Abstract
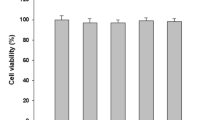
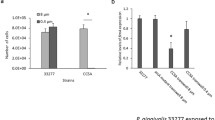
Streptococcus sanguinis is often found in subgingival biofilm including periodontopathogens, and is correlated with a delay in colonization by periodontopathogens. However, the effect of S. sanguinis on inflammation induced by periodontopathogens is poorly understood. Thus, this study investigated the effect of S. sanguinis peptidoglycan (PGN) on induction of TNF-α, IL-6, and IL-8 expression by lipopolysaccharide (LPS) of periodontal pathogens. LPS was extracted from Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, and Tannerella forsythia, and PGN was isolated from S. sanguinis. THP-1 cells, a monocytic cell-line, were cotreated with LPS of the periodontal pathogens and S. sanguinis PGN, and then the expression of inflammatory cytokines was analyzed by real-time RT-PCR. To analyze the underlying mechanism, the binding assay of the LPS to CD14 or LPS-binding protein (LBP) was performed in the presence or absence of the PGN after coating recombinant human CD14 and LBP on EIA plate. The PGN inhibited the binding of LPS to CD14 and LBP in a dose-dependent manner. Also, THP-1 cells were co-treated with the LPS in the presence of N-acetylmuramic acid and N-acetylglucosamine, as components of PGN, and the competition binding assay to CD14 and LBP was performed. N-acetylmuramic acid inhibited the induction of inflammatory cytokine expression by LPS and the binding of LPS to CD14 or LBP whereas N-acetylglucosamine did not show such effect. Collectively, the results suggest that S. sanguinis PGN inhibited the cytokine expression induced by the LPS of periodontopathogens due to the inhibition of LPS binding to LBP and CD14. N-acetylmuramic acid of PGN may play a role in inhibition of the LPS binding of periodontopathogens to CD14 and LBP.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akira, S. and Takeda, K. 2004. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511.
Chamaillard, M., Hashimoto, M., Horie, Y., Masumoto, J., Qiu, S., Saab, L., Ogura, Y., Kawasaki, A., Fukase, K., Kusumoto, S., et al. 2003. An essential role for nod1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat. Immunol. 4, 702–707.
Davis, K.M. and Weiser, J.N. 2011. Modifications to the peptidoglycan backbone help bacteria to establish infection. Infect. Immun. 79, 562–570.
Deng, H., Ding, Y., Fu, M.D., Xiao, X.R., Liu, J., and Zhou, T. 2004. purification and characterization of sanguicin–a bacteriocin produced by Streptococcus sanguis. Sichuan Da Xue Xue Bao Yi Xue Ban. 35, 555–558.
Dziarski, R. and Gupta, D. 2005. Peptidoglycan recognition in innate immunity. J. Endotoxin Res. 11, 304–310.
Hong, S.W., Baik, J.E., Kang, S.S., Yun, C.H., Seo, D.G., and Han, S.H. 2014. Lipoteichoic acid of Streptococcus mutans interacts with toll-like receptor 2 through the lipid moiety for induction of inflammatory mediators in murine macrophages. Mol. Immunol. 57, 284–291.
Kim, Y.J. and Lee, S.H. 2014. Reducing the bioactivity of Tannerella forsythia lipopolysaccharide by Porphyromonas gingivalis. J. Microbiol. 52, 702–708.
Kreth, J., Merritt, J., Shi, W., and Qi, F. 2005. Competition and coexistence between Streptococcus mutans and Streptococcus sanguinis in the dental biofilm. J. Bacteriol. 187, 7193–7203.
Kurata, S. 2014. Peptidoglycan recognition proteins in drosophila immunity. Dev. Comp. Immunol. 42, 36–41.
Lee, S.H. and Baek, D.H. 2013. Characteristics of Porphyromonas gingivalis lipopolysaccharide in co-culture with Fusobacterium nucleatum. Mol. Oral. Microbiol. 28, 230–238.
Lee, S.H., Jun, H.K., Lee, H.R., Chung, C.P., and Choi, B.K. 2010. Antibacterial and lipopolysaccharide (LPS)-neutralising activity of human cationic antimicrobial peptides against periodontopathogens. Int. J. Antimicrob. Agents 35, 138–145.
