Abstract
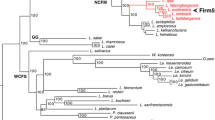
The Lactobacillus genus is widely used for fermentation of plant materials and dairy products. These species are typically found in highly specialized environments, with the bee gut serving as one of the niche locations in which Lactobacillus is detected. Lactobacillus species isolated from the bee gut and bee-related habitats were phylogenetically classified into three distinct groups, Lactobacillus kunkeei, Firm-4, and Firm-5. The L. kunkeei group was clearly differentiated from other members of the Lactobacillus buchneri group isolated from non-bee habitats. In comparison with non-bee members of the L. buchneri group, three bee-symbiotic Lactobacillus groups had a small-sized genome with low G + C content and showed a sharp reduction in the number of genes involved in energy production, carbohydrate transport and metabolism, and amino acid transport and metabolism. In addition, all three groups lacked the mutY gene, which encodes A/G-specific adenine glycosylase. The phylogenetic dendrogram based on the presence or absence of 1,199 functional genes indicated that these bee-symbiotic groups experienced convergent evolution. The occurrence of convergent evolution is thought to stem from the three bee-symbiotic groups sharing a similar habitat, i.e., the bee gut. The causative factor underlying genomic reduction was postulated to be mutY, which was absent in all three groups. Here, a novel strain, BHWM-4T, isolated from the gut of Bombus ignites was studied using polyphasic taxonomy and classified as a new member of the L. kunkeei group. The strain was Gram-positive, facultative anaerobic, and rod-shaped. The 16S ribosomal RNA gene sequence and genome analysis revealed that strain BHWM-4T was clustered into the L. kunkeei group, forming a compact cluster with L. kunkeei and Lactobacillus apinorum. Biochemical, chemotaxonomic, and genotypic data of strain BHWM-4T supports the proposal of a novel species, Lactobacillus bombintestini sp. nov., whose type strain is BHWM-4T (= KACC 19317 = NBRC 113067T).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahn, J.H., Hong, I.P., Bok, J.I., Kim, B.Y., Song, J., and Weon, H.Y. 2012. Pyrosequencing analysis of the bacterial communities in the guts of honey bees Apis cerana and Apis mellifera in Korea. J. Microbiol.50, 735–745.
Aravind, L., Walker, D.R., and Koonin, E.V. 1999. Conserved domains in DNA repair proteins and evolution of repair systems. Nucleic Acids Res.27, 1223–1242.
Auch, A.F., von Jan, M., Klenk, H.P., and Göker, M. 2010. Digital DNA-DNA hybridization for microbial species delineation by means of genome-to-genome sequence comparison. Stand. Genomic Sci.2, 117–134.
Canchaya, C., Claesson, M.J., Fitzgerald, G.F., van Sinderen, D., and O’Toole, P.W. 2006. Diversity of the genus Lactobacillus revealed by comparative genomics of five species. Microbiology152, 3185–3196.
Chin, C.S., Alexander, D.H., Marks, P., Klammer, A.A., Drake, J., Heiner, C., Clum, A., Copeland, A., Huddleston, J., Eichler, E.E., et al. 2013. Nonhybrid, finished microbial genome assemblies from long-read SMRT sequencing data. Nat. Methods10, 563–569
Chiou, T.Y., Suda, W., Oshima, K., Hattori, M., Matsuzaki, C., Yamamoto, K., and Takahashi, T. 2018. Lactobacillus kosoi sp. nov., a fructophilic species isolated from koso, a Japanese sugar-vegetable fermented beverage. Antonie van Leeuwenhoek111, 1149–1156.
Chun, J., Oren, A., Ventosa, A., Christensen, H., Arahal, D.R., da Costa, M.S., Rooney, A.P., Yi, H., Xu, X.W., De Meyer, S., et al. 2018. Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int. J. Syst. Evol. Microbiol.68, 461–466.
De Man, J.C., Rogosa, D., and Sharpe, M.E. 1960. A medium for the cultivation of lactobacilli. J. Appl. Microbiol.23, 130–135.
