Abstract
Purpose of Review
Coronary bifurcation lesions require complex interventions and are associated with higher complication rates. Compared to angiography alone, intravascular imaging with intravascular ultrasound (IVUS) and optical coherence tomography (OCT) allows better planning and optimization of bifurcation lesion interventions leading to improved results and long-term success. In this review, we aim to analyze the current data on utilization of intravascular imaging for optimization of interventions in complex bifurcation lesions and its impact on clinical outcomes.
Recent Findings
Evidence from meta-analyses and registries favor IVUS-guided percutaneous intervention (PCI) over angiography alone due to lower rates of cardiac death, myocardial infarction, and major adverse cardiac events. Current expert consensus supports the use of OCT to evaluate plaque characteristics and vessel anatomy. Both IVUS and OCT facilitate intervention planning by identifying high-risk lesions based on plaque characteristics and functional significance, improving stent sizing through more accurately measured vessel diameter and lesion length, and optimizing stent deployment by detecting stent underexpansion and malapposition.
Summary
Intravascular imaging in bifurcation lesions enhances planning, executing, and optimizing interventions, thereby contributing to improved clinical outcomes. Use of both IVUS and OCT should be considered in cases with complex anatomy such as bifurcations. With the advancement in technology such as high-resolution IVUS and 3D reconstruction for OCT, these modalities will likely have an increased role in bifurcation lesion interventions. Larger randomized studies, however, are needed to further define the role of intravascular imaging in bifurcation intervention.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary bifurcation lesions account for approximately 20% of all percutaneous coronary interventions (PCI). Despite significant improvements in interventional techniques and the advent of drug-eluting stents (DES), bifurcation lesions remain a challenge with lower procedural success and higher long-term complication rates, with some studies reporting a 6-month success rate of ~ 77% and restenosis rate of ~ 26% [1, 2]. Selecting the appropriate interventional technique for bifurcation lesions can be difficult due to anatomic complexity. Use of intravascular imaging techniques such as intravascular ultrasound (IVUS) or optical coherence tomography (OCT) during PCI has been shown to improve clinical outcomes for bifurcation lesions by providing more detailed information regarding coronary vessel anatomy, lesion characteristics, and stent sizing prior to deployment [3, 4•, 5,6,7,8,9].
In contrast to angiography alone, IVUS and OCT imaging provides greater detail of plaque morphology, stent expansion, and apposition, and the presence of coronary dissections, all of which are factors that may lead to complications following PCI. Accordingly, several meta-analyses have shown reduced rate of stent thrombosis (ST), myocardial infarction (MI), death, and major adverse cardiac events (MACE) when PCI with DES implantation was guided by IVUS or OCT [6,7,8,9].
This article will review current data regarding use of intravascular imaging techniques to optimize PCI in coronary bifurcation lesions.
Bifurcation Lesion Definition, Classification, and Intervention
A coronary artery bifurcation lesion, as defined by the European Bifurcation Club, is a narrowing adjacent to or involving the origin of a significant side branch, the loss of which would be consequential (symptoms, territory of ischemia, viability of the supplied myocardium, collateralizing vessel, left ventricular function, etc.) [10].
There are two strategies used in interventions of a bifurcation lesion: a one-stent approach (the provisional technique) and two-stent approaches. In the provisional technique, the main vessel (MV) is stented with subsequent side branch (SB) treatment only if the SB becomes compromised during MV intervention. With two-stent techniques, most notably the culotte and the double-kissing (DK) crush techniques, the aim is to intentionally stent both the MV and SB as part of the intervention [11•].
Several randomized control trials and meta-analyses found no difference in outcomes when comparing provisional and two-stent techniques [12•, 13•, 14•]. However, more recent meta-analysis shows decreased MI, MACE, and death in the provisional strategy compared to two-stent techniques [15]. Guidance with IVUS or OCT improves outcomes not only in PCI of non-complex lesions, but may also have a role in determining the need for planning and executing more complex bifurcation techniques [6,7,8,9, 16••].
Intravascular Ultrasound
Benefits of IVUS use in bifurcation lesions include greater anatomic detail of vessel lumen and lesion anatomy that facilitate intervention planning, optimization of stent positioning, and detection of post-PCI complications, all of which ultimately improve outcomes through decreased target lesion revascularization (TLR).
