbon accumulation. Therefore, it is important to determine if milled peatlands become functionally and structurally similar to their undisturbed counterparts. We measured plant functional type (PFT) cover and biomass, bryophyte production and CO2 fluxes on three rewetted peatlands in Estonia restored 4, 15, and 35 years before the measurements and compared observations at rewetted sites to two nearby reference bogs. We hoped to better understand whether structure and function return at rewetted sites over time. Differences in vegetation composition and CO2 fluxes between the sites were greater for rewetted than undisturbed sites. The most recently rewetted site was mainly covered in bare peat and Eriophorum vaginatum and was a CO2 source. On the rewetted site of 15 years, Sphagnum was present in addition to ombrotrophic sedges, and in the rewetted site of 35 years, lawn-hollow microtopography is starting to develop with various PFTs. Both of these sites were CO2 sinks. Lawn Sphagnum was abundant on the two older rewetted sites, and was connected with CO2 sink functioning in the rewetted sites. Still, hummock Sphagnum species, which were present in undisturbed bogs, were absent from all of the rewetted sites. With time, CO2 fluxes, microtopography and vegetation develop after rewetting in the direction of undisturbed bogs, while vegetation composition still differs from the reference sites even 35 years after rewetting.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Undisturbed peatlands are important carbon sinks in the long term (Yu 2012) as well as a suitable habitat for plant species that have adapted to survive in acidic and waterlogged conditions (Minayeva 2008). Northern peatlands have been widely affected by peat mining for horticulture or energy production (Leifeld et al. 2019), particularly since the 1950 s when peat milling became the main technique for peat extraction. This method involves drainage and the removal of vegetation in large areas, so thin layers of peat can be extracted every summer season.
Excavated peatlands have several negative environmental effects, such as peat loss through mineralisation, high CO2 emissions, fire hazard, no plant diversity and low aesthetic value. The natural revegetation of those site takes a long time, depends on the environmental conditions of the site and usually does not lead to mire-specific plant communities (Lavoie et al. 2003; Graf et al. 2008; Orru et al. 2016). Unrestored milled peatlands are important CO2 sources to the atmosphere due to the low water tables allowing peat mineralisation and sparse or absent vegetation (Strack et al. 2016; Rankin et al. 2018). In Estonian conditions, Salm et al. (2012) have measured the median annual emissions of active peat excavation sites to be 1 741 kg CO2-C ha−1. The main mitigation possibility for those negative impacts is peatland rewetting, which through higher water tables creates suitable conditions for revegetation and thus reduces CO2 emissions (Wilson et al. 2016) and the flammability of these sites (Granath et al. 2016). Although rewetting increases CH4 emissions on restored peatlands, CH4 is a short-lived gas in the atmosphere, rewetting of peatlands has potential to mitigate climate change in the long-term (Günther et al. 2020). Various criteria have been taken into account throughout the history of restoration ecology to assess restoration success. First, biodiversity measures and hydrology were used to indicate the success of restoring the ecosystem, which in recent decades have been integrated with greenhouse gas balances showing recovery of ecosystem functioning (Kløve et al. 2017; Renou-Wilson et al. 2018).
The initial response of the plant community and its diversity to rewetting is complex and depends also on the pre-rewetting state of the peatland (Tuittila et al. 2000). After peatland rewetting or self-recovery, plant cover increases over the time-scale of several decades (Orru et al. 2016; Priede et al. 2016). Furthermore, CO2 fluxes change in time after rewetting (Beyer and Höper 2015; Kløve et al. 2017) estimate based on their experience of greenhouse gas measurements in temperate peatland that rewetted peat extraction sites may become peat accumulating ecosystems after about 30 years. Even after 30 years post rewetting, milled peatlands can remain CO2 sources, but those emissions from rewetted sites tend to be smaller than from active peat extraction sites, especially if Sphagnum is dominating (Samaritani et al. 2011; Vanselow-Algan et al. 2015).
Milled peatlands have been ombrotrophic bogs and in some cases bog remnants border the peat extraction areas. Usually during peat extraction, layers of Sphagnum peat are removed, so more nutrient-rich peat deposits at the bottom of the former mire with varying thickness are left on the site. This leads to the development of wet minerotrophic vegetation (Tuittila et al. 2000; Renou-Wilson et al. 2018), which is different from typical raised bog vegetation (Renou-Wilson et al. 2018). With vegetation succession and distancing peatland surface from the water table minerotrophic vegetation will be replaced by bog species in time (Samaritani et al. 2011).
The rate of vegetation recovery likely depends on successfully raising the water table (Konvalinková and Prach 2014; Strack et al. 2014; Priede et al. 2016), the presence of plant propagules (Campbell and Rochefort 2003; Konvalinková and Prach 2014) and the nutrient status of the site (Komulainen et al. 1999; Konvalinková and Prach 2014; Kozlov et al. 2016; Priede et al. 2016). However, even after decades, rewetted boreal peatlands mostly differ from their reference ecosystem regarding carbon accumulation and vegetation structure (Moreno-Mateos et al. 2012). The quicker return of CO2 sink function has been reported on milled peatlands restored using the moss-layer-transfer technique (Strack et al. 2016; Nugent et al. 2018).
Due to the challenges related to raising and keeping the stable water table close to the peat surface throughout the year (Price et al. 2003; Brown et al. 2017), functional dryness of the residual peat layer (Price and Whitehead 2001; Price et al. 2003), the smaller water holding capacity of developed moss layer (Waddington et al. 2011; McCarter and Price 2015) and the moisture conditions being closely related with the microtopography of the site (Bugnon et al. 1997; Price et al. 1998; Price and Whitehead 2001; Purre and Ilomets 2018) environmental conditions in restored sites may be drier than in undisturbed sites. Restoration sites have generally lower bryophyte and shrub but higher graminoid cover than in undisturbed peatlands (Soini et al. 2010; González et al. 2013; Strack et al. 2016). Some studies report that vegetation in rewetted sites is more heterogeneous than in pristine peatlands due to the patchiness and incompleteness of the plant cover on restoration sites as vegetation cover starts to develop near the drainage ditches and close to existing vegetation (Soini et al. 2010; Laine et al. 2016).
Undisturbed peatlands are CO2 sinks at least over longer time-scales, but CO2 exchange of the peatland can vary annually (Wilson et al. 2016; Kløve et al. 2017; Nugent et al. 2018) stress the lack of knowledge about the greenhouse gas fluxes in rewetted peatlands, especially on sites that have been rewetted for decades. Restored milled peatlands can have larger (Soini et al. 2010; Strack et al. 2016; Wilson et al. 2016; Nugent et al. 2018, 2019) or smaller (Renou-Wilson et al. 2018; Nugent et al. 2019) CO2 uptake than similar eco-hydrologically undisturbed sites. Similar carbon sequestration as in undisturbed peatlands is reported to return sooner to rewetted milled peatlands than the development of typical raised bog species composition (Soini et al. 2010; Renou-Wilson et al. 2018). Although photosynthesis on restored milled peatlands develops akin to undisturbed peatland, ecosystem respiration stays lower (Strack et al. 2016). Therefore, on rewetted sites, Net Ecosystem Exchange (NEE) is reported to be driven by respiration rather than by photosynthesis (Samaritani et al. 2011; Wilson et al. 2016). Due to their transitional stages, rewetted sites have large interannual and within-site variations in CO2 fluxes (Wilson et al. 2016; Nugent et al. 2019), whereas undisturbed peatlands have reached a mature development stage regarding their vegetation, water retention capacity and CO2 balance (Wilson et al. 2016).
