Abstract
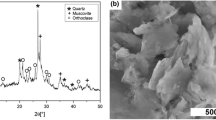
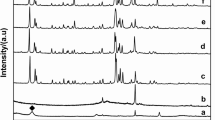
Resource saving and creation of environmentally safe environment are the guidelines of modern world policy in the field of waste management. This paper considers a possible method of utilization of a by-product of aluminum fluoride production—silica gel—as an alternative high-silica raw material for hydrothermal synthesis of LTA-type zeolites. To obtain synthetic zeolites, industrial silica gel was subjected to sulfuric acid purification and used to obtain one of the hydrogel components—sodium silicate. As a result of investigation of the influence of hydrogel molar composition on the phase composition of the obtained samples, the optimal molar ratios of SiO2:Al2O3, Na2O:Al2O3 and H2O:SiO2 were identified. The phase composition of the obtained samples and morphological pattern were evaluated using X-ray diffraction and scanning electron microscopy techniques. According to the obtained results, the use of hydrogel of composition 1.8SiO2:Al2O3:4Na2O:28H2O allows to obtain a monophase of LTA type zeolite with high ion exchange capacity, the particles of which have a regular cubic shape with the size of the main fraction up to 10 μm.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Data Availability
No datasets were generated or analysed during the current study.
References
Popov G, Bolobov V, Zhuikov I, Zlotin V (2023) Development of the kinetic equation of the groove corrosion process for predicting the residual life of oil-field pipelines. Energies 16:7067. https://doi.org/10.3390/en16207067
Bolobov VI, Latipov IU, Zhukov VS, Popov GG (2023) Using the magnetic anisotropy method to determine hydrogenated sections of a steel pipeline. Energies 16:5585. https://doi.org/10.3390/en16155585
Safiullin RN, Safiullin RR, Sorokin KV et al (2024) Integral assessment of influence mechanism of heavy particle generator on hydrocarbon composition of vehicles motor fuel. Int J Eng 37:1700–1706. https://doi.org/10.5829/IJE.2024.37.08B.20
Zubkova O, Alexeev A, Polyanskiy A et al (2021) Complex processing of saponite waste from a diamond-mining enterprise. Appl Sci 11:6615. https://doi.org/10.3390/app11146615
Cheremisina O, Litvinova T, Sergeev V et al (2021) Application of the organic waste-based sorbent for the purification of aqueous solutions. Water 13:3101. https://doi.org/10.3390/w13213101
Gerasimov A, Ustinov I, Zyryanova O (2023) Use of clay-containing waste as pozzolanic additives. J Min Inst 260:313–320. https://doi.org/10.31897/PMI.2023.33
Ponomareva MA, Cheremisina OV, Mashukova YA, Lukyantseva ES (2021) Increasing the efficiency of rare earth metal recovery from technological solutions during processing of apatite raw materials. J Min Inst 252:1–10. https://doi.org/10.31897/PMI.2021.6.13
Litvinova TE, Tsareva AA, Poltoratckaya ME, Rudko VA (2024) The mechanism and thermodynamics of ethyl alcohol sorption process on activated petroleum coke. J Min Inst. https://doi.org/10.21203/rs.3.rs-3308920/v1
Vaiciukynienė D, Jakevicius L, Kantautas A et al (2021) Conversion of silica by-product into zeolites by thermo-sonochemical treatment. Ultrason Sonochem 72. https://doi.org/10.1016/j.ultsonch.2020.105426
Gorlanov ЕS, Leontev LI (2024) Directions in the technological development of aluminium pots. J Min Inst 266:246–259
Rudelis V, Dambrauskas T, Grineviciene A, Baltakys K (2019) The prospective approach for the reduction of fluoride ions mobility in industrial waste by creating products of commercial value. Sustainability 11:634. https://doi.org/10.3390/su11030634