Growing evidence from registries and meta-analyses favor IVUS-guided PCI [17]. A recent study by Chen et al. demonstrated a lower rate of cardiac death, MI, revascularization, and MACE at 7-year follow-up in patients who had IVUS-guided bifurcation stenting compared to angiography-guided bifurcation stenting. Similarly, a Korean retrospective study reported reduction in overall mortality and rate of very late stent thrombosis in IVUS-guided DES bifurcation intervention [16••, 17••, 18••].
Intravascular ultrasound guidance should be considered in all bifurcation lesions, especially in cases where angiography may be ambiguous. IVUS can rule out pseudostenosis in the SB caused by coronary spasm or calcification and allow for selection of appropriate stenting method, i.e., one- vs two-stent technique. It also provides important information about lesion length and demarcation of healthy tissue, both of which are important in selecting the appropriate stent length and preventing edge dissection. In lesions that are initially treated with one stent technique, IVUS can identify the need for follow-up SB intervention. Post PCI, intravascular imaging aids the detection of stent underexpansion, malapposition, and dissection which can be subsequently corrected to prevent future complications. In all cases of stent failure, IVUS is strongly recommended to identify and correct the underlying factors [17, 19].
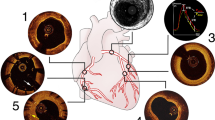
In clinical practice, IVUS is a useful adjunctive tool when treating bifurcation lesions. Both Figs. 1 and 2 demonstrate the use of IVUS in planning and confirmation of stent sizing and placement.
Intravascular ultrasound images of pre- and post-intervention in a left main bifurcation lesion using the culotte two-stent technique. (A) Angiogram showing lesion involving the distal left main (LM), ostial left circumflex (LCx), and ostial left anterior descending artery (LAD). (B) Post-intervention angiogram showing an optimum result. (a) Pre-intervention IVUS image of LCx artery; yellow line indicates the luminal diameter. (b), (c) IVUS images showing disease in the LAD and LCx segments. (d) IVUS image of bifurcation confluence. (e) Pre-intervention IVUS image of the LM artery; yellow line indicates the luminal diameter. (f) IVUS image of LM lumen. (g–l) Series of post-intervention IVUS images showing well-apposed stent placed in the LAD, LCx, and LM using the culotte two-stent technique
Intravascular ultrasound images of bifurcation intervention using the DK crush two-stent technique with pre-intervention measurements to aid in stent sizing. (A, B) IVUS images showing the measurement of lumen area (a) and media-adventitia border (b). (C) IVUS image of well-apposed stent in the side branch. (D) IVUS image of well-apposed main vessel stent. (E) IVUS image of stent at the bifurcation level. (F) IVUS image of stent in the left main lumen
Optical Coherence Tomography
Optical coherence tomography uses near-infrared light to create images. The current technique uses a high viscosity liquid such as contrast to purge blood from the coronary artery and acquire images with spatial resolution of nearly 10 μm, which is higher than the typical 40-μm resolution of IVUS [20••, 21••].
ILUMIEN 3 is a randomized control trial that showed OCT as non-inferior to IVUS in non-complex lesions [22]. Current consensus of expert opinion agrees that use of OCT to assess plaque characteristics as well as pre- and post-PCI anatomy confers clinical benefit in complex cases such as bifurcations [23, 24••]. Figure 3 demonstrates the use of OCT imaging in the assessment of stent optimization during a bifurcation lesion intervention.
Optical coherence tomography images showing intervention in a bifurcation lesion using a Tryton bifurcation stent. a Distal area of bifurcation showing the LAD and LCx vessels with coronary wires and stent struts. b, c OCT images of proximal bifurcation post-intervention. d–f OCT images of the left main lumen with good stent apposition
Novel applications for OCT have been proposed using post-imaging processing. Three-dimensional (3D) real-time reconstruction of OCT images provide greater anatomic detail that may assist complex maneuvers, such as positioning the wire through appropriate stent cells, and reduce stent malapposition [25]. One prospective, non-randomized, observational study of 150 bifurcation lesions investigated the use of 3D OCT imaging during PCI to optimize guidewire positioning through the jailed SB prior to kissing balloon inflation [26]. Compared with conventional 2D OCT imaging, 3D OCT imaging has a lower rate of incomplete stent apposition in left main lesions (8.9% vs 18.7%, p 0.014) [26]. Use of 3D OCT imaging in other bifurcation lesions trended toward lower rate of incomplete stent apposition but was not statistically significant; no benefit was seen in composite cardiovascular death, MI, ST, TVR, and in-stent restenosis [26]. Figure 4 shows a complex bifurcation lesion where OCT use helped plan and optimize strategy during intervention.