Plant communities vary by their photosynthesis and respiration rates. The highest photosynthesis on nutrient-poor peatlands is reported from Eriophorum communities (Beyer and Höper 2015; Wilson et al. 2016), but this is accompanied by higher respiration (Beyer and Höper 2015). Higher CO2 net sink function (Kivimäki et al. 2008) and soil organic matter accumulation (Andersen et al. 2013) have been recorded on plots with mixed graminoid and Sphagnum patches compared to pure graminoid patches. Sphagnum species have lower photosynthetic capacities (Korrensalo et al. 2016) and lower respiration, therefore plots with Sphagnum are larger CO2 sinks than plots with only graminoids (Beyer and Höper 2015). Vascular plants are also important in peatland CO2 exchange, especially by mitigating the effect of drought on CO2 sink functioning (Kuiper et al. 2014). In similar climatic conditions, correlating vegetation cover and CO2 fluxes differs between land-use types (Laine et al. 2016; Strack et al. 2016) — in restored sites, CO2 net uptake increases with vascular plant cover (Strack et al. 2014, 2016), while in undisturbed sites plots with higher moss cover are greater CO2 sinks (Strack et al. 2016).
In this case study, we analyse the success of rewetting by comparing CO2 fluxes and vegetation on relatively similar rewetted milled peatlands and initially eco-hydrologically similar undisturbed bogs located near the disturbed sites. This is done to assess if rewetting or ecosystem recovery leads to ecosystems that functionally converge to the state of reference bogs. For that, we established the following postulates:
-
1)
CO2 fluxes and vegetation structure on rewetted milled peatlands develop in the direction of undisturbed reference bogs;
-
2)
Higher amount of plant above-ground biomass, especially Sphagnum biomass, is related to higher CO2 sink function on rewetted milled peatlands.
Methods
Study Sites
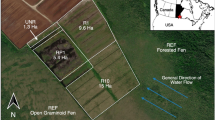
Two paired study sites (Kõrsa and Hara) were selected (Table 1), both of which include rewetted abandoned milled peatlands and remnant open raised bog areas (Fig. 1). The Kõrsa site is located in southwestern Estonia next to an active peat extraction site. The Kõrsa rewetted (KõrsaR) site has self-recovered after the end of peat extraction due to the water level being raised up to the peat surface in 1980 following a damming to create a firewater reservoir (Ramst et al. 2007). Revegetation began at KõrsaR two years later with the self-establishment of E. vaginatum tussocks in 1982. The Hara site is located in Northern Estonia, in Lahemaa National Park. The Hara rewetted site divides into a self-recovered northern part (HaraRN) and an actively rewetted southern part (HaraRS). The water table in HaraRN rose in 2000-2002 due to closing of the bordering ditch, and vegetation started to develop earlier in that area. HaraRS was rewetted in 2012 by the State Forestry Centre of Estonia. No additional restoration measures were applied in addition to rewetting.
The rewetted and undisturbed sites of both paired sites initially had a similar ecohydrological status (raised bog), which is still present in the undisturbed parts of Kõrsa (KõrsaP) and Hara (HaraP). Previously, the vegetation of the rewetted sites was described by Ramst et al. (2007), as the following:
-
HaraRS: Eriophorum vaginatum and bare peat;
-
HaraRN: E. vaginatum, Warnstorfia fluitans, Sphagnum riparium and Sphagnum cuspidatum;
-
KõrsaR: E. vaginatum, Chiloscyphus pallescens, Pleurozium schreberi, Polytrichum strictum, W. fluitans, Brachythecium mildeanum, S. cuspidatum and Sphagnum balticum.
The respective paired sites are located in the same mire-basin, and the distances between the rewetted and undisturbed sites range from 140 m in Hara to 500 m in Kõrsa.
Among the five research sites, in total, 18 plots were fluxed. Four measurement plots per site were established during the previous year (2015). As two of the plots in HaraRN became flooded during the measurement period, they were omitted from the study, and data from two measurement plots in HaraRN were used. The locations of the permanent measurement plots were chosen based on the dominant vegetation and by taking into account its variability between micro-topographic levels. In undisturbed sites and KõrsaR, where microtopography has already developed, two measurement plots were situated on the hummocks and two plots on the lawns of each site. In other sites, two replicates for each vegetation type were established.
Vegetation Analysis
Vegetation analyses were conducted on the plant functional type (PFT) level, and we used the PFT division described in Laine et al. (2012). In our study sites, the following PFTs were present from the larger number of PFTs described by Laine et al. (2012):
-
Hummock Sphagnum: S. rubellum, S. fuscum and S. capillifolium;
-
Lawn Sphagnum: S. angustifolium, S. balticum, S. medium, S. fallax, S. papillosum, S. squarrosum, S. riparium and S. cuspidatum;
-
True mosses: Polytrichum strictum, Warnstorfia fluitans, Chiloscyphus pallescens, Pleurozium schreberi and Brachythecium mildeanum;
-
Evergreen shrubs: Calluna vulgaris, Vaccinium oxycoccus and Andromeda polifolia;
-
Ombrotrophic forbs: Drosera rotundifolia and Rubus chamaemorus;
-
Minerotrophic forbs: Melampyrum spp., Menyanthes trifoliata and Thelypteris palustris;
-
Ombrotrophic sedges: Eriophorum vaginatum;
-
Trees: Pinus sylvestris, Betula pubescens, and Salix spp.
The nomenclature followed Ingerpuu and Vellak (1998) for bryophytes and Leht (2010) for vascular plants.
We measured the plant species coverage (%), vascular plant leaf area index (LAIvasc; m2 m−2), above-ground biomass of PFTs (AGB; g dm−2) and moss production (AGP; g dm−2 year−1) as well as the length increment of mosses (LI; mm year−1). The plant cover of measurement plots was determined visually at the peak of the 2016 growing season (end of July) from inside the CO2 flux measurement collars (four plots per each site/management type combination, but two plots in HaraRN). LAIvasc was determined according to Wilson et al. (2007a) during the CO2 flux measurement campaigns.
Biomass samples were collected from near the measurement plots with vegetation as similar as possible to those in the collars. Vascular plant biomass samples were collected at the end of July 2016 and bryophyte samples at the beginning of October 2016 to capture the maximum biomass of each plant group. We used two plot sizes for the AGB measurements of vascular plants (15 cm radius circular plot) and bryophytes (2.5 cm circular round plot) and collected one vascular plant sample and three bryophyte samples per measurement point (a total of four vascular plant and 12 bryophyte samples from HaraRS, HaraP, KõrsaR and KõrsaP each, and two vascular plant and six bryophyte samples in HaraRN). Only the capitula for Sphagnum species was used as there is no clear distinction between the live and dead material of Sphagnum (Clymo 1970), and the upper 2 cm layer for other bryophytes to obtain biomass samples up to the similar depth of biomass as Sphagnum were collected to determine bryophyte biomass similar to Moore et al. (2002); Laine et al. (2012); Purre et al. (2019b). The collected samples were divided into species level. The sampling and laboratory analysis of biomass is described in Purre et al. (2019b). During the data analysis, the biomass of different species was compiled into PFTs. From the air-dried (65ºC) bryophyte samples, the border of AGP and LI was determined using the innate markers method (Clymo 1970; Pouliot et al. 2010), then separated from the rest of the biomass and weighed (AGP). The LI of ten individuals from the dominant species of each sample were measured with a digital caliper. In HaraRS, bryophytes were absent and thus biomass AGP and LI were considered to be zero.
