Abstract
Background
Bronchial hyperresponsiveness (BHR) is a fundamental pathophysiological characteristic of asthma. Although several factors such as airway caliber can affect BHR, no study has established age-dependent cutoff values of BHR to methacholine for the diagnosis of asthma in children. We investigated the cutoff values of the methacholine challenge test (MCT) in the diagnosis of asthma according to age.
Methods
A total of 2383 individuals aged from 6 to 15 years old were included in this study. MCTs using the five-breath technique were performed in 350 children with suspected asthma based on symptoms by pediatric allergists and in 2033 healthy children from a general population-based cohort. We determined the provocative concentration of methacholine producing a 20% decrease in forced expiratory volume in 1 second from baseline (PC20). A modified Korean version of the International Study of Asthma and Allergies in Childhood questionnaire was used to distinguish asthmatics and healthy subjects. Receiver-operator characteristic curve analysis was used to assess the cutoff value of PC20 for the diagnosis of asthma.
Results
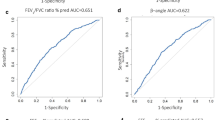
Cutoff values of methacholine PC20, which provided the best combination of diagnostic sensitivity and specificity, showed an increasing pattern with age: 5.8, 9.1, 11.8, 12.6, 14.9, 21.7, 23.3, 21.1, 21.1, and 24.6 mg/mL at ages 6, 7, 8, 9, 10, 11, 12, 13, 14, and 15 years, respectively.
Conclusion
The application of different cutoff values of methacholine PC20 depending on age might be a practical modification for the diagnosis of asthma in children and adolescents with asthmatic symptoms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
van Aalderen WM. Childhood asthma: diagnosis and treatment. Scientifica (Cairo) 2012;2012:674204.
Warrier PJ, Bellamkonda P, Stokes JR, Casale TB. Utility and limitations of objective measures of asthma. Ann Allergy Asthma Immunol 2009;102:518–522.
Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, et al. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med 2000;161:309–329.
Trigg CJ, Bennett JB, Tooley M, Sibbald B, D’Souza MF, Davies RJ. A general practice based survey of bronchial hyperresponsiveness and its relation to symptoms, sex, age, atopy, and smoking. Thorax 1990;45:866–872.
O’Connor G, Sparrow D, Taylor D, Segal M, Weiss S. Analysis of dose-response curves to methacholine. An approach suitable for population studies. Am Rev Respir Dis 1987;136:1412–1417.
Cerveri I, Bruschi C, Zoia MC, Zanon P, Maccarini L, Grassi M, et al. Distribution of bronchial nonspecific reactivity in the general population. Chest 1988;93:26–30.
Perpina M, Pellicer C, de Diego A, Compte L, Macian V. Diagnostic value of the bronchial provocation test with methacholine in asthma. A Bayesian analysis approach. Chest 1993;104:149–154.
Jayet PY, Schindler C, Kunzli N, Zellweger JP, Brandli O, Perruchoud AP, et al. Reference values for methacholine reactivity (SAPALDIA study). Respir Res 2005;6:131.
Kim BJ, Seo JH, Jung YH, Kim HY, Kwon JW, Kim HB, et al. Air pollution interacts with past episodes of bronchiolitis in the development of asthma. Allergy 2013;68:517–523.
Kim WK, Kwon JW, Seo JH, Kim HY, Yu J, Kim BJ, et al. Interaction between IL13 genotype and environmental factors in the risk for allergic rhinitis in Korean children. J Allergy Clin Immunol 2012;130:421–426.e5.
Hewitt DJ. Interpretation of the "positive" methacholine challenge. Am J Ind Med 2008;51:769–781.
Avital A, Noviski N, Bar-Yishay E, Springer C, Levy M, Godfrey S. Nonspecific bronchial reactivity in asthmatic children depends on severity but not on age. Am Rev Respir Dis 1991;144:36–38.
Tantisira KG, Colvin R, Tonascia J, Strunk RC, Weiss ST, Fuhlbrigge AL, et al. Airway responsiveness in mild to moderate childhood asthma: sex influences on the natural history. Am J Respir Crit Care Med 2008;178:325–331.
Mead J. Dysanapsis in normal lungs assessed by the relationship between maximal flow, static recoil, and vital capacity. Am Rev Respir Dis 1980;121:339–342.
Hibbert M, Lannigan A, Raven J, Landau L, Phelan P. Gender differences in lung growth. Pediatr Pulmonol 1995;19:129–134.
Parker AL, Abu-Hijleh M, McCool FD. Ratio between forced expiratory flow between 25% and 75% of vital capacity and FVC is a determinant of airway reactivity and sensitivity to methacholine. Chest 2003;124:63–69.
Tan KS, McFarlane LC, Lipworth BJ. Loss of normal cyclical beta 2 adrenoceptor regulation and increased premenstrual responsiveness to adenosine monophosphate in stable female asthmatic patients. Thorax 1997;52:608–611.
Lieberman D, Kopernic G, Porath A, Levitas E, Lazer S, Heimer D. Influence of estrogen replacement therapy on airway reactivity. Respiration 1995;62:205–208.
Tan KS, McFarlane LC, Lipworth BJ. Paradoxical downregulation and desensitization of beta2-adrenoceptors by exogenous progesterone in female asthmatics. Chest 1997;111:847–851.