Gineika A, Dambrauskas T, Baltakys K (2021) Synthesis and characterisation of wollastonite with aluminium and fluoride ions. Ceram Int 47:22900–22910. https://doi.org/10.1016/j.ceramint.2021.05.003
Gineika A, Baltakys K, Dambrauskas T (2019) The application of silica gel waste for the two-step synthesis of wollastonite in temperature range of 200–950 °C. J Therm Anal Calorim 138:2263–2273. https://doi.org/10.1007/s10973-019-08481-5
Dambrauskas T, Baltakys K, Grineviciene A, Rudelis V (2021) The effect of various hydroxide and salt additives on the reduction of fluoride ion mobility in industrial waste. Sustainability 13:1554. https://doi.org/10.3390/su13031554
MamchenkovProkof’ev EAVY (2019) Sodium silicate manufacturing from modified silica gel as by-product of aluminum fluoride. ChemChemTech 62:89–93. https://doi.org/10.6060/ivkkt.20196203.5949
Wong EY, Stenstrom MK (2021) The usage of calcium phosphate systems for onsite defluoridation treatment. J Environ Sci Heal Part A 56:1189–1195. https://doi.org/10.1080/10934529.2021.1973311
Kakasor Ismael Jaf D, Ismael Abdulrahman P, Salih Mohammed A et al (2023) Machine learning techniques and multi-scale models to evaluate the impact of silicon dioxide (SiO2) and calcium oxide (CaO) in fly ash on the compressive strength of green concrete. Constr Build Mater 400:132604. https://doi.org/10.1016/j.conbuildmat.2023.132604
Kornev YV, Semenov NA, Vlasov AN, Valiev KK (2021) Reinforcing effects in elastomeric composites, filled with particles of mineral fillers, based on silicon dioxide and carbon. J Phys Conf Ser 1942:012031. https://doi.org/10.1088/1742-6596/1942/1/012031
Endzhievskaya IG, Demina AV, Lavorenko AA (2020) Synthesis of a mineralizing agent for Portland cement from aluminum production waste. IOP Conf Ser Mater Sci Eng 945:012062. https://doi.org/10.1088/1757-899X/945/1/012062
Kakasor Ismael Jaf D, Abdulrahman AS, Abdulrahman PI et al (2023) Effitioned soft computing models to evaluate the impact of silicon dioxide (SiO2) to calcium oxide (CaO) ratio in fly ash on the compressive strength of concrete. J Build Eng 74:106820. https://doi.org/10.1016/j.jobe.2023.106820
de Aquino TF, Estevam ST, Viola VO et al (2020) CO2 adsorption capacity of zeolites synthesized from coal fly ashes. Fuel 276:118143. https://doi.org/10.1016/j.fuel.2020.118143
Tran NM, Nam Y, Yoo H (2022) Fabrication of dendritic fibrous silica nanolayer on optimized water-glass-based synthetic nanosilica from rice husk ash. Ceram Int 48:32409–32417. https://doi.org/10.1016/j.ceramint.2022.07.184
Rajan HS, Kathirvel P (2021) Sustainable development of geopolymer binder using sodium silicate synthesized from agricultural waste. J Clean Prod 286:124959. https://doi.org/10.1016/j.jclepro.2020.124959
Tran-Nguyen PL, Ly K-P, Thanh LHV et al (2021) Facile synthesis of zeolite NaX using rice husk ash without pretreatment. J Taiwan Inst Chem Eng 123:338–345. https://doi.org/10.1016/j.jtice.2021.05.009
Tran YT, Lee J, Kumar P et al (2019) Natural zeolite and its application in concrete composite production. Compos Part B Eng 165:354–364. https://doi.org/10.1016/j.compositesb.2018.12.084
Narayanan S, Tamizhdurai P, Mangesh VL et al (2021) Recent advances in the synthesis and applications of mordenite zeolite – review. RSC Adv 11:250–267. https://doi.org/10.1039/D0RA09434J
Cao P, Li G, Jiang H et al (2021) Extraction and value-added utilization of alumina from coal fly ash via one-step hydrothermal process followed by carbonation. J Clean Prod 323:129174. https://doi.org/10.1016/j.jclepro.2021.129174