CO2 Flux Measurements and Data Processing
CO2 measurements were carried out at least once a month during the growing season (May-October 2016). NEE and ecosystem respiration (RECO) was measured on 60 × 60 cm square aluminium collars inserted to about a 20 cm depth, with the rim filled with water to ensure an air-tight fit during flux measurements. We measured CO2 concentrations with the infrared gas analyser Li-6400 (Li-Cor (USA)) from transparent Plexiglas chamber (60 × 60 × 30 cm) with a cooling system. The measurements period was two minutes, and the CO2 content in the chamber was recorded with an interval of 15 s. After measuring the CO2 concentrations in full-light, NEE was measured on two lower irradiation levels by using one or two shades that reduced the photosynthetically active radiation (PAR (µmol m−2 s−1)) reaching the vegetation in the chamber at an average of 65% and 88%, respectively. Lastly, RECO was measured by covering the chamber with an opaque hood. Between each measurement period, the measurement chamber was ventilated. During the measurement campaigns, plant parameters for determining LAIvasc inside the measurement collars were measured according to Wilson et al. (2007a) in addition to recording PAR, the temperature inside the chamber, peat temperatures at 5 and 15 cm depths and the water table (cm).
Input data (PAR, TAIR) for CO2 flux reconstruction were measured with hourly intervals in stations belonging to the Estonian Weather Service. For Hara, the temperature data was obtained from the nearest station in Vanaküla (about 10 km from the site) and radiation data from Harku meteorological station (about 70 km from Hara). For Kõrsa, all meteorological data was obtained from Pärnu-Sauga meteorological station located about 15 km from the site. Those stations were the closest to the study sites where PAR and TAIR were continuously measured, and they were located within a 10 km distance from the sea similarly to the study sites.
The flux rates were estimated based on linear change in CO2 concentrations in time. The linear method was chosen, as this method was considered suitable by Kandel et al. (2016) for CO2 flux calculations in the case of short (few minutes) chamber closure periods (2 min in current study). The measured NEE and RECO fluxes were considered suitable according to the following quality criteria: variation of PAR during the flux measurement not exceeding ±15%, variation of inside temperature of the chamber not varying more than ±5 °C and the determination coefficient (R2) of the measured flux of at least 0.9. Very small fluxes (±0.2 ppm s−1) were accepted regardless of their R2 value. Similar quality criteria in respect of R2 values were used by Järveoja et al. (2016). A total of 215 CO2 flux measurements fulfilled the set criteria and were used for CO2 flux reconstructions. Photosynthesis (Pg) was calculated by adding RECO to NEE.
CO2 fluxes were reconstructed for the period from the beginning of May until the end of September 2016 at each site. With these reconstructions, based on measured and calculated CO2 fluxes and other parameters (PAR, LAIvasc and air temperature (TAIR)), models were created for relating differences in measured CO2 fluxes with differences in input parameters for reconstructing the whole growing season CO2 fluxes. CO2 flux and LAIvasc reconstruction was carried out in program R version 3.2.2 package nlme (Linear and Nonlinear Mixed Effects Models, ver. 3.1 – 121; Pinheiro et al. (2015)). Gaussian curves were fitted to LAIvasc values, which were calculated according to the vegetation parameters measured during the CO2 measurement campaigns for reconstructing the change in LAIvasc during the vegetation season as described by Wilson et al. (2007a) in each measurement collar.
The gross photosynthesis (Pg (mg CO2 m−2 h−1)) model uses the saturating response to PAR (Eq. 1) and records the change in LAIvasc during the vegetation season:
where Pmax is the maximum photosynthesis at light saturation, k and s are respectively the PAR and LAIvasc values when Pg reaches half of its maximum level.
The respiration model (Eq. 2) expresses an exponential response of ecosystem respiration (RECO (mg CO2 m−2 h−1)) to the temperature inside the chamber (TAIR).
Where parameters r0 and b are respectively the respiration at the 0 °C temperature and the sensitivity of respiration to air temperature, and TAIR is the air temperature (°C). CO2 measurements and reconstructions are described in more detail in Purre et al. (2019a, b).
Data Analysis
Data analyses were conducted with IBM SPSS ver. 23. As the data did not fulfil the requirements for parametric data analysis according to the Shapiro-Wilk test, non-parametric data analysis methods were chosen. The Kruskal-Wallis and Mann-Whitney tests with Bonferroni correction for the pairwise comparison of vegetation parameters and CO2 fluxes between the sites were applied. Spearman correlations were used to relate separate plant group abundances with different parameters of CO2 fluxes (NEE, Pg, RECO) in rewetted and undisturbed peatlands. Generalized linear mixed models (GLMMs) were applied on data from rewetted milled peatlands to determine the effect of site, microtopography and time since rewetting (fixed factors) on CO2 fluxes (growing season NEE, Pg or RECO as target variables), biomass of studied plant functional types (PFTs) were incorporated in the models as random factors. For information criterion of the GLMMs log-likelihood was used, lower log-likelihood values showing better model fit. The results were considered statistically significant if p < 0.05. Average values are reported with standard errors.
The multivariate analysis methods Redundancy Analysis (RDA) and Detrended Correspondence Analysis (DCA) were applied in PC-ORD ver. 7 to relate the abundances of PFTs and CO2 fluxes on rewetted and undisturbed sites. In RDA, the response variables were standardised and a randomisation test was applied to test for any significant relationship between the PFT and CO2 flux matrices. DCA was used to find the main gradients in PFT and CO2 flux data using time since rewetting and the site as supplementary variables.
Results
Vegetation
Vegetation varied between rewetted and undisturbed sites and between all rewetted sites, while small differences also occurred between both undisturbed sites (Fig. 2). More PFTs were present on undisturbed and older rewetted sites, while many PFTs such as Sphagnum and evergreen shrubs were absent from the recently rewetted HaraRS. Evergreen shrubs such as C. vulgaris and A. polifolia had higher cover in undisturbed sites, while V. oxycoccus was present with low cover only in KõrsaR. Evergreen shrub biomass was absent or lower in rewetted sites compared to undisturbed sites (Appendix S1). Ombrotrophic forbs R. chamaemorus and D. rotundifolia were only present in undisturbed plots, but with relatively low cover (0.5-3%). Only in KõrsaR minerotrophic forbs like Melampyrum species and T. palustris were present. Tree seedling of Salix spp., Betula spp. and P. sylvestris had about 1% cover on all sites, or were absent.
Average cover (in %) of vascular plant (a) and bryophyte (b) plant functional types of the study sites (±SE). Different small case letters indicate statistically significant differences between the sites (p < 0.05). Statistical significance was tested using the Kruskal-Wallis test, and pairwise comparison was concluded using the Mann-Whitney test with Bonferroni correction
In undisturbed sites, hummock (S. fuscum, S. rubellum, S. angustifolium) and lawn (S. medium, S. balticum, S. papillosum) Sphagnum species were present in relatively similar cover (ranging from 5% (S. balticum in Kõrsa) to 45% (S. rubellum in Kõrsa)). Only lawn species (S. medium, S. fallax, and S. squarrosum) were present in KõrsaR and HaraRN site. True mosses (P. strictum and P. schreberi) had low cover (1-3%) on KõrsaR and HaraRN but were absent from all of the other study sites.