Post-intervention OCT images showing well-placed stent in a bifurcation lesion involving the left main using the culotte two-stent technique. (A) Angiogram showing pre-intervention bifurcation lesion. (B) Angiogram showing wide open vessels following bifurcation intervention. (a) OCT image of LAD showing well-apposed stent. (b) OCT image of LCx showing well-placed stent struts. (c, d) OCT images of proximal bifurcation into the left main lumen
Intervention Planning
Pre-PCI
Utilizing intravascular imaging techniques prior to PCI helps identify culprit lesions by evaluating plaque morphology and the functional significance of intermediate lesions, provide more accurate anatomic measurements to optimize stent sizing, evaluate SB anatomy to identify lesions that may require a two-stent approach, and aid the use of adjunctive therapies such as atherectomy. Recent high-quality data on the use of IVUS in PCI to optimize stent implantation in bifurcation lesions remains conflicted and has not clarified the utility of IVUS in PCI. This is reflected in the current European guidelines that recommend IVUS in selected patients as a Class IIa recommendation [23]. In contrast, studies of OCT use in bifurcation lesions are limited to observational data. However, consensus recommendation from both the European and Japanese Bifurcation Clubs supports the use of OCT in bifurcation lesions [24••].
Plaque Morphology
Intravascular imaging evaluates plaque morphology in greater detail that may identify at-risk or culprit lesions. Thin cap fibroatheromas (TCFA) are the type of plaque at highest risk for rupture and subsequent acute coronary syndrome (ACS) [25]. Plaque erosions are a less frequent cause of ACS and occur when the endothelium covering the plaque is lost, exposing the underlying smooth muscle layer [26]. IVUS and OCT can measure thickness of the fibrous cap to identify TCFA, absence of fibrous cap rupture to identify plaque erosion, and presence of thrombus [27, 28]. Higher resolution OCT images afford greater detail and closer measurement of the fibrous cap thickness when compared with IVUS and angiography [29].
IVUS can be used to assess calcified lesions for the degree of stenosis and extent of calcification to evaluate the need for atherectomy and subsequent stent sizing. When compared with angiography-guided atherectomy alone, IVUS guidance produces increased post-PCI MLD and a trend toward decreased MACE and TVR [30].
Intermediate Lesion Assessment
IVUS and OCT can evaluate functional significance of intermediate lesions, particularly in the left main (LM). IVUS is superior to angiography alone in evaluating the LM. In contrast, OCT is limited in the proximal LM due to difficulty displacing intravascular blood through contrast injection, although OCT images near the distal LM bifurcation are adequate for use in PCI [31, 32]. IVUS measurements of minimum luminal area (MLA) less than 5.9 mm2 and minimum luminal diameter (MLD) less than 2.8 mm correlate with functionally significant lesions as measured by FFR and strongly predict significance of left main lesions [33]. OCT measurements are less predictive of functionally significant lesions. However, ischemia depends on additional factors such as lesion length, myocardial territory, and collateral supply which potentially limits intravascular imaging assessment of functional ischemia [17, 24••].
Vessel Sizing
IVUS and OCT can provide more accurate measurements of vessel size to optimize stent sizing. Appropriately sizing the stent is paramount to intervention planning since stent dimensions predict restenosis rates [4•, 34, 35]. IVUS provides more precise measurement of vessel diameter than angiography resulting in larger minimum stent area (MSA) and lower TLR [4•, 36]. In contrast, OCT can underestimate the true vessel diameter due to incomplete visualization of the vessel walls in lipid-rich lesions, resulting in smaller stent deployment [37]. However, new techniques have been developed to overcome this shortcoming. Ali et al. measured external elastic lamina diameter proximal and distal to the lesions and chose a stent size based on the smaller of the measurements [38••]. This resulted in MSA that was non-inferior to IVUS or angiography [38••].
