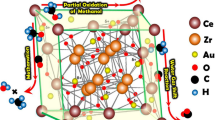
Abstract
The catalytic behavior of ultra-low-gold-loaded Au/CeO2 during CO oxidation was studied using HRTEM, TGA, in-situ FTIR, CO–TPD, O2–TPD, and O2–TPO. Herein, water, which facilitates CO oxidation, is present on the catalyst rather than obtained from the reactant gas. The experimental results show that the presence of water on the catalyst enhances the adsorption and activation of O2 to produce Oact on the catalyst surface, resulting in excellent catalytic activity. Different reaction pathways for CO oxidation over Au/CeO2 may be present; however, the dominant pathway is influenced by the species on the catalyst surface.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bond, G. C.; Louis, C.; Thompson, D. T. Catalysis by Gold. London, 2006.
Carabineiro, S. A. C.; Bogdanchikova, N.; Avalos-Borja, M.; Pestryakov, A.; Tavares, P. B.; Figueiredo, J. L. Gold supported on metal oxides for carbon monoxide oxidation. Nano. Res. 2011, 4, 180–193.
Wu, B. H.; Zhang, H.; Chen, C.; Lin, S. C.; Zheng, N. F. Interfacial activation of catalytically inert Au (6.7 nm)-Fe3O4 dumbbell nanoparticles for CO oxidation. Nano. Res. 2009, 2, 975–983.
Peng, S.; Lee, Y. M.; Wang, C.; Yin, H. F.; Dai, S.; Sun, S. H. A facile synthesis of monodisperse Au nanoparticles and their catalysis of CO oxidation. Nano. Res. 2008, 1, 229–234.
Kast, P.; Kucerová, G.; Behm, R. J. On the nature of the active Au species: CO oxidation on cyanide leached Au/TiO2 catalysts. Catal. Today 2015, 244, 146–160.
Gavril, D.; Georgakab, A.; Loukopoulosb, V.; Karaiskakisb, G.; Nieuwenhuysa, B. E. On the mechanism of selective CO oxidation on nanosized Au/γ-Al2O3 catalysts. Gold. Bull. 2006, 39, 192–199.
Gavril, D. CO oxidation on nanosized Au/Al2O3 by surface hydroxyl groups and in the absence of O2, studied by inverse gas chromatography. Catal. Today 2015, 244, 36–46.
Hutchings, G. J. Catalysis by gold. Catal. Today. 2005, 100, 55–61.
Chen, M. S.; Goodman, D. W. The structure of catalytically active gold on titania. Science. 2004, 306, 252–255.
Vijay, A.; Mills, G.; Metiu, H. Adsorption of gold on stoichiometric and reduced rutile TiO2 (110) surfaces. J. Chem. Phys. 2003, 118, 6536–6551.
Varganov, S. A.; Olson, R. M.; Gordon, M. S.; Metiu, H. The interaction of oxygen with small gold clusters. J. Chem. Phys. 2003, 119, 2531–2537.
Li, Q. L.; Zhang, Y. H.; Chen, G. X.; Fan, J. Q.; Lan, H. Q.; Yang, Y. Q. Ultra-low-gold loading Au/CeO2 catalysts for ambient temperature CO oxidation: Effect of preparation conditions on surface composition and activity. J. Catal. 2010, 273, 167–176.
Date, M.; Haruta, M. Moisture effect on CO oxidation over Au/TiO2 catalyst. J. Catal. 2001, 201, 221–224.
Kung, H. H.; Kung, M. C.; Costello, C. K. Supported Au catalysts for low temperature CO oxidation. J. Catal. 2003, 216, 425–432.
Calla, J. T.; Davis, R. J. Oxygen-exchange reactions during CO oxidation over titania-and alumina-supported Au nanoparticles. J. Catal. 2006, 241, 407–416.
Date, M.; Okumura, M.; Tsubota, S.; Haruta, M. Vital role of moisture in the catalytic activity of supported gold nanoparticles. Angew. Chem. Int. Ed. 2004, 43, 2129–2132.
Gao, F.; Wood, T. E.; Goodman, D. W. The effects of water on CO oxidation over TiO2 supported Au catalysts. Catal. Lett. 2010, 134, 9–12.
Liu, L. M.; McAllister, B.; Ye, H. Q.; Hu, P. Identifying an O2 supply pathway in CO oxidation on Au/TiO2(110): A density functional theory study on the intrinsic role of water. J. Am. Chem. Soc. 2006, 128, 4017–4022.
Bongiorno, A.; Landman, U. Water-enhanced catalysis of CO oxidation on free and supported gold nanoclusters. Phys. Rev. Lett. 2005, 95, 106102–106105.
Ojeda, M.; Zhan, B. Z.; Lglesia, E. Mechanistic interpretation of CO oxidation turnover rates on supported Au clusters. J. Catal. 2012, 285, 92–102.
Saavedra, J.; Doan, H. A.; Pursell, C. J.; Grabow, L. C.; Chandler, B. D. The critical role of water at the gold-titania interface in catalytic CO oxidation. Science. 2014, 345, 1599–1602.
Karpenko, A.; Leppelt, R.; Plzak, V.; Behm, R. J. The role of cationic Au3+ and nonionic Au0 species in the low temperature water-gas shift reaction on Au/CeO2 catalysts. J. Catal. 2007, 252, 231–242.
Ntho, T. A.; Anderson, J. A.; Scurrell, M. S. CO oxidation over titanate nanotube supported Au: Deactivation due to bicarbonate. J. Catal. 2009, 261, 94–100.
Fu, Q.; Saltsburg, H.; Flytzani-Stephanopoulos, M. Active nonmetallic Au and Pt species on ceria-based water-gas shift catalysts. Science. 2003, 301, 935–938.
Karpenko, A.; Denkwitz, Y.; Plzak, V.; Cai, J.; Leppelt, R.; Schumacher, B.; Behm, R. J. Low-temperature water-gas shift reaction on Au/CeO2 catalysts-the influence of catalyst pre-treatment on the activity and deactivation in idealized reformate. Catal. Lett. 2007, 116, 105–115.
Boccuzzi, F.; Chiorino, A.; Manzoli, M.; Lu, P.; Akita, T.; Ichikawa, S.; Haruta, M. Au/TiO2 nanosized samples: A catalytic, TEM, and FTIR study of the effect of calcination temperature on the CO oxidation. J. Catal. 2001, 202, 256–267.
Kim, C. H.; Thompson, L. T. On the importance of nanocrystalline gold for Au/CeO2 water–gas shift catalysts. J. Catal. 2006, 244, 248–250.
Parida, K. M.; Sahu, N.; Mohapatra, P.; Scurrell, M. S. Low temperature CO oxidation over gold supported mesoporous Fe–TiO2. J. Molecular. Catal. A: Chem. 2010, 319, 92–97.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, Q., Xie, W., Chen, G. et al. The behaviors of ultra-low-gold-loaded catalysts (Au/CeO2) for CO oxidation in the presence of water on the catalysts. Nano Res. 8, 3075–3084 (2015). https://doi.org/10.1007/s12274-015-0813-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-015-0813-z