Small differences in plant cover and AGB occurred between the measurement plots in hummocks and lawns. Hummocks had higher AGB (15.3 ± 1.3 g dm−2), AGP of Sphagnum (3.4 ± 0.5 g dm−2 year−1) and mosses (3.5 ± 0.5 g dm−2 year−1) than lawns (AGB 9.9 ± 0.8 g dm−2. AGP of Sphagnum 1.5 ± 0.5 g dm−2 year−1 and mosses 1.5 ± 0.4 g dm−2 year−1) (p < 0.05). In contrast, the cover of lawn Sphagna was higher in lawns (72 ± 14%) than in hummocks (21 ± 15%) (p < 0.05).
Carbon Dioxide Fluxes
Measured NEE and RECO varied spatially to a larger extent in rewetted rather than in undisturbed sites (Appendix S2). CO2 net uptake and Pg increased with higher PAR on rewetted and undisturbed sites.
Reconstructed Pg and NEE did not differ (p > 0.05) between the rewetted and the undisturbed sites, whereas RECO was higher in the rewetted than in the undisturbed sites (p < 0.05; Fig. 3). The respiration model’s parameter r0 was higher in the rewetted (41.8 ± 11.4 mg CO2 m−2 h−1; p < 0.05) than in the undisturbed sites (9.4 ± 3.1 mg CO2 m−2 h−1; Appendix S3). In KõrsaR the reconstructed Pg was higher than in the undisturbed sites and at HaraRS (p < 0.05). Also, KõrsaR had higher RECO than KõrsaP, whereas all of the other sites had a similar RECO. Although there were no differences in the model parameters between HaraRN, HaraRS and HaraP (p > 0.05), Pmax and r0 were higher in KõrsaR than in KõrsaP (p < 0.05) indicating a higher maximum CO2 uptake in case of light saturation and also a higher minimum respiration rate in rewetted sites. Undisturbed sites did not differ according to their CO2 fluxes (p > 0.05). There were no differences in CO2 fluxes between the hummocks and the lawns in the undisturbed sites and KõrsaR (p < 0.05).
Cumulative reconstructed growing season CO2 fluxes in study sites ±SE. Different lower-case letters indicate a statistically significant (p < 0.05) difference in certain CO2 flux component between the sites. The statistical significance was tested using the Kruskal-Wallis test, and pairwise comparison was concluded with the Mann-Whitney test and Bonferroni correction. NEE = Pg – RECO, note that Pg and RECO are always positive for clarity
GLMMs were used specify the effect of site, microtopography and time since rewetting on growing season CO2 flux components (RECO, Pg, NEE) on rewetted peatlands. Although none of the fixed effects and GLMMs were statistically significant, time since rewetting had strongest effect on all of the CO2 flux components (Table 2). In addition to time since rewetting, microtopography and combination of microtopography and site had also relatively strong, but still statistically non-significant effect on RECO.
CO2 Fluxes and Vegetation
CO2 fluxes correlate with every PFT differently between the undisturbed and rewetted plots (Fig. 4, Appendix. S4). In the undisturbed sites, Pg was higher in measurement plots with higher ombrotrophic sedge (E. vaginatum) cover and biomass but lower with higher tree cover, which was related with the higher values of the parameter k indicating the PAR value when Pg reaches half of its maximum value. In the rewetted sites, NEE was higher in the case of higher Sphagnum abundance, and higher photosynthesis rates were connected with the cover of minerotrophic forbs.
Redundancy analysis of plant functional type cover, CO2 fluxes (NEE, Pg, RECO) and CO2 model parameters (Pmax, k, s, r0, b) in the undisturbed (a) and rewetted (b) sites. Only plant functional types, which are significant predictors explaining CO2 fluxes and model parameters, are shown. Minerotrophic forbs and true mosses in the case of undisturbed sites, and ombrotrophic forbs and hummock Sphagnum in the case of rewetted sites, were omitted from the analysis due to their absence from the respective sites. NEE – net ecosystem exchange, Pg – gross photosynthesis, RECO – ecosystem respiration, ombro_sedge – ombrotrophic sedges, Minero_forb – minerotrophic forbs, True_moss – true mosses, Lawn_Sph – lawn Sphagnum
Pg increases with higher bryophyte and vascular biomass in rewetted sites, whereas this correlation was non-significant in the undisturbed sites (Fig. 5). In the undisturbed sites, higher RECO was measured on plots with higher vascular plant biomass, whereas this correlation was insignificant in the rewetted sites. There were no other statistically significant correlations between vascular plant, bryophyte and plant biomass, and Pg, RECO and NEE in the rewetted nor in the undisturbed plots.
With time since rewetting, communities evolve in the direction of undisturbed mires, where several PFTs are present, including Sphagnum and evergreen trees (Fig. 6). HaraRS is characterised by high RECO and biomass of ombrotrophic sedges, HaraRN and KõrsaR contain lawn Sphagnum and Pg, while undisturbed sites (HaraP and KõrsaP) have higher NEE along with the presence of hummock Sphagnum, ombrotrophic forbs and evergreen shrubs. With this transition, high RECO is replaced with higher Pg, and eventually with higher NEE, indicating CO2 sink function during the growing season.
Detrended correspondence analysis (DCA) of plant functional types and CO2 fluxes (NEE, Pg, RECO) and its model parameters (Pmax, k, s, r0, b) in the study sites. The eigenvalues for the first and second axes are 0.181 and 0.067, respectively. NEE – net ecosystem exchange, Pg – gross photosynthesis, RECO – ecosystem respiration, Ombro_sedge – ombrotrophic sedges, Omb_forb – ombrotrophic forbs, Minero_forb – minerotrophic forbs, Ever_shrub – evergreen shrubs, True_moss – true mosses, Lawn_Sph – lawn Sphagnum, Hummock_Sph – hummock Sphagnum
Discussion
Vegetation
Vegetation differed between the undisturbed and rewetted sites. The undisturbed sites had oligotrophic raised-bog vegetation, whereas rewetted sites contained vegetation assemblages more characteristic of mesotrophic environments with higher water tables, an observation previously reported also by others (Tuittila et al. 2000; Samaritani et al. 2011; Renou-Wilson et al. 2018). Commonly, the less humified Sphagnum peat has been removed from abandoned milled peatlands, as the mineral-rich substrate supports the establishment and development of more nutrient demanding plant species. Contrary, oligotrophic vegetation is prevailing in bogs where the peat layer is more nutrient-poor and the water level deeper. After rewetting, vegetation establishment is more rapid and species rich in sites with more nutrients (Komulainen et al. 1999; Kozlov et al. 2016). This could have caused the relatively rapid vegetation succession on KõrsaR where peat ash content is reported to be about twofold higher (2-3%) than in Hara rewetted sites (about 1-2%; Orru 1995). In KõrsaR, a rather diverse peatland community with a thick Sphagnum mat had developed in about 35 years.
Of note, in KõrsaR a thin layer of new peat – an acrotelm – has formed, which suggests that the site is functionally (but not structurally) quite similar to a pristine bog. According to results reported by Lucchese et al. (2010), about a 19 cm thick bryophyte layer would be needed in the Bois-des-Bel restored milled peatland in Canada to mitigate summer water level drawdown; this could be reached about 17 years after restoration. Throughout the study period in KõrsaR and HaraRS, the water level stayed inside the moss layer, mainly near the moss surface, therefore not inhibiting the moss growth during the summer period. In the rewetted sites with thick moss layer in the current study, the moss layer was looser than in the undisturbed reference sites. This was probably due to the higher water table along with the high abundance of hollow Sphagna in the rewetted sites. Hollow Sphagnum could be affected by extreme droughts to a larger degree due to their larger pore size and less connectivity with the residual peat layer (McCarter and Price 2015) than the denser Sphagnum cover of undisturbed bogs, therefore making CO2 exchange on rewetted sites more susceptible to drought impacts.