Dufresne, A., Garczarek, L., and Partensky, F. 2005. Accelerated evolution associated with genome reduction in a free-living prokaryote. Genome Biol.6, R14.
Edwards, C.G., Haag, K.M., Collins, M.D., Hutson, R.A., and Huang, Y.C. 1998. Lactobacillus kunkeei sp. nov.: A spoilage organism associated with grape juice fermentations. J. Appl. Microbiol.84, 698–702.
Ellegaard, K.M. and Engel, P. 2019. Genomic diversity landscape of the honey bee gut microbiota. Nat. Commun.10, 446.
Ellegaard, K.M., Tamarit, D., Javelind, E., Olofsson, T.C., Andersson, S.G.E., and Vasquez, A. 2015. Extensive intra-phylotype diversity in lactobacilli and bifidobacteria from the honeybee gut. BMC Genomics16, 284.
Endo, A. 2012. Fructophilic lactic acid bacteria inhabit fructose-rich niches in nature. Microb. Ecol. Heal. Dis.23, 18563.
Endo, A., Futagawa-Endo, Y., and Dicks, L.M.T. 2009. Isolation and characterization of fructophilic lactic acid bacteria from fructose-rich niches. Syst. Appl. Microbiol.32, 593–600.
Endo, A., Tanizawa, Y., Tanaka, N., Maeno, S., Kumar, H., Shiwa, Y., Okada, S., Yoshikawa, H., Dicks, L., Nakagawa, J., et al. 2015. Comparative genomics of Fructobacillus spp. and Leuconostoc spp. reveals niche-specific evolution of Fructobacillus spp. BMC Genomics16, 1117.
Falsen, E., Pascual, C., Sjödén, B., Ohlén, M., and Collins, M.D. 1999. Phenotypic and phylogenetic characterization of a novel Lactobacillus species from human sources: Description of Lactobacillus iners sp. nov. Int. J. Syst. Bacteriol.49, 217–221.
Felsenstein, J. 1981. Evolutionary trees from DNA sequences: A maximum likelihood approach. J. Mol. Evol.17, 368–376.
Felsenstein, J. 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution39, 783–791.
Felske, A., Rheims, H., Wolterink, A., Stackebrandt, E., and Akkermans, A.D.L. 1997. Ribosome analysis reveals prominent activity of an uncultured member of the class Actinobacteria in grassland soils. Microbiology143, 2983–2989.
Fitch, W.M.. Toward defining the course of evolution: Minimum change for a specific tree topology. Syst. Biol., 406–416.
Gerhardt, P., Murray, R.G.E., Wood, W.A., and Krieg, N.R. (eds.). 1994. Methods for General and Molecular Bacteriology. American Society for Microbiology, Washington, DC, USA.
Germond, J.E., Lapierre, L., Delley, M., Mollet, B., Felis, G.E., and Dellaglio, F. 2003. Evolution of the bacterial species Lactobacillus delbrueckii: A partial genomic study with reflections on prokaryotic species concept. Mol. Biol. Evol.20, 93–104.
Heo, J., Saitou, S., Tamura, T., Cho, H., Kim, J.S., Joa, J.H., Kim, J.S., Kwon, S.W., and Kim, S.J. 2018. Lactobacilus nuruki sp. nov., isolated from nuruk, a Korean fermentation starter. Int. J. Syst. Evol. Microbiol.68, 3273–3278.
Hutchinson, G.E. 1957. A treatise on limnology. 1. Geography, Physics, and Chemistry, New York, USA.
Hyatt, D., Chen, G.L., LoCascio, P.F., Land, M.L., Larimer, F.W., and Hauser, L.J. 2010. Prodigal: Prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics11, 119.
Kawasaki, S., Kurosawa, K., Miyazaki, M., Sakamoto, M., Ohkuma, M., and Niimura, Y. 2011. Lactobacillus ozensis sp. nov., isolated from mountain flowers. Int. J. Syst. Evol. Microbiol.61, 2435–2438.