Defining Bifurcation Geometry and Side Branch Details
Intravascular imaging imparts greater anatomic detail of the complex 3D bifurcation structure that informs the choice of approach. Acute SB occlusion during PCI using the provisional strategy is a serious complication unique to bifurcation interventions. The ability to predict acute SB occlusion could direct interventions for at-risk lesions from one-stent strategies to more complex two-stent strategies. Measurement of the plaque thickness and SB diameter using IVUS have been shown to independently predict SB occlusion [39]. Similarly, measurement of the proximal MV maximal lipid arc and SB diameter using OCT has been shown to independently predict SB occlusion [40].
In summary, the use of IVUS and OCT prior to bifurcation PCI provides crucial information to guide the decision to intervene, optimize stent size, determine the technical approach, and assist in adjunctive therapies.
PCI Procedure
Wire re-crossing through the jailed SB at an appropriate point near the carina for kissing balloon inflation is a key step during bifurcation intervention which, when done correctly, prevents stent struts from being pushed inappropriately into the MV. Intravascular imaging during the procedure can help direct control of wire re-crossing through the jailed side branch. IVUS can repeatedly track the SB wire position between the proximal MV and SB and further guide this step [17]. The potential benefit of OCT guidance in selecting the re-crossing point has been assessed in some studies; however, the evidence is mainly limited to observational data. Alegria-Barrero et al. in their study of 52 patients undergoing bifurcation lesion treatment re-crossed 12 cases with OCT guidance and noted significantly lower number of malapposed stent struts when compared to angiography guidance alone [41]. Reports from a prospective registry with 3D OCT acquisition show an 89.9% feasibility of assessment of guidewire re-crossing following MV stenting. Although several randomized trials are ongoing to further assess the issue, current joint European and Japanese bifurcation clubs statements recommend that 3D OCT be used after MV stenting to determine re-crossing position before the final kissing balloon [24••].
Post-PCI
Stent underexpansion predisposes to stent thrombosis and in-stent restenosis [42, 43] Despite ample post-dilatation and good angiographic result, stents may still be underexpanded. Small MSA of less than 5–5.5 mm2 is generally consistent with stent underexpansion and is an independent predictor of restenosis. In LM lesions, this value for optimal results is reported as greater than 8.7 mm2. In a study by Kang et al. looking at IVUS-MSA values to predict ISR, various segments had different cut-offs. In the LM, it was 8.2 mm2, for ostial left circumflex 5.0 mm2, and for ostial left anterior descending 6.3 mm2 [44]. With OCT, stent expansion is the minimum stent cross-sectional area which can be an absolute measure or relative to a predefined reference area. In general, greater absolute stent expansion (obtained as an absolute measure) is related to lower risk of stent failure [24••]. Using published intravascular imaging criteria to detect stent underexpansion can help prevent future stent complications.
Malapposition refers to incomplete stent strut contact with the vessel wall, which can be an independent finding or co-exist with underexpansion. Although conflicting evidence exists regarding the role of acute malapposition in stent thrombosis, multiple studies of stent thrombosis have identified malapposition as an underlying stent abnormality. Malapposition is better identified by OCT as compared to IVUS (50% vs 15% respectively). The risk of malapposition is higher in bifurcation PCI due to the requirement of rewiring freshly implanted stents, which may result in accidental abluminal rewiring leading to malapposition. Stent apposition distance of < 300 μm acutely is likely to resolve; however, if the distance is > 300 μm, additional post-dilatation should be considered [17, 24••].
Tissue prolapse, defined as tissue extrusion from inside the stent area, may include lesion or athero-thrombotic material protrusion, the latter being the case in the context of ACS. This phenomenon, seen on imaging following stent placement, has been related to early stent thrombosis. IVUS is generally less superior in identifying tissue prolapse compared to OCT, and ACS-related tissue prolapse is more likely to have clinical consequences compared to non-ACS-related tissue prolapse [45••].