Some PFTs were lacking or had very low abundances in the rewetted sites but were present in the reference sites. We found significantly lower biomass and cover of evergreen shrubs on the rewetted than in the undisturbed sites, similar to results by Soini et al. (2010) and González et al. (2013), and they were absent from the most recently rewetted sites. Hummock Sphagna, which was present in both undisturbed bog sites was completely absent from the rewetted sites. The low occurrence and dying-off of hummock Sphagnum due to high water tables has been reported previously by Soini et al. (2010) and González et al. (2013). In contrast, Karofeld et al. (2015) recorded relatively high cover of hummock Sphagna and the presence of shrubs on restored milled peatland site where those species were dispersed using the moss-layer-transfer technique (Rochefort et al. 2003). Therefore, the application of this technique could lead to a more diverse vegetation composition of restoration sites.
While vegetation differs between the rewetted sites, being more diverse in the older sites, the vegetation in both undisturbed sites was similar with hummock and hollow vegetation pattern. Hummocks on the two undisturbed sites are typical Calluna vulgaris-Sphagnum fuscum communities, the most common plant associations in Estonian bogs (Masing 1982), and are comparable to the high hummock communities described by Korrensalo et al. (2018). Lawns in the undisturbed sites belong to the tussocky Eriophorum community or the Sphagnum balticum-Sphagnum rubellum community (Masing 1982), described also by Korrensalo et al. (2018) in an undisturbed bog in central Finland as lawn and high lawn communities. A large variation in vegetation between the measurement plots occurred in rewetted sites, especially in the most recently rewetted HaraRS. However, this could also be caused by the relatively low number of measurement plots in each study site and their positioning on the site. In recovering milled peatlands, vegetation is developing in patterns due to large variations in suitable substrate conditions for plant growth (Tuittila et al. 2000; Purre and Ilomets 2018) and the presence of nurse-plant species (Tuittila et al. 2000; Groeneveld et al. 2007), whereas in undisturbed bogs microtopography explains the largest portion of variation in vegetation composition (Korrensalo et al. 2018; Mežaka et al. 2018).
Sphagnum has been considered a keystone genus of peatland restoration (Rochefort 2000). In the newly rewetted HaraRS site, Sphagnum was not yet present in the measurement plots, although some patches of lawn Sphagnum (mainly Sphagnum cuspidatum) were present in depressions with high water level. After rewetting, the height of the water table should remain a few centimetres below the peat surface, which leads to optimal conditions for Sphagnum growth and peat accumulation (Beyer and Höper 2015). Sphagnum has relatively high immigration potential (Campbell et al. 2003) and is abundant on the undisturbed plots bordering the rewetted ones, so further colonisation of Sphagna in recently rewetted sites is expected. In both older rewetting sites, Sphagnum had almost total cover. In addition, in the oldest KõrsaR site, lawn Sphagnum species have created some relatively high hummocks and overgrow E. vaginatum tussocks. The AGP and IL of Sphagnum in the rewetted sites was similar to those reported by Ilomets (1982) in Estonian undisturbed peatlands, while we measured about double the production and somewhat higher IL of Sphagna on the undisturbed sites. This probably results from different methods used for growth measurements (Pouliot et al. 2010), variations in weather conditions (Vitt 1990; Bengtsson et al. 2020) and species composition (Lindholm and Vasander 1990; Bengtsson et al. 2020).
Carbon Dioxide Fluxes
Both the undisturbed sites (KõrsaP and HaraP) and the older rewetted sites (KõrsaR and HaraRN) were CO2 net sinks during the growing season, while the more recently rewetted site (HaraRS) was still a CO2 source. Variations in CO2 fluxes between the rewetted sites are largely due to differences in vegetation, weather and water levels — while some sites are important CO2 sinks (Tuittila et al. 1999; Beyer and Höper 2015; Wilson et al. 2016; Lee et al. 2017; Purre et al. 2019a), others could be small CO2 sources (Tuittila et al. 1999; Waddington and Warner 2001; Beyer and Höper 2015; Purre et al. 2019a). Although rewetted sites could be CO2 sources in the first decades after rewetting, they should become a CO2 net sink with time (Samaritani et al. 2011). Similar (Komulainen et al. 1999) or higher (Soini et al. 2010; Strack et al. 2016) CO2 net uptake on rewetted sites as in reference sites has been reported about ten years after rewetting, which is consistent with our results.
NEE in the rewetted sites is likely connected with differences in RECO rather than Pg (Samaritani et al. 2011; Wilson et al. 2016). Similar to our results from the Hara rewetted site, lower CO2 net uptake due to higher RECO has been reported from newly rewetted sites than from undisturbed bogs (Urbanová et al. 2012). In contrast, in the studies by Soini et al. (2010), Christen et al. (2016) and Strack et al. (2016), higher Pg compensated for high RECO, therefore leading to a higher CO2 net uptake on a rewetted site, which is consistent with our results from the KõrsaR.
CO2 fluxes and model parameters varied more strongly between the measurement plots of the rewetted sites compared to undisturbed sites, as also reported by Soini et al. (2010), Laine et al. (2016) and Strack et al. (2016). Observations may be driven by larger variations in PFT cover in the rewetted sites. Unvegetated plots on rewetted sites remain CO2 sources (Wilson et al. 2016; Purre et al. 2019a) but measurement plots turn from a CO2 source to a sink with increasing plant cover (Strack et al. 2016; Purre et al. 2019a). Respiration on younger sites still containing fragmented vegetation cover and lower diversity of plant species is largely influenced by peat temperature and water table depth, whereas those factors have a smaller effect on sites where vegetation has recovered well (Waddington and Warner 2001; Samaritani et al. 2011; Vanselow-Algan et al. 2015). Therefore, it could be expected that the CO2 sink function will increase and be more stable with secondary succession after rewetting, especially as the actual acrotelm is formed with time.
We detected some effect of site status on plant above-ground biomass, which on rewetted sites had a strong positive correlation with Pg, whereas in undisturbed plots the correlation between plant biomass and Pg was insignificant. Similarly to our rewetted sites, Marinier et al. (2004) reported higher photosynthesis in plots with higher AGB, but plots with high AGB have also been reported to have higher RECO (Marinier et al. 2004; Strack et al. 2016; Brown et al. 2017). This was not the case in our rewetted sites, although in the undisturbed sites, RECO and vascular plant biomass had a strong positive correlation. The lack of correlations between the RECO and vascular plant biomass on rewetted milled peatlands is probably due to the domination of heterotrophic respiration on such sites (Wilson et al. 2007b; Järveoja et al. 2016; Purre et al. 2019a, b; Laine et al. 2016; Strack et al. 2016) also reported interaction between peatland management (undisturbed, rewetted), PFTs and carbon sequestration. According to Järveoja et al. (2016), those correlations depend on water level depth — if the water level is high in restored milled peatlands, bryophyte cover correlates with NEE, Pg and autotrophic respiration, whereas with deeper water table CO2 fluxes correlate with vascular plant cover. Therefore, the different correlations on rewetted and undisturbed sites are consistent with previous studies (Strack et al. 2016) and could be related to differences in water table height and fluctuations on sites with different management.
