Abstract
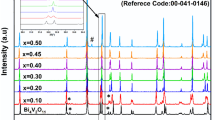
The solid-state reaction method was employed to successfully synthesize a range of compounds, Bi4Six/2Gex/2V2-xO11-x/2, where 0.1 ≤ x ≤ 0.6. The main aim of this investigation is to examine how the structural and optical properties are influenced by the double substitution on vanadium site. The analyses using room temperature X-ray diffraction (XRD) revealed existence of α-monoclinic and β-orthorhombic phases in the composition range of 0.1 ≤ x ≤ 0.3 and 0.4 ≤ x ≤ 0.5, respectively. The α to β structural transition, as a function of the substitution rate, was further confirmed through Raman and infrared spectroscopies at room temperature, which both verified the presence of these two distinct phases depending on the composition. The X-ray diffraction (XRD) analysis, versus temperature, highlighted the α → β and β → γ phase transitions with increasing the temperature. These phase transitions were confirmed by DTA and Raman investigations as a function of temperature. Diffuse reflectance spectroscopy showed that the band gap varies around 2.1eV and that the compound with x = 0.1 has the lowest value of 2.08 eV.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Data Availability
No datasets were generated or analysed during the current study.
References
Aurivillius B (1949) Mixed bismuth oxides with layer lattices. Arki Kemi 2
Kikuchi T, Watanabe A, Uchida K (1977) A family of mixed-layer type bismuth compounds. Mater Res Bull 12:299–304. https://doi.org/10.1016/0025-5408(77)90148-9
Ahsaine HA (2017) Etude de tungstate de bismuth (Bi, Lu)2WO6 : evolution des propriétés physicochimiques, luminescentes et photocatalytiques. Universite Ibn Zohr
Abraham F, Debreuille-Gresse MF, Mairesse G, Nowogrocki G (1988) Phase transitions and ionic conductivity in Bi4V2O11 an oxide with a layered structure. Solid State Ionics 28–30:529–532. https://doi.org/10.1016/S0167-2738(88)80096-1
Touboul M, Vachon C (1988) The Bi2O3-V2O5 system and crystal data about some bismuth vanadates. Thermochim Acta 133:61–66. https://doi.org/10.1016/0040-6031(88)87137-5
Mairesse G, Roussel P, Vannier RN et al (2003) Crystal structure determination of α-, β- and γ-Bi4V2O11 polymorphs. Part II: crystal structure of α-Bi4V2O11. Solid State Sci 5:861–869. https://doi.org/10.1016/S1293-2558(03)00016-5
Paydar MH, Hadian AM, Fafilek G (2001) Studies on preparation, characterisation and ion conductivity of TI-CU double substituted Bi4V2O11. J Eur Ceram Soc 21:1821–1824. https://doi.org/10.1016/S0955-2219(01)00123-6
Roy M, Sahu S, Barbar SK, Jangid S (2013) Synthesis, electrical and thermal properties of Bi4V2−xMexO11 (x = 0.0 and 0.02) ceramics. J Therm Anal Calorim 113:873–879. https://doi.org/10.1007/s10973-012-2833-1
Abrahams I (2003) A model for the mechanism of low temperature ionic conduction in divalent-substituted γ-BIMEVOXes. Solid State Ionics 157:139–145. https://doi.org/10.1016/S0167-2738(02)00201-1
Abrahams I, Bush AJ, Krok F et al (1998) Effects of preparation parameters on oxygen stoichiometry in Bi4V2O11−δ. J Mater Chem 8:1213–1217. https://doi.org/10.1039/a801614c
Boivin J (1998) Electrode-electrolyte BIMEVOX system for moderate temperature oxygen separation. Solid State Ionics 113–115:639–651. https://doi.org/10.1016/S0167-2738(98)00330-0