IVUS identified stent dissections are not always detected by angiography. In a study by Lui et al., IVUS identified post stent dissections after 9.2% of DES, of which 39% were not identified by angiography [46]. Chamie et al. showed high rates of stent edge dissections post PCI with OCT guidance, usually due to the presence of atherosclerosis at stent edges and to PCI technique [47]. Smaller edge dissections found on OCT that are undetected by angiography likely do not have clinical impact. Some factors to consider when determining the significance of edge dissection include longitudinal (> 3 mm) and circumferential (> 60°) extension, respective intra-dissection lumen area (< 90% of reference), and the depth of dissection [38••]. In the CLI-OPCI II trial with OCT, dissection was defined as > 0.2 mm wide linear tissue rim clearly separated from the vessel wall. Significant IVUS criteria for edge dissection associated with acute stent thrombosis include lumen narrowing to < 4.0 mm2 or dissection angle > 60°. Additional stent implantation should be considered in these situations to reduce the risk of stent thrombosis [17, 24••].
Limitations of Imaging
Although the value of intravascular imaging for PCI guidance is widely appreciated, there are certain potential limitations to keep in mind. Additional time required for imaging, notable cost of IVUS and OCT and need for adequate training in acquisition and interpretation of images are some caveats. The potential additional time requirement can be overcome by early anticipation and advance preparation in planned interventions. For IVUS-guided PCI with DES, dedicated analysis has shown cost-effectiveness, especially when used in patients at higher risk of PCI complications. Operator training is part of most interventional fellowships which provide a fair degree of experience to new trainees. Another potential limitation is catheter deliverability in some complex lesion subsets such as tortuous, heavily calcified, and angulated coronaries. Imaging-related complications are infrequent and mostly self-limiting with no major adverse events. Advancement in technology with more deliverable catheters, ability to co-register with angiography, and higher resolution imaging will further the use of intravascular imaging [45••, 48].
Conclusion
Intravascular imaging in bifurcation lesions has great application in planning, executing, and optimizing intervention, thereby contributing to improved clinical outcomes. Use of both IVUS and OCT should be considered in complex anatomy and complex cases. With the advancement in technology such as high-resolution IVUS and 3D reconstruction for OCT, these modalities will likely have an increased role in bifurcation lesion intervention.
Abbreviations
- ACS:
-
Acute coronary syndrome
- DES:
-
Drug eluting stent
- DK:
-
Double kissing
- FFR:
-
Fractional flow reserve
- LM:
-
Left main
- IVUS:
-
Intravascular ultrasound
- MACE:
-
Major adverse cardiac events
- MI:
-
Myocardial infarction
- MLA:
-
Minimum luminal area
- MLD:
-
Minimum luminal diameter
- MSA:
-
Minimum stent area
- MV:
-
Main vessel
- OCT:
-
Optical coherence tomography
- PCI:
-
Percutaneous coronary intervention
- SB:
-
Side branch
- ST:
-
Stent thrombosis
- TCFA:
-
Thin cap fibroatheroma
- TLR:
-
Target lesion revascularization
- TVR:
-
Target vessel revascularization
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Sawaya F, Lefevre T, Chevalier B, Garot P, Hovasse T, Morice M-C, et al. Contemporary approach to coronary bifurcation lesion treatment. JACC Cardiovasc Interv. 2016;9(18):1861–78.
Colombo A, Moses J, Morice MC, Ludwig J, Holmes D, Spanos V, et al. Randomized study to evaluate sirolimus-eluting stents implanted at coronary bifurcation lesions. Circulation. 2004;109:1244–9.
Chieffo A, Latib A, Caussin C, Presbitero P, Galli S, Menozzi A, et al. A prospective, randomized trial of intravascular-ultrasound guided compared to angiography guided stent implantation in complex coronary lesions: the AVIO trial. Am Heart J. 2013;165:65–72.
• Hong SJ, Kim BK, Shin DH, Nam CM, Kim JS, Ko YG, et al. Effect of intravascular ultrasound-guided vs angiography-guided everolimus-eluting stent implantation: the IVUS-XPL randomized clinical trial. JAMA. 2015;314:2155–63 This study compares outcomes of DES implantation with angiography alone vs IVUS guidance. In patients requiring longer stents, there was lower composite incidence of MACE and cardiac death at 1 yr (2.9% vs 5.8%, hazard ratio [HR] 0.48 [95% CI, 0.28 to 0.83], P = .007). This favors the use of IVUS in complex lesions.
Patel Y, Depta JP, Novak E, Yeung M, Lavine K, Banerjee S, et al. Long-term outcomes with use of intravascular ultrasound for the treatment of coronary bifurcation lesions. Am J Cardiol. 2012;109:960–5.