There are large differences in photosynthetic capacities between PFTs. In the undisturbed sites, we measured higher photosynthesis and maximum photosynthesis rates (Pmax) in plots exhibiting higher E. vaginatum cover. Vascular plant, especially graminoid biomass, has a relatively large impact on NEE in comparison with their abundance (Laine et al. 2012; Hassanpour Fard et al. 2020), due to their high photosynthetic capacity (Komulainen et al. 1999; Kivimäki et al. 2008; Urbanová et al. 2012; Strack et al. 2014; Laine et al. 2016). As E. vaginatum was present or abundant on most of the rewetted plots, the lack of correlation between the sedge cover and photosynthesis on the rewetted sites was unexpected. In addition to having high Pmax, this sedge species also has high light use efficiency (parameter k in the photosynthesis model) (Kivimäki et al. 2008) and high respiration rate (Jordan et al. 2016). Still, in the case of a high water table, rewetted sites with high E. vaginatum cover have a CO2 net sink function, even in unfavourable habitat conditions such as the occasionally lower water table during drought periods (Tuittila et al. 1999).
In the rewetted sites, higher Pg and Pmax were measured with higher evergreen shrub cover. Evergreen shrubs stand out from other vascular plants with low photosynthesis and respiration rates (Laine et al. 2016), while in contrast Korrensalo et al. (2016) reported high Pmax on evergreen shrubs like A. polifolia, C. vulgaris and V. oxycoccus, which are also present in the undisturbed sites and KõrsaR in our study. According to Korrensalo et al. (2016), the Pmax of evergreen shrubs varies between species belonging to the same PFT. Still, the cause of controversies between different studies remains unclear and could be result of a rather low number of measurements that do not cover the whole ecosystem variation.
High photosynthesis in the case of higher evergreen shrub cover in this study could also be connected with higher plant cover and the number of PFTs on the measurement plots in KõrsaR where evergreen shrubs were present. According to Kivimäki et al. (2008), the presence of different PFTs lowers the RECO/Pg ratio, so creating conditions for higher CO2 net uptake as in Kõrsa, while in monostands of E. vaginatum this ratio is higher, which also explains a lower CO2 net uptake, as well as CO2 net emissions from the younger site in this study. According to Hassanpour Fard et al. (2020), the presence of some key species or PFTs either in monostand or in mixed community support the larger carbon accumulation during the growing season than the mixed communities with a different number of PFTs lacking such certain species. Whereas most vascular plants, especially sedges, have high Pg during summer when their LAI is highest, the importance of Sphagnum in CO2 sequestration expresses itself during spring and autumn, when LAIvasc is low (Korrensalo et al. 2017).
In the rewetted sites, CO2 net sink function was larger in plots with higher Sphagnum cover. Sphagnum has lower photosynthetic capacities than vascular plants (Laine et al. 2012; Christen et al. 2016; Korrensalo et al. 2016) and also low respiration rates (Waddington and Warner 2001; Laine et al. 2016), and by increasing soil moisture content, a Sphagnum carpet could reduce soil respiration (Waddington and Warner 2001). However, restoring the Sphagnum carpet may not be enough for CO2 sequestering (Samaritani et al. 2011), especially as a newly formed Sphagnum carpet is sensitive to drier conditions (Tuittila et al. 2004). Therefore, constant high water tables are necessary, which support CO2 accumulation of those sites early on after restoration activities (Günther et al. 2017).
Conclusion
Although vegetation structure on rewetted milled peatlands approached the reference condition with time, some plant functional types present in the undisturbed reference sites such as late succession shrubs and hummock Sphagnum could be absent even decades after rewetting. Vegetation composition developing with time affects the carbon accumulation of rewetted sites. During the studied growing season, milled peatlands rewetted more than 10-years prior were carbon sinks similar to the reference sites, whereas the most recently rewetted site was still a carbon source to the atmosphere. Although graminoids play an important role in the photosynthesis of rewetted sites, as they do in undisturbed reference bogs, the carbon accumulation of rewetted peatlands is related with development of the Sphagnum mat, which is present in the reference sites. A well-developed Sphagnum mat also reflects the development of other environmental variables, of a functioning acrotelm and the development of a C sink function. Thus, a well-developed Sphagnum lawn could be used as an indicator of successful restoration. However, general plant functional type composition can still differ from reference sites in some accounts even several decades after rewetting.
Data Availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
Code Availability
The codes used for data modelling and analysis are available from the corresponding author upon reasonable request.
References
Andersen R, Pouliot R, Rochefort L (2013) Above-ground net primary production from vascular plants shifts the balance towards organic matter accumulation in restored Sphagnum bogs. Wetlands 33:811–821
Bengtsson F, Rydin H, Baltzer JL, Bragazza L, Bu Z-J, Caporn SJ, Dorrepaal E, Flatberg KI, Galanina O, Gałka M, Ganeva A, Goia I, Goncharova N, Hájek M, Haraguchi A, Harris LI, Humphreys E, Jiroušek M, Kajukało K, Karofeld E, Koronatova NG, Kosykh NP, Laine AM, Lamentowicz M, Lapshina E, Limpens J, Linkosalmi M, Ma J-Z, Mauritz M, Mitchell EAD, Munir TM, Natali SM, Natcheva R, Payne RJ, Philippov DA, Rice SK, Robinson S, Robroek BJM, Rochefort L, Singer D, Stenøien HK, Tuittila E-S, Vellak K, Waddington JM, Granath G (2020) Environmental drivers of Sphagnum growth in peatlands across the Holarctic region. Journal of Ecology. https://doi.org/10.1111/1365-2745.13499
Beyer C, Höper H (2015) Greenhouse gas exchange of rewetted bog peat extraction sites and a Sphagnum cultivation site in northwest Germany. Biogeosciences 12:2101–2117
Brown C, Strack M, Price J (2017) The effects of water management on the CO2 uptake of Sphagnum moss in a reclaimed peatland. Mires and Peat 20:1–15
Bugnon J-L, Rochefort L, Price JS (1997) Field experiment of Sphagnum reintroduction on a dry abandoned peatland in Eastern Canada. Wetlands 17:513–517
Campbell DR, Rochefort L (2003) Germination and seedling growth of bog plants in relation to the recolonization of milled peatlands. Plant Ecology 169:71–84
Campbell DR, Rochefort L, Lavoie C (2003) Determining the immigration potential of plants colonizing disturbed environments: the case of milled peatlands in Quebec. Journal of Applied Ecology 40:78–91
Christen A, Jassal RS, Black TA, Grant NJ, Hawthorne I, Johnson MS, Lee SC, Merkens M (2016) Summertime greenhouse gas fluxes from an urban bog undergoing restoration through rewetting. Mires and Peat 17:1–24