Kikuchi, Y., Hosokawa, T., Nikoh, N., Meng, X.Y., Kamagata, Y., and Fukatsu, T. 2009. Host-symbiont co-speciation and reductive genome evolution in gut symbiotic bacteria of acanthosomatid stinkbugs. BMC Biol.7, 2.
Killer, J., Dubná, S., Sedláček, I., and Švec, P. 2014a. Lactobacillus apis sp. nov., from the stomach of honeybees, having an in vitro inhibitory effect on the causative agents of American and European foulbrood. Int. J. Syst. Evol. Microbiol.64, 152–157.
Killer, J., Votavová, A., Valterová, I., Vlková, E., Rada, V., and Hroncová, Z. 2014b. Lactobacillus bombi sp. nov., from the digestive tract of laboratory-reared bumblebee queens (Bombus terrestris). Int. J. Syst. Evol. Microbiol.64, 2611–2617.
Kumar, S., Stecher, G., and Tamura, K. 2016. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol.33, 1870–1874.
Kwong, W.K. and Moran, N.A. 2016. Gut microbial communities of social bees. Nat. Rev. Microbiol.14, 374–384.
Lee, I., Kim, Y.O., Park, S.C., and Chun, J. 2016. OrthoANI: an improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol.66, 1100–1103.
Lind, P.A. and Andersson, D.I. 2008. Whole-genome mutational biases in bacteria. Proc. Natl. Acad. Sci. USA105, 17878–17883.
Maeno, S., Dicks, L., Nakagawa, J., and Endo, A. 2017. Lactobacillus apinorum belongs to the fructophilic lactic acid bacteria. Biosci. Microbiota Food Heal.36, 147–149.
Maeno, S., Tanizawa, Y., Kanesaki, Y., Kanesaki, Y., Kubota, E., Kumar, H., Dicks, L., Salminen, S., Nakagawa, J., Arita, M., and Endo, A. 2016. Genomic characterization of a fructophilic bee symbiont Lactobacillus kunkeei reveals its niche-specific adaptation. Syst. Appl. Microbiol., 516–526.
Martinson, V.G., Danforth, B.N., Minckley, R.L., Rueppell, O., Tingek, S., and Moran, N.A. 2011. A simple and distinctive microbiota associated with honey bees and bumble bees. Mol. Ecol.20, 619–628.
McCutcheon, J.P. and Moran, N.A. 2012. Extreme genome reduction in symbiotic bacteria. Nat. Rev. Microbiol.10, 13–26.
McFrederick, Q.S., Vuong, H.Q., and Rothman, J.A. 2018. Lactobacillus micheneri sp. nov., Lactobacillus timberlakei sp. nov. and Lactobacillus quenuiae sp. nov., lactic acid bacteria isolated from wild bees and flowers. Int. J. Syst. Evol. Microbiol.68, 1879–1884.
Meier-Kolthoff, J.P., Auch, A.F., Klenk, H.P., and Goker, M. 2013. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics, 60.
Minnikin, D.E., O’Donnell, A.G., Goodfellow, M., Alderson, G., Athalye, M., Schaal, A., and Parlett, J.H. 1984. An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J. Microbiol. Methods2, 233–241.
Nikoh, N., Hosokawa, T., Oshima, K., Hattori, M., and Fukatsu, T. 2011. Reductive evolution of bacterial genome in insect gut environment. Genome Biol. Evol.3, 702–714.
Olofsson, T.C., Alsterfjord, M., Nilson, B., Butler, È., and Vásquez, A. 2014. Lactobacillus apinorum sp. nov., Lactobacillus mellifer sp. nov., Lactobacillus mellis sp. nov., Lactobacillus melliventris sp. nov., Lactobacillus kimbladii sp. nov., Lactobacillus helsingborgensis sp. nov. and Lactobacillus kullabergensis sp. nov., isolated from the honey stomach of the honeybee Apis mellifera. Int. J. Syst. Evol. Microbiol.64, 3109.3019.
Overbeek, R., Olson, R., Pusch, G.D., Olsen, G.J., Davis, J.J., Disz, T., Edwards, R.A., Gerdes, S., Parrello, B., Shukla, M., et al. 2014. The SEED and the Rapid Annotation of microbial genomes using Subsystems Technology (RAST). Nucleic Acids Res.42, 206–214.