Klersy C, Ferlini M, Raisaro A, Scotti V, Balduini A, Curti M, et al. Use of IVUS guided coronary stenting with drug eluting stent: a systematic review and meta-analysis of randomized controlled clinical trials and high quality observational studies. Int J Cardiol. 2013;170:54–63.
Jang J-S, Song Y-J, Kang W, Jin H-Y, Seo J-S, Yang T-H, et al. Intravascular ultrasound-guided implantation of drug-eluting stents to improve outcome. JACC Cardiovasc Interv. 2014;7(3):233–43.
Ahn J-M, Kang S-J, Yoon S-H, Park HW, Kang SM, Lee J-Y, et al. Meta-analysis of outcomes after intravascular ultrasound-guided versus angiography-guided drug-eluting stent implantation in 26,503 patients enrolled in three randomized trials and 14 observational studies. Am J Cardiol. 2014;113:1338–47.
Alsidawi S, Effat M, Rahman S, Abdallah M, Leesar M. The role of vascular imaging in guiding routine percutaneous coronary interventions: a meta-analysis of bare metal stent and drug-eluting stent trials. Cardiovasc Ther. 2015;33:360–6.
Louvard Y, Thomas M, Dzavik V, Hildick-Smith D, Galassi A, Pan M, et al. Classification of coronary artery bifurcation lesions and treatments: time for a consensus! Catheter Cardiovasc Interv. 2008;71:175–83.
• Rab T, Sheiban I, Louvard Y, Sawaya FJ, Zhang JJ, Chen SL. Current interventions for the left main bifurcation. JACC Cardiovasc Interv. 2017;10:849–65. This paper reviews the LM bifurcation intervention with detailed account of various techniques and use of intravascular imaging. It highlights the safety of LM bifurcation intervention with second generation DES in comparison to CABG, however with continued concern for late TLR. This emphasizes the safe use of LM bifurcation PCI in patients who may not be able to or opt not to undergo CABG.
• Hildick-Smith D, Behan M, Lassen J, Chieffo A, Lefevre T, Stankovic G et al. The EBC TWO Study (European Bifurcation Coronary TWO) A randomized comparison of provisional T-stenting versus a systematic 2 stent culotte strategy in large caliber true bifurcations. Circ Cardiovasc Interv. 2016;9. This RCT is a comparison of proximal T-stent strategy with an upfront two stent Cullote strategy for large caliber bifurcation lesions with significant side branch disease. No difference was found in composite end point of death, MI or TLR in both groups. This shows the efficacy of either technique when planning an intervention.
Colombo A, Bramucci E, Sacca S, Violini R, Lettieri C, Zanini R, et al. Randomized study of the crush technique versus provisional side-branch stenting in true coronary bifurcations the CACTUS (coronary bifurcations: application of the crushing technique using sirolimus-eluting stents) study. Circulation. 2009;119:71–8.
Katritsis D, Siontis G, Ioannidis J. Double versus single stenting for coronary bifurcation lesions. Circ Cardiovasc Interv. 2009;2009(2):409–15.
Nairooz R, Saad M, Elgendy I, Mahmoud A, Habash F, Sardar P, et al. Long-term outcomes of provisional stenting compared with a two-stent strategy for bifurcation lesions: a meta-analysis of randomised trials. Heart. 2017;2017(103):1427–34.
•• Chen L, Xu T, Xue XJ, et al. Intravascular ultrasound-guided drug-eluting stent implantation is associated with improved clinical outcomes in patients with unstable angina and complex coronary artery true bifurcation lesions. Int J Cardiovasc Imaging. 2018;11:1685–96 This study compares IVUS guided bifurcation intervention to angiography guided bifurcation intervention. Results show lower adverse event rate in IVUS guided vs angiography group at 7 yr follow up (MACE 15.2% vs 22.4%, p=0.01; Cardiac death 1.3% vs 6.5%,p=0.002; MI 2.3% vs 8.4%, p=<0.001). This study favors routine use of IVUS for bifurcation intervention.
Legutko J, Yamawaki M, Costa RA, et al. IVUS in bifurcation stenting: what have we learned? EuroIntervention. 2015;11(Suppl V):V55–8.
Kim SH, Kim YH, Kang SJ, Park DW, Lee SW, Lee CW, et al. Long-term outcomes of intravascular ultrasound-guided stenting in coronary bifurcation lesions. Am J Cardiol. 2010;106:612–8.
