Abstract
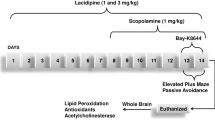
Alzheimer’s disease (AD) is the most prevalent cause of dementia in the elderly people. The disease is pathologically characterized by extracellular deposition of beta-amyloid peptide (Aβ), cholinergic neurodegeneration and elevation of acetylcholine esterase (AChE) activity in the affected regions. In this study, we investigated the effects of obovatol on memory dysfunction, which was caused by scopolamine. Obovatol (0.2, 0.5 and 1 mg/kg for 7 day) attenuated scopolamine (1 mg/kg, i.p.)-induced amnesia in a dose-dependent manner, as revealed by the Morris water maze test and step-through passive avoidance test. Mechanism studies exhibited that obovatol dose-dependently alleviated scopolamine-induced increase in Aβ generation and β-secretase activity in the cortex and hippocampus. Obovatol also attenuated scopolamine-induced rise in AChE activity in the cortex and hippocampus. Obovatol might rescue scopolamine-mediated impaired learning and memory function by attenuating Aβ accumulation and stabilizing cholinergic neurotransmission, which suggests that the natural compound could be a useful agent for the prevention of the development or progression of AD neurodegeneration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmed, T. and Gilani, A. H., Inhibitory effect of curcuminoids on acetylcholinesterase activity and attenuation of scopolamine-induced amnesia may explain medicinal use of turmeric in Alzheimer’s disease. Pharmacol. Biochem. Behav., 91, 554–559 (2009).
Auld, D. S., Kornecook, T. J., Bastianetto, S., and Quirion, R., Alzheimer’s disease and the basal forebrain cholinergic system: relations to beta-amyloid peptides, cognition, and treatment strategies. Prog. Neurobiol., 68, 209–245 (2002).
Bailey, J. A., Ray, B., Greig, N. H., and Lahiri, D. K., Rivastigmine lowers Abeta and increases sAPPalpha levels, which parallel elevated synaptic markers and metabolic activity in degenerating primary rat neurons. PLoS One, 6, e21954 (2011).
Beatty, W. W., Butters, N., and Janowsky, D. S., Patterns of memory failure after scopolamine treatment: implications for cholinergic hypotheses of dementia. Behav. Neural Biol., 45, 196–211 (1986).
Choi, D. Y., Lee, J. W., Lin, G., Lee, Y. J., Ban, J. O., Lee, Y. H., Choi, I. S., Han, S. B., Jung, J. K., Lee, W. S., Lee, S. H., Kwon, B. M., Oh, K. W., and Hong, J. T., Obovatol improves cognitive functions in animal models for Alzheimer’s disease. J. Neurochem., 120, 1048–1059 (2012a).
Choi, D. Y., Lee, J. W., Lin, G., Lee, Y. K., Lee, Y. H., Choi, I. S., Han, S. B., Jung, J. K., Kim, Y. H., Kim, K. H., Oh, K. W., Hong, J. T., and Lee, M. S., Obovatol attenuates LPSinduced memory impairments in mice via inhibition of NF-kappaB signaling pathway. Neurochem. Int., 60, 68–77 (2012b).
Cole, S. L. and Vassar, R., The role of amyloid precursor protein processing by BACE1, the beta-secretase, in Alzheimer disease pathophysiology. J. Biol. Chem., 283, 29621–29625 (2008).
Ellman, G. L., Courtney, K. D., Andres, V., Jr., and Feather-Stone, R. M., A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol., 7, 88–95 (1961).
Ensinger, H. A., Doods, H. N., Immel-Sehr, A. R., Kuhn, F. J., Lambrecht, G., Mendla, K. D., Muller, R. E., Mutschler, E., Sagrada, A., Walther, G., and Hammer, R., WAL 2014—a muscarinic agonist with preferential neuron-stimulating properties. Life Sci., 52, 473–480 (1993).
Glabe, C. G., Structural classification of toxic amyloid oligomers. J. Biol. Chem., 283, 29639–29643 (2008).
Ho, K. Y., Tsai, C. C., Chen, C. P., Huang, J. S., and Lin, C. C., Antimicrobial activity of honokiol and magnolol isolated from Magnolia officinalis. Phytother. Res., 15, 139–141 (2001).
Hock, C., Maddalena, A., Heuser, I., Naber, D., Oertel, W., Von Der Kammer, H., Wienrich, M., Raschig, A., Deng, M., Growdon, J. H., and Nitsch, R. M., Treatment with the selective muscarinic agonist talsaclidine decreases cerebrospinal fluid levels of total amyloid beta-peptide in patients with Alzheimer’s disease. Ann. N. Y. Acad. Sci., 920, 285–291 (2000).
Huber, K. and Superti-Furga, G., After the grape rush: sirtuins as epigenetic drug targets in neurodegenerative disorders. Bioorg. Med. Chem., 19, 3616–3624 (2011).
Kaduszkiewicz, H., Zimmermann, T., Beck-Bornholdt, H. P., and Van Den Bussche, H., Cholinesterase inhibitors for patients with Alzheimer’s disease: systematic review of randomised clinical trials. BMJ, 331, 321–327 (2005).