Nazir LSM, Yeong YF, Chew TL (2020) Methods and synthesis parameters affecting the formation of FAU type zeolite membrane and its separation performance: a review. J Asian Ceram Soc 8:553–571. https://doi.org/10.1080/21870764.2020.1769816
Wang J, Li M, Fu Y et al (2021) An ambient pressure method for synthesizing NaY zeolite. Microporous Mesoporous Mater 320:111073. https://doi.org/10.1016/j.micromeso.2021.111073
Kudinova AA, Poltoratckaya ME, Gabdulkhakov RR et al (2022) Parameters influence establishment of the petroleum coke genesis on the structure and properties of a highly porous carbon material obtained by activation of KOH. J Porous Mater 29:1599–1616. https://doi.org/10.1007/s10934-022-01287-1
Rozhkovskaya A, Rajapakse J, Millar GJ (2021) Synthesis of high-quality zeolite LTA from alum sludge generated in drinking water treatment plants. J Environ Chem Eng 9:104751. https://doi.org/10.1016/j.jece.2020.104751
Elboughdiri N (2020) The use of natural zeolite to remove heavy metals Cu (II), Pb (II) and Cd (II), from industrial wastewater. Cogent Eng 7. https://doi.org/10.1080/23311916.2020.1782623
Murukutti MK, Jena H (2022) Synthesis of nano-crystalline zeolite-A and zeolite-X from Indian coal fly ash, its characterization and performance evaluation for the removal of Cs+ and Sr2+ from simulated nuclear waste. J Hazard Mater 423:127085. https://doi.org/10.1016/j.jhazmat.2021.127085
Koohsaryan E, Anbia M, Maghsoodlu M (2020) Application of zeolites as non-phosphate detergent builders: a review. J Environ Chem Eng 8:104287. https://doi.org/10.1016/j.jece.2020.104287
Yusriadi Y, Ridayanti D, Sulastri E, Aanisah N (2022) The effect of tablet detergent wastewater using zeolite from rice husk ash on the chemical water quality and the growth of water hyacinth. Tenside Surfactants Deterg 59:433–440. https://doi.org/10.1515/tsd-2022-2424
Mordor Intelligence (2024) Sodium Silicate Market Size & Share Analysis - Growth Trends & Forecasts (2024 - 2029). https://www.mordorintelligence.com/industry-reports/sodium-silicate-market. Accessed 23.05.2024
Mordor Intelligence (2024) Zeolite Market Size & Share Analysis - Growth Trends & Forecasts (2024 - 2029). https://www.mordorintelligence.com/industry-reports/zeolites-market. Accessed 23.05.2024
Pyagay IN, Sizyakov VM, Svakhina YA et al (2023) Study of the process of obtaining water glass from silica gel for use in metallurgy. iPolytech J 27:598–610. https://doi.org/10.21285/1814-3520-2023-3-598-610
Mirfendereski M, Mohammadi T (2016) Effects of synthesis parameters on the characteristics of naa type zeolite nanoparticles. World Congr Recent Adv Nanotechnol 1–8. https://doi.org/10.11159/icnnfc16.113
Aminullah RE, Yuliarto B, Irzaman (2018) Reduction of silicon dioxide from bamboo leaves and its analysis using energy dispersive x-ray and fourier transform-infrared. IOP Conf Ser Earth Environ Sci 209:012048. https://doi.org/10.1088/1755-1315/209/1/012048
Lee Y, DuMont JW, George SM (2015) Atomic layer etching of AlF3 using sequential, self-limiting thermal reactions with Sn(acac)2 and hydrogen fluoride. J Phys Chem C 119:25385–25393. https://doi.org/10.1021/acs.jpcc.5b07236
Merkulova M, Boudon V, Manceron L (2023) Analysis of high-resolution spectra of SiF4 combination bands. J Mol Spectrosc 391:111738. https://doi.org/10.1016/j.jms.2023.111738
Kononov P, Kononova I, Moshnikov V et al (2022) Step-by-step modeling and demetallation experimental study on the porous structure in zeolites. Molecules 27:8156. https://doi.org/10.3390/molecules27238156