Praet, J., Meeus, I., Cnockaert, M., Houf, K., Smagghe, G., and Van damme, P. 2015. Novel lactic acid bacteria isolated from the bumble bee gut: Convivina intestini gen. nov., sp. nov., Lactobacillus bombicola sp. nov., and Weissella bombi sp. nov. Antonie van Leeuwenhoek107, 1337–1349.
Pruesse, E., Peplies, J., and Glöckner, F.O. 2012. SINA: Accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics28, 1823–1829.
Saitou, N. and Nei, M. 1987. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol.4, 406–425.
Salvetti, E., Harris, H.M.B., Felis, G.E., and O’Toole, P.W. 2018. Comparative genomics of the genus Lactobacillus reveals robust phylogroups that provide the basis for reclassification. Appl. Environ. Microbiol.84, e00993–18.
Sasser, M. 1990. Identification of Bacteria by Gas Chromatography of Cellular Fatty Acids. Technical Note 101, Microbial MIDI Inc., Newark, DE, USA.
Siezen, R.J., Tzeneva, V.A., Castioni, A., Wels, M., Phan, H.T.K., Rademaker, J.L.W., Starrenburg, M.J.C., Kleerebezem, M., and van Hylckama Vlieg, J.E.T. 2010. Phenotypic and genomic diversity of Lactobacillus plantarum strains isolated from various environmental niches. Environ. Microbiol.12, 758–773.
Tamarit, D., Ellegaard, K.M., Wikander, J., Olofsson, T., Vásquez, A., and Andersson, S.G.E. 2015. Functionally structured genomes in Lactobacillus kunkeei colonizing the honey crop and food products of honeybees and stingless bees. Genome Biol. Evol.7, 1455–1473.
Tatusova, T., Dicuccio, M., Badretdin, A., Chetvernin, V., Nawrocki, E.P., Zaslavsky, L., Lomsadze, A., Pruitt, K.D., Borodovsky, M., and Ostell, J. 2016. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 44, 6614–6624
Toft, C. and Andersson, S.G.E. 2010. Evolutionary microbial genomics: insights into bacterial host adaptation. Nat. Rev. Genet.11, 465–475.
Vásquez, A., Forsgren, E., Fries, I., Paxton, R.J., Flaberg, E., Szekely, L., and Olofsson, T.C. 2012. Symbionts as major modulators of insect health: Lactic acid bacteria and honeybees. PLoS One7, e33188.
Vuong, H.Q. and McFrederick, Q.S. 2019. Comparative genomics of wild bee and flower isolated Lactobacillus reveals potential adaptation to the bee host. Genome Biol. Evol, 11, 2151–2161.
Wang, C., Huang, Y., Li, L., Guo, J., Wu, Z., Deng, Y., Dai, L., and Ma, S. 2018. Lactobacillus panisapium sp. nov., from honeybee Apis cerana bee bread. Int. J. Syst. Evol. Microbiol.68, 703–708
Yoon, S.H., Ha, S.M., Kwon, S., Lim, J., Kim, Y., Seo, H., and Chun, J. 2017. Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol.67, 1613–1617.
Acknowledgments
We thank professor Bernhard Schink and Aharon Oren for advice on the etymology of the new names proposed.
This study was carried out with the support (PJ013549) of National Institute of Agricultural Sciences, Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
12275_2020_9596_MOESM1_ESM.pdf
Comparative genomics of Lactobacillus species as bee symbionts and description of Lactobacillus bombintestini sp. nov., isolated from the gut of Bombus ignitus
Rights and permissions
About this article
Cite this article
Heo, J., Kim, SJ., Kim, JS. et al. Comparative genomics of Lactobacillus species as bee symbionts and description of Lactobacillus bombintestini sp. nov., isolated from the gut of Bombus ignitus. J Microbiol. 58, 445–455 (2020). https://doi.org/10.1007/s12275-020-9596-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-020-9596-3