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. EuroIntervention. 2019;14:1435–534.
•• Koganti S, Kotecha T, Rakhit RD. Choice of intracoronary imaging: when to use intravascular ultrasound or optical coherence tomography. Interv Cardiol. 2016;11:11–6 This review elaborates the use of IVUS and OCT in various coronary intervention suggesting IVUS use in left main and OCT use in smaller vessels based on available MLA data. Also emphasizes the safety of intravascular imaging which in future may be routinely incorporated in interventions.
•• Shrestha R, Shrestha A, Kan J, Chen S. A review in enormity of OCT and its enduring understanding of vulnerable plaque in coronary bifurcation lesion. Int J Cardiovasc Imaging. 2018;34:1679–84 This review summarizes current role of OCT in assessment of site and composition of vulnerable plaque at coronary bifurcation lesions to aide with clinical decision and planning of PCI.
Maehara A, Ben-Yehuda O, Ali Z, Wijns W, Bezerra H, Shite J, et al. Comparison of stent expansion guided by optical coherence tomography versus intravascular ultrasound. JACC Cardiovasc Imaging. 2015;8(13):1704–14.
Kolh P, Windecker S, Alfonso F, Collet J-P, Crener J, Falk V, et al. 2014 ESC/EACTS guidelines on myocardial revascularization The Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur J Cardiothorac Surg. 2014;46:517–92.
•• Onuma Y, Katagiri Y, Burzotta F, et al. Joint consensus on the use of OCT in coronary bifurcation lesions by the European and Japanese bifurcation clubs. EuroIntervention. 2019;14:e1568–77 This consensus document from the European bifurcation club and Japanese bifurcation club support the feasibility of use of OCT in guiding complex procedures in bifurcations, however encourage the need for further work in obtaining dedicated evidence for OCT use for bifurcation treatment.
Virmani R, Kolodgie FD, Burke AP, et al. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol. 2000;20:162–75.
Falk E, Nakano M, Bentzon JF, Finn AV, Virmani R. Update on acute coronary syndromes: the pathologists’ view. Eur Heart J. 2013;34:719–28.
Inaba S, Mintz GS, Burke AP, Stone GW, Virmani R, Matsumura M, et al. Intravascular ultrasound and near-infrared spectroscopic characterization of thin-cap fibroatheroma. Am J Cardiol. 2017;119:372–8.
Ino Y, Kubo T, Tanaka A, Kuroi A, Tsujioka H, Ikejima H, et al. Difference of culprit lesion morphologies between ST-segment elevation myocardial infarction and non-ST-segment elevation acute coronary syndrome: an optical coherence tomography study. JACC Cardiovasc Interv. 2011;4:76–82.
Kubo T, Imanishi T, Takarada S, Kuroi A, Ueno S, Yamano T, et al. Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J Am Coll Cardiol. 2007;50:933–9.
Shlofmitz E, Martinsen B, Lee M, Généreux P, Behrens A, Kumar G, et al. Utilizing intravascular ultrasound imaging prior to treatment of severely calcified coronary lesions with orbital atherectomy: an ORBIT II sub-analysis. J Interv Cardiol. 2017;30:570–6.
Oviedo C, Maehara A, Mintz G, Araki H, Choi S-Y, Tsujita K, et al. Intravascular ultrasound classification of plaque distribution in left main coronary artery bifurcations. Circulation Cardiovasc Interv. 2010;3:105–12.
Burzotta F, Dato I, Trani C, Pirozzolo G, Maria GLD, Porto I, et al. Frequency domain optical coherence tomography to assess non-ostial left main coronary artery. Eurointervention. 2015;10:1–8.
Jasti V, Ivan E, Yalamanchili V, Wongpraparut N, Leesar MA. Correlatons between fractional flow reserve and intravascular ultrasound in patients with an ambiguous left main coronary artery stenosis. Circulation. 2004;110:2831–6.
De Feyter PJ, Kay P, Disco C, et al. Reference chart derived from post-stent-implantation intravascular ultrasound predictors of 6-month expected restenosis on quantitative coronary angiography. Circulation. 1999;100:1777–83.
Sonoda S, Morino Y, Ako J, Terashima M, Hassan AHM, Bonneau HN, et al. Impact of final stent dimensions on long-term results following sirolimus-eluting stent implantation: serial intravascular ultrasound analysis from the Sirius trial. J Am Coll Cardiol. 2004;43:1959–63.