Collins F, Rozhkovskaya A, Outram JG, Millar GJ (2020) A critical review of waste resources, synthesis, and applications for Zeolite LTA. Microporous Mesoporous Mater 291:109667. https://doi.org/10.1016/j.micromeso.2019.109667
Dali Youcef L, López-Galindo A, Verdugo-Escamilla C, Belaroui LS (2020) Synthesis and characterization of zeolite LTA by hydrothermal transformation of a natural Algerian palygorskite. Appl Clay Sci 193:105690. https://doi.org/10.1016/j.clay.2020.105690
Rozhkovskaya A, Rajapakse J, Millar GJ (2021) Process engineering approach to conversion of alum sludge and waste glass into zeolite LTA for water softening. J Water Process Eng 43:102177. https://doi.org/10.1016/j.jwpe.2021.102177
Osacky M, Palkova H, Hudec P et al (2020) Effect of alkaline synthesis conditions on mineralogy, chemistry and surface properties of phillipsite, P and X zeolitic materials prepared from fine powdered perlite by-product. Microporous Mesoporous Mater 294:109852. https://doi.org/10.1016/j.micromeso.2019.109852
Al-Jubouri SM, Sabbar HA, Laft HA, Waisi BI (2019) Effect of synthesis parameters on the formation 4A zeolite crystals: characterization analysis and heavy metals uptake performance study for water treatment. Desalin Water Treat 165:290–300. https://doi.org/10.5004/dwt.2019.24566
Kong X, Qiu H, Zhang Y et al (2021) Seeded synthesis of all-silica CHA zeolites in diluted mother liquor. Microporous Mesoporous Mater 316:110914. https://doi.org/10.1016/j.micromeso.2021.110914
Ueno K, Yamada S, Negishi H et al (2020) Fabrication of pure-silica *BEA-type zeolite membranes on tubular silica supports coated with dilute synthesis gel via steam-assisted conversion. Sep Purif Technol 247:116934. https://doi.org/10.1016/j.seppur.2020.116934
Ayele L, Pérez-Pariente J, Chebude Y, Diaz I (2016) Synthesis of zeolite A using kaolin from Ethiopia and its application in detergents. New J Chem 40:3440–3446. https://doi.org/10.1039/C5NJ03097H
Xue Z, Li Z, Ma J et al (2014) Effective removal of Mg2+ and Ca2+ ions by mesoporous LTA zeolite. Desalination 341:10–18. https://doi.org/10.1016/j.desal.2014.02.025
Acknowledgements
The investigation was conducted at the Scientific Center “Issues of Mineral and Technogenic Resources Processing” with the involvement of the laboratory base of the Center for Collective Use of Empress Catherine II Saint Petersburg Mining University.
Funding
The authors state that they received no financial support for the research and preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. Material preparation,data collection and formal analysis were carried out by M.E., V.V. and V.R.. Data curation and validation was done by I.N., Ya.A. and M.E. Ya.A. and M.E. wrote the main text of the manuscript, I.N. edited and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The authors confirm that this manuscript is original, has not been previously published in whole or in part, and is not currently under consideration for publication in another journal.
Consent to Participate and Publication
The authors consent to participation and publication of the manuscript.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pyagay, I.N., Svakhina, Y.A., Titova, M.E. et al. Effect of Hydrogel Molar Composition on the Synthesis of LTA-type Zeolites in the Utilization of Technogenic Silica Gel. Silicon 16, 4811–4819 (2024). https://doi.org/10.1007/s12633-024-03053-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-024-03053-1