Clymo RS (1970) The growth of Sphagnum: methods of measurement. Journal of Ecology 58:13–49
González E, Henstra SW, Rochefort L, Bradfield GE, Poulin M (2013) Is rewetting enough to recover Sphagnum and associated peat-accumulating species in traditionally exploited bogs? Wetlands Ecology and Management 22:49–62
Graf MD, Rochefort L, Poulin M (2008) Spontaneous revegetation of cutwaway peatlands of North America. Wetlands 28(1):28–39
Granath G, Moore PA, Lukenbach MC, Waddington JM (2016) Mitigating wildfire carbon loss in managed northern peatlands through restoration. Scientific Reports 6:28498
Groeneveld EVG, Massé A, Rochefort L (2007) Polytrichum strictum as a nurse-plant in peatland restoration. Restoration Ecology 15:709–719
Günther A, Barthelmes A, Huth V, Joosten H, Jurasinski G, Koebsch F, Couwenberg J (2020) Prompt rewetting of drained peatlands reduces climate warming despite methane emissions. Nature Communications 11:1644
Günther A, Jurasinski G, Albrecht K, Gaudig G, Krebs M, Glatsel S (2017) Greenhouse gas balance of an establishing Sphagnum culture on a former bog grassland in Germany. Mires and Peat 18:1–16
Hassanpour Fard G, Farries E, Bérubé V, Rochefort L, Strack M (2020) Key species superpose the effect of species richness and species interaction on carbon fluxes in a restored Minerotrophic Peatland. Wetlands 40:333–349
Ilomets M (1982) The productivity of Sphagnum communities and the rate of peat accumulation in Estonian bogs. In: Masing V (ed) Peatland ecosystems. Research into the plant cover of Estonian bogs and their productivity. Valgus, Tallinn, pp 102–116
Ingerpuu N, Vellak K (1998) Key of Estonian Bryophytes (Estonian). Tartu, Eesti Loodusfoto
Järveoja J, Peichl M, Maddison M, Soosaar K, Vellak K, Karofeld E, Teemusk A, Mander Ü (2016) Impact of water table level on annual carbon and greenhouse gas balances of a restored peat extraction area. Biogeosciences 13:2637–2651
Jordan S, Strömgren M, Fiedler J, Lundin L, Lode E, Nilsson T (2016) Ecosystem respiration, methane and nitrous oxide fluxes from ecotopes in a rewetted extracted peatland in Sweden. Mires and Peat 17:1–23
Kandel TP, Lærke PE, Elsgaard L (2016) Effect of chamber enclosure time on soil respiration flux: A comparison of linear and non-linear flux calculation methods. Atmospheric Environment 141:245–254
Karofeld E, Müür M, Vellak K (2015) Factors affecting re-vegetation dynamics of experimentally restored extracted peatland in Estonia. Environmental Science and Pollution Research 23:13706–13717
Kivimäki SK, Yli-Petäys M, Tuittila E-S (2008) Carbon sink function of sedge and Sphagnum patches in a restored cut-away peatland: increased functional diversity leads to higher production. Journal of Applied Ecology 45:921–929
Kløve B, Berglund K, Berglund Ö, Weldon S, Maljanen M (2017) Future options for cultivated Nordic peat soils: Can land management and rewetting control greenhouse gas emissions? Environmental Science and Policy 69:85–93
Komulainen V-M, Tuittila E-S, Vasander H, Laine J (1999) Restoration of drained peatlands in southern Finland: initial effects on vegetation change and CO2 balance. Journal of Applied Ecology 36:634–648
Konvalinková P, Prach K (2014) Environmental factors determining spontaneous recovery of industrially mined peat bogs: A multi-site analysis. Ecological Engineering 69:38–45
Korrensalo A, Alekseychik P, Hájek T, Rinne J, Vesala T, Mehtätalo L, Mammarella I, Tuittila E-S (2017) Species-specific temporal variation in photosynthesis as a moderator of peatland carbon sequestration. Biogeosciences 14:257–269
Korrensalo A, Hájek T, Vesala T, Mehtätalo L, Tuittila E-S (2016) Variation in photosynthetic properties among bog plants. Botany 94:1127–1139
Korrensalo A, Kettunen L, Laiho R, Alekseychik P, Vesala T, Mammarella I, Tuittila E-S (2018) Boreal bog plant communities along a water table gradient differ in their standing biomass but not their biomass production. The Journal of Vegetation Science 29:136–146
Kozlov SA, Lundin L, Avetov NA (2016) Revegetation dynamics after 15 years of rewetting in two extracted peatlands in Sweden. Mires and Peat 18:1–17
Kuiper JJ, Mooij WM, Bragazza L, Robroek BJM (2014) Plant functional types define magnitude of drought response in peatland CO2 exchange. Ecology 95:123–131
Laine AM, Bubier J, Riutta T, Nilsson MB, Moore TR, Vasander H, Tuittila E-S (2012) Abundance and composition of plant biomass as potential controls for mire net ecosystem CO2 exchange. Botany 90:63–74
Laine AM, Tolvanen A, Mehtätalo L, Tuittila E-S (2016) Vegetation structure and photosynthesis respond rapidly to restoration in young coastal fens. Ecology and Evolution 6:6880–6891
Lavoie C, Grosvernier P, Girard M, Marcoux K (2003) Spontaneous revegetation of mined peatlands: An useful restoration tool? Wetlands Ecology and Management 11(1–2):97–107
Lee SC, Christen A, Black AT, Johnson MS, Jassal RS, Ketler R, Nesic Z, Merkens M (2017) Annual greenhouse gas budget for a bog ecosystem undergoing restoration by rewetting. Biogeosciences 14:2799–2814
Leht M (2010) Key of estonian plants. Eesti Loodusfoto, Tartu, Eesti Maaülikool
Leifeld J, Wüst-Galley C, Page S (2019) Intact and managed peatland soils as a source and sink of GHGs from 1850 to 2100. Nature Climate Change 9:945–947
Lindholm T, Vasander H (1990) Production of eight species of Sphagnum at Suurisuo mire, southern Finland. Annales Botanici Fennici 27:145–157
Lucchese M, Waddington JM, Poulin M, Pouliot R, Rochefort L, Strack M (2010) Organic matter accumulation in a restored peatland: Evaluating restoration success. Ecological Engineering 36:482–488
Marinier M, Glatzel S, Moore TR (2004) The role of cotton-grass (Eriophorum vaginatum) in the exchange of CO2 and CH4 at two restored peatlands, eastern Canada. Ecoscience 11:141–149
Masing V (1982) The plant cover of Estonian bogs. A structural analysis. In: Masing V (ed) Peatland ecosystems. Research into the plant cover of Estonian bogs and their productivity. Valgus, Tallinn, pp 50–92
McCarter CPR, Price JS (2015) The hydrology of the Bois-des-Bel peatland restoration: hydrophysical properties limiting connectivity between regenerated Sphagnum and remnant vacuum harvested peat deposit. Ecohydrology 8:173–187
Mežaka A, Priede A, Dobkeviča L, Bader MY (2018) Environmental controls of raised-bog vegetation in the Baltic boreo-nemoral zone. Folia Geobotanica. https://doi.org/10.1007/s12224-017-9305-0
Minayeva T (2008) Peatlands and biodiversity. In: Parish et al (eds) Assessment on peatlands, biodiversity and climate change: main report. Global Environment Centre, Kuala Lumpur and Wetlands International, Wageningen, pp 60–98
Moore TR, Bubier JL, Frolking SE, Lafleur PM, Roulet NT (2002) Plant biomass and production and CO2 exchange in an ombrotrophic bog. J Ecol 90:25–36
Moreno-Mateos D, Power ME, Comín FA, Yockteng R (2012) Structural and functional loss in restored wetland ecosystems. PLOS Biology 10:1–8
Nugent KA, Strachan IB, Roulet NT, Strack M, Frolking S, Helbig M (2019) Prompt active restoration of peatlands substantially reduces climate impact. Environmental Research Letters 14:124030