Löfberg A, Bodet H, Pirovano C et al (2006) Catalytic dense membranes of doped Bi4V2O11 (BIMEVOX) for selective partial oxidation: chemistry of defects versus catalysis. Top Catal 38:169–179. https://doi.org/10.1007/s11244-006-0082-x
Kant R, Singh K, Pandey O (2008) Synthesis and characterization of bismuth vanadate electrolyte material with aluminium doping for SOFC application. Int J Hydrogen Energy 33:455–462. https://doi.org/10.1016/j.ijhydene.2007.07.025
Karoum R, Pirovano C, Vannier RN et al (2009) Low temperature electrochemical catalysts using a BITAVOX electrolyte. Catal Today 146:359–366. https://doi.org/10.1016/j.cattod.2009.05.024
Chmielowiec J, Paściak G, BujŁo P (2009) BIMEVOX materials for application in SOFCS. Mater Sci Pol 27:1251–1256
Agnaou A, Mhaira W, Essalim R et al (2023) Correction: new silicon substituted BiMeVO x : synthesis and study of structural properties in relation to ionic conductivity. RSC Adv 13:8943–8943. https://doi.org/10.1039/D3RA90021E
Agnaou A, Mhaira W, Essalim R et al (2023) Structural study and ionic conductivity of Bi4V2−xSix/2Px/2O11−δ (0.0 ≤ x ≤ 0.5) compounds. J Solid State Chem 318:123730. https://doi.org/10.1016/j.jssc.2022.123730
Yue Y, Dzięgielewska A, Hull S et al (2022) Local structure in a tetravalent-substituent BIMEVOX system: BIGEVOX. J Mater Chem A 10:3793–3807. https://doi.org/10.1039/D1TA07547K
Anwar K, Naqvi FK, Beg S, Haneef S (2023) Photocatalytic degradation of MB dye and paracetamol drug, via hydrothermally synthesised praseodymium doped Bi4V2O11 nanoparticles. J Mol Struct 1272:134183. https://doi.org/10.1016/j.molstruc.2022.134183
Agnaou A, Mhaira W, Essalim R et al (2023) Effect of the doping element on the structure and UV–visible properties in the system Bi4V1.7(Si, Me)0.3O11-δ (Me = Si, P, Cu, and Co). Ionics (Kiel) 29:4923–4932. https://doi.org/10.1007/s11581-023-05185-7
Mhaira W, Agnaou A, Essalim R et al (2023) High-conducting Bi4V1.8Cu0.2-xSbxO10.7+3x/2 ceramics: structural, microstructural, electrical and optical properties. Ceram Int 49:39205–39213. https://doi.org/10.1016/j.ceramint.2023.09.264
Mhaira W, Agnaou A, Essalim R et al (2023) Structural, electrical, optical, and microstructural properties of Bi4V2-xCux/2Sbx/2O11–3x/4. J Solid State Electrochem 0–11. https://doi.org/10.1007/s10008-023-05768-4
Kharitonova EP, Voronkova VI (2007) Synthesis and electrical properties of Bi2V1−xGexO5+y solid solutions. Inorg Mater 43:55–59. https://doi.org/10.1134/S0020168507010128
Alga M, Ammar A, Essalim R et al (2005) Synthesis, sintering and electrical properties of P-doped Bi4V2O11 ceramics. Solid State Sci 7:1173–1179. https://doi.org/10.1016/j.solidstatesciences.2005.06.011
Shannon RD (1976) Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr Sect A 32:751–767. https://doi.org/10.1107/S0567739476001551
Agnaou A, Mhaira W, Essalim R et al (2023) Influence of Si4+ and Ga3+ doped in the BiSiGaVOx system on the structure and ionic conductivity. Silicon 1–8. https://doi.org/10.1007/s12633-023-02759-y
Ismunandar, Rusli R, Ramdan H (2011) Temperature dependent study of Bi2V1-xGaxO5.5 (x = 0.1 and 0.15) oxides. In: 2011 2nd International Conference on Instrumentation, Communications, Information Technology, and Biomedical Engineering. IEEE, pp 338–341
Rojas SS, De Souza JE, Andreeta MRB, Hernandes AC (2010) Influence of ceria addition on thermal properties and local structure of bismuth germanate glasses. J Non Cryst Solids 356:2942–2946. https://doi.org/10.1016/j.jnoncrysol.2010.05.101