Costa RA, Mintz GS, Carlier SG, Lansky AJ, Moussa I, Fujii K, et al. Bifurcation coronary lesions treated with the “crush” technique: an intravascular ultrasound analysis. J Am Coll Cardiol. 2005;46:599–605.
Wijns W, Shite J, Jones MR, Lee SWL, Price MJ, Fabbiocchi F, et al. Optical coherence tomography imaging during percutaneous coronary intervention impacts physician decision-making: ILUMIEN I study. Eur Heart J. 2015;36:3346–55.
•• Ali ZA, Maehara A, Genereux P, et al. Optical coherence tomography compared with intravascular ultrasound and with angiography to guide coronary stent implantation (ILUMIEN III: OPTIMIZE PCI): a randomized controlled trial. Lancet. 2016;388:2618–28 This RCT compares OCT guided PCI and IVUS guided PCI with angiography alone. OCT shows non-inferiority to IVUS guidance, however is not superior to angiography alone. This however warrants further large RCTs comparing OCT use to angiography alone.
Sakamoto N, Hoshino Y, Mizukami H, Sugimoto K, Yamaki T, Kunii H, et al. Intravascular ultrasound predictors of acute side branch occlusion in coronary artery bifurcation lesions just after single stent crossover. Catheter Cardiovasc Interv. 2016;87:243–50.
Kini A, Yoshimura T, Vengrenyuk Y, Amirian J, Hasan C, Baber U, et al. Plaque morphology predictors of side branch occlusion after main vessel stenting in coronary bifurcation lesions. JACC Cardiovasc Interv. 2016;9(8):861–8.
Alegria-Barrero E, Foin N, Chan PH, et al. Optical coherence tomography for guidance of distal cell recrossing in bifurcation stenting: choosing the right cell matters. EuroIntervention. 2012;8:205–13.
Cook S, Wenaweser P, Togni M, et al. Incomplete stent apposition and very late stent thrombosis after drug-eluting stent implantation. Circulation. 2007;115:2426–34.
Sarno G, Garg S, Gomez-Lara J, Garcia Garcia HM, Ligthart J, Bruining N, et al. Intravascular ultrasound radiofrequency analysis after optimal coronary stenting with initial quantitative coronary angiography guidance: an ATHEROREMO sub-study. EuroIntervention. 2011;6:977–84.
Kang SJ, Ahn JM, Song H, Kim WJ, Lee JY, Park DW, et al. Comprehensive intravascular ultrasound assessment of stent area and its impact on restenosis and adverse cardiac events in 403 patients with unprotected left main disease. Circulation Cardiovascular Interv. 2011;4:562–9.
•• Raber L, Mintz GS, Koskinas KC, et al. Clinical use of intracoronary imaging. Part 1: guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. Eur Heart J. 2018;39:3281–300 An expert consensus document highlighting the use of IVUS or OCT for stent optimization and sizing and planning procedural strategy. It gives an outline of the strengths and limitations of intravascular imaging for guiding PCI and assessing stent failure with emphases on further research.
Lui X, Tsujita K, Maehara A, et al. Intravascular ultrasound assessment of the incidence and predictors of edge dissections after drug-eluting stent implantation. JACC Cardiovasc Interv. 2009;2:997–1004.
Chamie D, Bezerra HG, Attizzani GF, et al. Incidence, predictors, morphological characteristics, and clinical outcomes of stent edge dissections detected by optical coherence tomography. JACC Cardiovasc Interv. 2013;6:800–13.
Alberti A, Giudice P, Gelera A, Stefanini L, Priest V, Simmonds M, et al. Understanding the economic impact of intravascular ultrasound (IVUS). Eur J Health Econ. 2016;17:185–93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors have no conflict of interest to declare.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Intravascular Imaging
Rights and permissions
About this article
Cite this article
Darlington, A.M., Balouch, M.A. & Shroff, A.R. Optimization of Coronary Bifurcation Intervention Using Intravascular Imaging. Curr Cardiovasc Imaging Rep 12, 26 (2019). https://doi.org/10.1007/s12410-019-9504-6
Published:
DOI: https://doi.org/10.1007/s12410-019-9504-6