Nugent KA, Strachan IB, Strack M, Roulet NT, Rochefort L (2018) Multi-year net ecosystem carbon balance of a restored peatland reveals a return to carbon sink. Global Change Biology 24:5751–5768
Orru M (1995) Eesti Turbasood (Estonian mires). Eesti Geoloogiakeskus. Estonian Geological Survey, Tallinn
Orru M, Ots K, Orru H (2016) Re-vegetation processes in cutaway peat production fields in Estonia in relation to peat quality and water regime. Environmental Monitoring and Assessment 188. https://doi.org/10.1007/s10661-016-5669-5
Pinheiro J, Bates D, Debroy S, Sarkar D, R Core Team (2015) nlme: Linear and nonlinear mixed effects models. R package version 3.1–121. https://CRAN.R-project.org/package=nlme
Pouliot R, Marchand-Roy M, Rochefort L, Gauthier G (2010) Estimating moss growth in arctic conditions: a comparison of three methods. Bryologist 113:322–332
Price J, Heathwaite AL, Baird AJ (2003) Hydrological processes in abandoned and restored peatlands: An overview of management approaches. Wetlands Ecology and Management 11:65–83
Price J, Rochefort L, Quinty F (1998) Energy and moisture considerations on cutover peatlands: surface microtopography, mulch cover and Sphagnum regeneration. Ecological Engineering 10:293–312
Price J, Whitehead GS (2001) Developing hydrologic thresholds for Sphagnum recolonization on an abandoned cutover bog. Wetlands 21:32–40
Priede A, Mežaka A, Dobkeviča L, Grīnberga L (2016) Spontaneous revegetation of cutaway fens: can it result in valuable habitats? Mires and Peat 18:1–14
Purre A-H, Ilomets M (2018) Relationships between bryophyte production and substrate properties in restored milled peatlands. Restoration Ecology 26:858–864
Purre A-H, Pajula R, Ilomets M (2019a) Carbon dioxide sink function in restored milled peatlands - the significance of weather and vegetation. Geoderma 346:30–42
Purre A-H, Penttilä T, Ojanen P, Minkkinen K, Aurela M, Lohila A, Ilomets M (2019b) Carbon dioxide fluxes and vegetation structure in rewetted and pristine peatlands in Finland and Estonia. Boreal Environment Research 24:243–261
Ramst R, Orru M, Salo V, Halliste L (2007) Eesti mahajäetud turbatootmisalade revisjon. 3. Etapp. Viljandi, Pärnu, Saare ja Hiiu maakond. OÜ Eesti Geoloogiakeskus, Tallinn
Rankin T, Strachan IB, Strack M (2018) Carbon dioxide and methane exchange at a post-extraction, unrestored peatland. Ecological Engineering 122:241–251
Renou-Wilson F, Moser G, Fallon D, Farrell CA, Müller C, Wilson D (2018) Rewetting degraded peatlands for climate and biodiversity benefits: Results from two raised bogs. Ecological Engineering. https://doi.org/10.1016/j.ecoleng.2018.02.014
Rochefort L (2000) Sphagnum — A keystone genus in habitat restoration. Bryologist 103:503–508
Rochefort L, Quinty F, Campeau S, Johnson K, Malterer T (2003) North American approach to the restoration of Sphagnum dominated peatlands. Wetland Ecology and Management 11:3–20
Salm J-O, Maddison M, Tammik S, Soosaar K, Truu J, Mander Ü (2012) Emissions of CO2, CH4 and N2O from undisturbed, drained and mined peatlands in Estonia. Hydrobiologia 692:41–55
Samaritani E, Siegenthaler A, Yli-Petäys M, Buttler A, Christin P-A, Mitchell EAD (2011) Seasonal net ecosystem carbon exchange of a regenerating cutaway bog: How long does it take to restore the C-sequestration function? Restoration Ecology 19:440–449
Soini P, Riutta T, Yli-Petäys M, Vasander H (2010) Comparison of vegetation and CO2 dynamics between a restored cut-away peatland and a pristine fen: Evaluation of the restoration success. Restoration Ecology 18:894–903
Strack M, Cagampan J, Hassanpour Fard G, Keith AM, Nugent K, Rankin T, Robinson C, Strachan IB, Waddington JM, Xu B (2016) Controls on plot-scale growing season CO2 and CH4 fluxes in restored peatlands: Do they differ from unrestored and natural sites? Mires and Peat 17:1–18
Strack M, Keith AM, Xu B (2014) Growing season carbon dioxide and methane exchange at a restored peatland on the Western Boreal Plain. Ecological Engineering 64:231–239
Tuittila E-S, Komulainen V-M, Vasander H, Laine J (1999) Restored cut-away peatland as a sink for atmospheric CO2. Oecologia 120:563–574
Tuittila E-S, Vasander H, Laine J (2004) Sensitivity of C sequestration in reintroduced Sphagnum to water-level variation in a cutaway peatland. Restoration Ecology 12:483–493
Tuittila E-S, Vasander H, Laine J (2000) Impact of rewetting on the vegetation of a cut-away peatland. Applied Vegetation Science 3:205–212
Urbanová Z, Picek T, Hájek T, Bufková I, Tuittila E-S (2012) Vegetation and carbon gas dynamics under a changed hydrological regime in central European peatlands. Plant Ecology and Diversity 5:89–103
Vaneslow-Algan M, Schmidt SR, Greven M, Fiencke C, Kutzbach L, Pfeiffer E-M (2015) High methane emissions dominated annual greenhouse gas balances 30 years after bog rewetting. Biogeosciences 12:4361–4371
Vitt DH (1990) Growth and production dynamics of boreal mosses over climatic, chemical and topographic gradients. Botanical Journal of the Linnean Society 104:35–59
Waddington JM, Lucchese MC, Duval TP (2011) Sphagnum moss moisture retention following the re-vegetation of degraded peatlands. Ecohydrology 4:359–366
Waddington JM, Warner KD (2001) Atmospheric CO2 sequestration in restored mined peatlands. Ecoscience 8:359–368
Wilson D, Alm J, Riutta T, Laine J, Byrne KA, Farrell EP, Tuittila E-S (2007) A high resolution green area index for modelling the seasonal dynamics of CO2 exchange in peatland vascular plant communities. Plant Ecology 190:37–51
Wilson D, Farrell CA, Fallon D, Moser G, Müller C, Renou-Wilson F (2016) Multiyear greenhouse gas balances at a rewetted temperate peatland. Global Change Biology 22:4080–4095
Wilson D, Tuittila E-S, Alm J, Laine J, Farrell EP, Byrne KA (2007b) Carbon dynamics of a restored maritime peatland. Ecoscience 14:71–80
Yu ZC (2012) Northern peatland carbon stocks and dynamics: a review. Biogeosciences 9:4071–4085
Acknowledgements
We thank the Estonian Forest Management Centre for rewetting the Hara site and AS Jiffy Products Estonia for allowing access to the Kõrsa rewetted site for the study. Aleksandra Krijer helped us with biomass measurements in the study sites.
Author information
Authors and Affiliations
Contributions
Both authors contributed to the study conception and design. Anna-Helena Purre conducted the material preparation, fieldwork, laboratory and data analysis, and she wrote the first draft of the manuscript. Mati Ilomets commented on previous versions of the manuscript. Both authors read and approved the final manuscript.
Animal Research (Ethics): Not applicable.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Plant Reproducibility
Not applicable.
Clinical Trials Registration
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Peatlands.
Supplementary Information
ESM 1
(DOCX 106 KB)
Rights and permissions
About this article
Cite this article
Purre, AH., Ilomets, M. Vegetation Composition and Carbon Dioxide Fluxes on Rewetted Milled Peatlands — Comparison with Undisturbed Bogs. Wetlands 41, 120 (2021). https://doi.org/10.1007/s13157-021-01518-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-021-01518-2