Dimitrov V, Dimitriev Y, Montenero A (1994) IR spectra and structure of V2O5-GeO2-Bi2O3 glasses. J Non Cryst Solids 180:51–57. https://doi.org/10.1016/0022-3093(94)90396-4
Kumar B, Sharma R, Bhoi H et al (2023) Synthesis, structural, morphological and optical properties of environment friendly yellow inorganic pigment Bi4Zr3O12. Opt Mater (Amst) 142:114040. https://doi.org/10.1016/j.optmat.2023.114040
Essalim R, Ammar A, Zamama M, Mauvy F (2020) A study on structural properties, conductivity and FT-IR spectroscopy of Cu–Al doubly substituted Bi4V2O11. J Solid State Chem 288:121405. https://doi.org/10.1016/j.jssc.2020.121405
ElBatal FH, Marzouk MA, Abdelghany AM (2011) UV–visible and infrared absorption spectra of gamma irradiated V2O5-doped in sodium phosphate, lead phosphate, zinc phosphate glasses: a comparative study. J Non Cryst Solids 357:1027–1036. https://doi.org/10.1016/j.jnoncrysol.2010.11.040
Mhaira W, Agnaou A, Essalim R et al (2023) Effect of simultaneous Cu and Nb doping Bi4V2O11 on structural and electrical properties of Bi4V2−xCux/2Nbx/2O11−3x/4. J Solid State Chem 320:123878. https://doi.org/10.1016/j.jssc.2023.123878
Russu S (2008) High-throughput synthesis and characterization of vanadium mixed metal oxide pigments using synchroton radiation
Trzciński K, Borowska-Centkowska A, Sawczak M, Lisowska-Oleksiak A (2015) Photoelectrochemical properties of BIMEVOX (ME=Cu, Zn, Mn) electrodes in contact with aqueous electrolyte. Solid State Ionics 271:63–68. https://doi.org/10.1016/j.ssi.2014.10.008
Hardcastle FD, Wachs IE, Eckert H, Jefferson DA (1991) Vanadium(V) environments in bismuth vanadates: a structural investigation using Raman spectroscopy and solid state 51V NMR. J Solid State Chem 90:194–210. https://doi.org/10.1016/0022-4596(91)90135-5
Hardcastle F, Wachs I (1991) Determination of niobium-oxygen bond distances and bond orders by Raman spectroscopy. Solid State Ionics 45:201–213. https://doi.org/10.1016/0167-2738(91)90153-3
Wójcik NA, Polcyn K, Karczewski J et al (2022) High-conducting Bi4V2−xFexO11-δ ceramics containing Fe2O3 nanocrystals: structure and properties. J Eur Ceram Soc 42:5015–5022. https://doi.org/10.1016/j.jeurceramsoc.2022.05.012
Tripathy D, Saikia A, Tado GT, Pandey A (2019) Role of Al and Ti doping in modulating electrical properties of BIVOX system. J Adv Ceram 8:489–499. https://doi.org/10.1007/s40145-019-0329-1
Acknowledgements
The authors would like to express their gratitude to the Cadi Ayyad University Analysis and Characterization Center (CAC) for providing access to the materials characterization techniques used in this study.
Funding
None.
Author information
Authors and Affiliations
Contributions
A. AGNAOU: investigation, writing original draft, formal analysis; W. MHAIRA: helped the interpretation of results; R. ESSALIM: writing review and editing; M. ZAGHRIOUI: writing review and editing; T. CHARTIER: investigation; C. AUTRET: investigation; A. AMMAR: conceived the idea and supervision.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Agnaou, A., Mhaira, W., Essalim, R. et al. Structural and Optical Properties of Bi4Six/2Gex/2V2-xO11-x/2 Compounds: Effect of Double Substitution and Phase Transitions. Silicon 16, 4635–4645 (2024). https://doi.org/10.1007/s12633-024-03035-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-024-03035-3