Abstract
Background
Idiopathic nephrotic syndrome is the most common glomerular disease in children. This study was undertaken to observe the efficacy and side-effects of rituximab (RTX) in treating children with different types of refractory primary nephrotic syndrome.
Methods
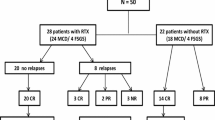
Twelve patients with steroid dependent nephrotic syndrome (SDNS), frequently relapsing nephritic syndrome (FRNS), and steroid resistant nephrotic syndrome (SRNS) were enrolled in our study. There were obvious drug side-effects, and proteinuria remained difficult to control. RTX was administered at a dose of 375 mg/m2 body surface area, once or twice weekly.
Results
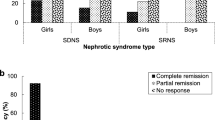
The male to female ratio was 3:1, and the onset age was 1.6–8.9 years. There were 9 patients with steroid sensitive nephrotic syndrome (SDNS or FRNS), and 3 patients with SRNS. There were 7 patients with minimal change disease (MCD), 3 patients with focal segmental glomerular sclerosis (FSGS), 1 with focal proliferative glomerulonephritis, and 1 without renal biopsy. The total effective treatment rate of RTX was 91.67%, and for 77.78% of the patients, steroid dosage could be reduced. Six months before and after RTX infusion, the mean steroid dosage was significantly decreased (P=0.014) and the recurrence number was significantly reduced (P<0.001). The results were better in MCD patients than in FSGS patients (P=0.045). There was no significant difference between FRNS/SDNS and SRNS patients (P=0.175). During RTX administration, 3 patients developed skin rashes, 1 developed hypotension, and 1 developed a fever. One patient experienced a persistent decrease in serum immunoglobulin level but without serious infection.
Conclusion
RTX was effective in the treatment of refractory nephrotic syndrome, and it could significantly reduce the use of steroid and immunosuppressants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
van Husen M, Kemper MJ. New therapies in steroid-sensitive and steroid-resistant idiopathic nephrotic syndrome. Pediatr Nephrol 2011;26:881–892.
Kyrieleis HA, Levtchenko EN, Wetzels JF. Long-term outcome after cyclophosphamide treatment in children with steroiddependent and frequently relapsing minimal change nephrotic syndrome. Am J Kidney Dis 2007;49:592–597.
Grimbert P, Audard V, Remy P, Lang P, Sahali D. Recent approaches to the pathogenesis of minimal-change nephrotic syndrome. Nephrol Dial Transplant 2003;18:245–248.
Benz K, Dötsch J, Rascher W, Stachel D. Change of the course of steroid-dependent nephrotic syndrome after rituximab therapy. Pediatr Nephrol 2004;19:794–797.
Gilbert RD, Hulse E, Rigden S. Rituximab therapy for steroiddependent minimal change nephrotic syndrome. Pediatr Nephrol 2006;21:1698–1700.
Pescovitz MD, Book BK, Sidner RA. Resolution of recurrent focal segmental glomerulosclerosis proteinuria after rituximab treatment. N Engl J Med 2006;354:1961–1963.
Smith GC. Is there a role for rituximab in the treatment of idiopathic childhood nephrotic syndrome? Pediatr Nephrol 2007;22:893–898.
Bagga A, Sinha A, Moudgil A. Rituximab in patients with the steroid-resistant nephrotic syndrome. N Engl J Med 2007;356:2751–2752.
Guigonis V, Dallocchio A, Baudouin V, Dehennault M, Hachon-Le Camus C, Afanetti M, et al. Rituximab treatment for severe steroid- or cyclosporine-dependent nephrotic syndrome: a multicentric series of 22 cases. Pediatr Nephrol 2008;23:1269–1279.
Kamei K, Ito S, Nozu K, Fujinaga S, Nakayama M, Sako M, et al. Single dose of rituximab for refractory steroid-dependent nephrotic syndrome in children. Pediatr Nephrol 2009;24:1321–1328.
Ito S, Kamei K, Ogura M, Sato M, Fujimaru T, Ishikawa T, et al. Maintenance therapy with mycophenolate mofetil after rituximab in pediatric patients with steroid-dependent nephrotic syndrome. Pediatr Nephrol 2011;26:1823–1828.
Liu GL, Mao S, Xia ZK, Fan ZM, Ren XG, He X, et al. Rituximab for steroid-dependentnephrotic syndrome in children. Yi Xue Yan Jiu Sheng Xue Bao 2011;24:482–484. [In Chinese]
Guan N, Ding J. Anti-CD20 monoclonal antibody in treatment of refractory nephrotic syndrome. Er Ke Yao Xue Za Zhi 2010;16:1–3. [In Chinese]
The Subspecialty Group of Nephrology, Society of Pediatrics, Chinese Medical Association. Evidence-based guidelines on diagnosis and treatment of childhood common renal disease (I): Guideline on diagnosis and treatment of steroid-sensitive, relapsing/steroid-dependent nephrotic syndrome. Zhonghua Er Ke Za Zhi 2009;47:167–170. [In Chinese]
The Subspecialty Group of Nephrology, Society of Pediatrics, Chinese Medical Association. Evidence-based guidelines on diagnosis and treatment of childhood common renal diseases (III): Guideline on diagnosis and treatment of steroid-resistant nephrotic syndrome. Zhonghua Er Ke Za Zhi 2010;48:72–75.
Gulati A, Sinha A, Jordan SC, Hari P, Dinda AK, Sharma S, et al. Efficacy and safety of treatment with rituximab for difficult steroid-resistant and -dependent nephrotic syndrome: multicentric report. Clin J Am Soc Nephrol 2010;5:2207–2212.
Prytuła A, Iijima K, Kamei K, Geary D, Gottlich E, Majeed A, et al. Rituximab in refractory nephrotic syndrome. Pediatr Nephrol 2010;25:461–468.
Nakayama M, Kamei K, Nozu K, Matsuoka K, Nakagawa A, Sako M, et al. Rituximab for refractory focal segmental glomerulosclerosis. Pediatr Nephrol 2008;23:481–485.
Kari JA, El-Morshedy SM, El-Desoky S, Alshaya HO, Rahim KA, Edrees BM. Rituximab for refractory cases of childhood nephrotic syndrome. Pediatr Nephrol 2011;26:733–737.
Sellier-Leclerc AL, Macher MA, Loirat C, Guérin V, Watier H, Peuchmaur M, et al. Rituximab efficiency in children with steroid-dependent nephrotic syndrome. Pediatr Nephrol 2010;25:1109–1115.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, L., Xu, H., Shen, Q. et al. Efficacy of rituximab therapy in children with refractory nephrotic syndrome: a prospective observational study in Shanghai. World J Pediatr 10, 59–63 (2014). https://doi.org/10.1007/s12519-014-0453-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-014-0453-5