Cockcroft DW, Davis BE, Todd DC, Smycniuk AJ. Methacholine challenge: comparison of two methods. Chest 2005;127:839–844.
Cockcroft DW, Davis BE. The bronchoprotective effect of inhaling methacholine by using total lung capacity inspirations has a marked influence on the interpretation of the test result. J Allergy Clin Immunol 2006;117:1244–1248.
Salome CM, Xuan W, Gray EJ, Belooussova E, Peat JK. Perception of airway narrowing in a general population sample. Eur Respir J 1997;10:1052–1058.
Hopp RJ, Townley RG, Biven RE, Bewtra AK, Nair NM. The presence of airway reactivity before the development of asthma. Am Rev Respir Dis 1990;141:2–8.
Jansen DF, Timens W, Kraan J, Rijcken B, Postma DS. (A) symptomatic bronchial hyper-responsiveness and asthma. Respir Med 1997;91:121–134.
Zhong NS, Chen RC, Yang MO, Wu ZY, Zheng JP, Li YF. Is asymptomatic bronchial hyperresponsiveness an indication of potential asthma? A two-year follow-up of young students with bronchial hyperresponsiveness. Chest 1992;102:1104–1109.
Laprise C, Boulet LP. Asymptomatic airway hyperresponsiveness: a three-year follow-up. Am J Respir Crit Care Med 1997;156:403–409.
Pin I, Radford S, Kolendowicz R, Jennings B, Denburg JA, Hargreave FE, et al. Airway inflammation in symptomatic and asymptomatic children with methacholine hyperresponsiveness. Eur Respir J 1993;6:1249–1256.
Brand PL. Airway inflammation in symptomatic and asymptomatic children with methacholine hyperresponsiveness. Eur Respir J 1994;7:829.
de Gooijer A, Brand PL, Gerritsen J, Koeter GH, Postma DS, Knol K. Changes in respiratory symptoms and airway hyperresponsiveness after 27 years in a population-based sample of school children. Eur Respir J 1993;6:848–854.
Henderson JC, O’Connell F, Fuller RW. Decrease of histamine induced bronchoconstriction by caffeine in mild asthma. Thorax 1993;48:824–826.
Cheung D, Dick EC, Timmers MC, de Klerk EP, Spaan WJ, Sterk PJ. Rhinovirus inhalation causes long-lasting excessive airway narrowing in response to methacholine in asthmatic subjects in vivo. Am J Respir Crit Care Med 1995;152:1490–1496.
Clough JB, Hutchinson SA, Williams JD, Holgate ST. Airway response to exercise and methacholine in children with respiratory symptoms. Arch Dis Child 1991;66:579–583.
Salo PM, Arbes SJ Jr, Jaramillo R, Calatroni A, Weir CH, Sever ML, et al. Prevalence of allergic sensitization in the United States: results from the National Health and Nutrition Examination Survey (NHANES) 2005-2006. J Allergy Clin Immunol 2014;134:350–359.
Bodtger U. Prognostic value of asymptomatic skin sensitization to aeroallergens. Curr Opin Allergy Clin Immunol 2004;4:5–10.
Godfrey S, Springer C, Noviski N, Maayan C, Avital A. Exercise but not methacholine differentiates asthma from chronic lung disease in children. Thorax 1991;46:488–492.
Avital A, Springer C, Bar-Yishay E, Godfrey S. Adenosine, methacholine, and exercise challenges in children with asthma or paediatric chronic obstructive pulmonary disease. Thorax 1995;50:511–516.
Gilbert R, Auchincloss JH Jr. Post-test probability of asthma following methacholine challenge. Chest 1990;97:562–565.
Motomura C, Odajima H, Tezuka J, Murakami Y, Moriyasu Y, Kando N, et al. Effect of age on relationship between exhaled nitric oxide and airway hyperresponsiveness in asthmatic children. Chest 2009;136:519–525.
Anderson SD. Provocative challenges to help diagnose and monitor asthma: exercise, methacholine, adenosine, and mannitol. Curr Opin Pulm Med 2008;14:39–45.
Zou KH, O’Malley AJ, Mauri L. Receiver-operating characteristic analysis for evaluating diagnostic tests and predictive models. Circulation 2007;115:654–657.
Fruchter O, Yigla M. Seasonal variability of the methacholine challenge test. J Asthma 2009;46:951–954.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding: This research was supported by a fund (2014-ER5602-00) from Research of Korea Centers for Disease Control and Prevention.
Ethical approval: This study was performed according to the guideline of Asan Medical Center, which abides by the Helsinki Declaration on ethical principles for medical research.
Competing interest: The authors declare that they have no competing interests.
Contributors: Hong SJ (Email: sjhong@amc.seoul.kr) and Kwon JW contributed equally to this work as co-corresponding authors. Hong SJ and Kwon JW designed and supervised the execution of the study. Lee E and Han S performed the analyses. Lee E wrote the manuscript. Kim YH, Yang SI, Jung YH, Seo JH, Kim HB, and Lee SY participated in the interpretations.
Rights and permissions
About this article
Cite this article
Lee, E., Kim, YH., Han, S. et al. Different cutoff values of methacholine bronchial provocation test depending on age in children with asthma. World J Pediatr 13, 439–445 (2017). https://doi.org/10.1007/s12519-017-0026-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-017-0026-5