Lee, S.H., Kim, K.K., Rhyu, I.C., Koh, S., Lee, D.S., and Choi, B.K. 2006. Phenol/water extract of Treponema socranskii subsp. socranskii as an antagonist of toll-like receptor 4 signalling. Microbiology 152, 535–546.
Lerouge, I. and Vanderleyden, J. 2002. O-antigen structural variation: Mechanisms and possible roles in animal/plant-microbe interactions. FEMS Microbiol. Rev. 26, 17–47.
Leung, C.H., Lam, W., Ma, D.L., Gullen, E.A., and Cheng, Y.C. 2009. Butyrate mediates nucleotide-binding and oligomerisation domain (nod) 2-dependent mucosal immune responses against peptidoglycan. Eur. J. Immunol. 39, 3529–3537.
Marsh, P.D. 2006. Dental plaque as a biofilm and a microbial community - implications for health and disease. BMC Oral Health. 6 Suppl 1, S14.
Moynihan, P.J., Sychantha, D., and Clarke, A.J. 2014. Chemical biology of peptidoglycan acetylation and deacetylation. Bioorg. Chem. 54, 44–50.
Muhvic, D., El-Samalouti, V., Flad, H.D., Radosevic-Stasic, B., and Rukavina, D. 2001. The involvement of CD14 in the activation of human monocytes by peptidoglycan monomers. Mediators Inflamm. 10, 155–162.
Mychajlonka, M., McDowell, T.D., and Shockman, G.D. 1980. Conservation of cell wall peptidoglycan by strains of Streptococcus mutans and Streptococcus sanguis. Infect. Immun. 28, 65–73.
Rietschel, E.T., Schletter, J., Weidemann, B., El-Samalouti, V., Mattern, T., Zahringer, U., Seydel, U., Brade, H., Flad, H.D., Kusumoto, S., et al. 1998. Lipopolysaccharide and peptidoglycan: CD14-dependent bacterial inducers of inflammation. Microb. Drug. Resist. 4, 37–44.
Schytte Blix, I.J., Helgeland, K., Hvattum, E., and Lyberg, T. 1999. Lipopolysaccharide from Actinobacillus actinomycetemcomitans stimulates production of interleukin-1beta, tumor necrosis factor-alpha, interleukin-6 and interleukin-1 receptor antagonist in human whole blood. J. Periodontal. Res. 34, 34–40.
Socransky, S.S. and Haffajee, A.D. 1992. The bacterial etiology of destructive periodontal disease: Current concepts. J. Periodontol. 63, 322–331.
Socransky, S.S. and Haffajee, A.D. 2005. Periodontal microbial ecology. Periodontol 2000 38, 135–187.
Stingu, C.S., Eschrich, K., Rodloff, A.C., Schaumann, R., and Jentsch, H. 2008. Periodontitis is associated with a loss of colonization by Streptococcus sanguinis. J. Med. Microbiol. 57, 495–499.
Stinson, M.W., Safulko, K., and Levine, M.J. 1991. Adherence of Porphyromonas (Bacteroides) gingivalis to Streptococcus sanguis in vitro. Infect. Immun. 59, 102–108.
Volz, T., Nega, M., Buschmann, J., Kaesler, S., Guenova, E., Peschel, A., Rocken, M., Gotz, F., and Biedermann, T. 2010. Natural Staphylococcus aureus-derived peptidoglycan fragments activate NOD2 and act as potent costimulators of the innate immune system exclusively in the presence of TLR signals. FASEB J. 24, 4089–4102.
Weber, J.R., Freyer, D., Alexander, C., Schroder, N.W., Reiss, A., Kuster, C., Pfeil, D., Tuomanen, E.I., and Schumann, R.R. 2003. Recognition of pneumococcal peptidoglycan: An expanded, pivotal role for LPS binding protein. Immunity 19, 269–279.
Wu, Z., Pan, D., Guo, Y., Sun, Y., and Zeng, X. 2015. Peptidoglycan diversity and anti-inflammatory capacity in Lactobacillus strains. Carbohydr. Polym. 128, 130–137.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SH. Antagonistic effect of peptidoglycan of Streptococcus sanguinis on lipopolysaccharide of major periodontal pathogens. J Microbiol. 53, 553–560 (2015). https://doi.org/10.1007/s12275-015-5319-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-015-5319-6