Kwon, S. H., Lee, H. K., Kim, J. A., Hong, S. I., Kim, H. C., Jo, T. H., Park, Y. I., Lee, C. K., Kim, Y. B., Lee, S. Y., and Jang, C. G., Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur. J. Pharmacol., 649, 210–217 (2010).
Lee, B., Shim, I., Lee, H., and Hahm, D. H., Rehmannia glutinosa ameliorates scopolamine-induced learning and memory impairment in rats. J. Microbiol. Biotechnol., 21, 874–883 (2011).
Lee, S. K., Kim, H. N., Kang, Y. R., Lee, C. W., Kim, H. M., Han, D. C., Shin, J., Bae, K., and Kwon, B. M., Obovatol inhibits colorectal cancer growth by inhibiting tumor cell proliferation and inducing apoptosis. Bioorg. Med. Chem., 16, 8397–8402 (2008).
Lee, Y. K., Choi, I. S., Kim, Y. H., Kim, K. H., Nam, S. Y., Yun, Y. W., Lee, M. S., Oh, K. W., and Hong, J. T., Neurite outgrowth effect of 4-O-methylhonokiol by induction of neurotrophic factors through ERK activation. Neurochem. Res., 34, 2251–2260 (2009a).
Lee, Y. K., Yuk, D. Y., Kim, T. I., Kim, Y. H., Kim, K. T., Kim, K. H., Lee, B. J., Nam, S. Y., and Hong, J. T., Protective effect of the ethanol extract of Magnolia officinalis and 4-O-methylhonokiol on scopolamine-induced memory impairment and the inhibition of acetylcholinesterase activity. J. Nat. Med., 63, 274–282 (2009b).
Lee, Y. K., Song, J. K., Choi, I. S., Jeong, J. H., Moon, D. C., Yun, Y. P., Han, S. B., Oh, K. W., and Hong, J. T., Neurotrophic activity of obovatol on the cultured embryonic rat neuronal cells by increase of neurotrophin release through activation of ERK pathway. Eur. J. Pharmacol., 649, 168–176 (2010).
Lin, L., Leblanc, C. J., Deacon, T. W., and Isacson, O., Chronic cognitive deficits and amyloid precursor protein elevation after selective immunotoxin lesions of the basal forebrain cholinergic system. Neuroreport, 9, 547–552 (1998).
Liskowsky, W. and Schliebs, R., Muscarinic acetylcholine receptor inhibition in transgenic Alzheimer-like Tg2576 mice by scopolamine favours the amyloidogenic route of processing of amyloid precursor protein. Int. J. Dev. Neurosci., 24, 149–156 (2006).
Matsui, N., Takahashi, K., Takeichi, M., Kuroshita, T., Noguchi, K., Yamazaki, K., Tagashira, H., Tsutsui, K., Okada, H., Kido, Y., Yasui, Y., Fukuishi, N., Fukuyama, Y., and Akagi, M., Magnolol and honokiol prevent learning and memory impairment and cholinergic deficit in SAMP8 mice. Brain Res., 1305, 108–117 (2009).
Messamore, E., Warpman, U., Ogane, N., and Giacobini, E., Cholinesterase inhibitor effects on extracellular acetylcholine in rat cortex. Neuropharmacology, 32, 745–750 (1993).
Muller, D. M., Mendla, K., Farber, S. A., and Nitsch, R. M., Muscarinic M1 receptor agonists increase the secretion of the amyloid precursor protein ectodomain. Life Sci., 60, 985–991 (1997).
Nitsch, R. M., Deng, M., Tennis, M., Schoenfeld, D., and Growdon, J. H., The selective muscarinic M1 agonist AF102B decreases levels of total Abeta in cerebrospinal fluid of patients with Alzheimer’s disease. Ann. Neurol., 48, 913–918 (2000).
Nitsch, R. M., Slack, B. E., Wurtman, R. J., and Growdon, J. H., Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science, 258, 304–307 (1992).
Nordberg, A. and Svensson, A. L., Cholinesterase inhibitors in the treatment of Alzheimer’s disease: a comparison of tolerability and pharmacology. Drug Saf., 19, 465–480 (1998).
Ock, J., Han, H. S., Hong, S. H., Lee, S. Y., Han, Y. M., Kwon, B. M., and Suk, K., Obovatol attenuates microglia-mediated neuroinflammation by modulating redox regulation. Br. J. Pharmacol., 159, 1646–1662 (2010).
Park, S. J., Kim, D. H., Jung, J. M., Kim, J. M., Cai, M., Liu, X., Hong, J. G., Lee, C. H., Lee, K. R., and Ryu, J. H., The ameliorating effects of stigmasterol on scopolamine-induced memory impairments in mice. Eur. J. Pharmacol., 676, 64–70 (2012).
Petersen, R. C., Scopolamine induced learning failures in man. Psychopharmacology (Berl.), 52, 283–289 (1977).
Preston, G. C., Brazell, C., Ward, C., Broks, P., Traub, M., and Stahl, S. M., The scopolamine model of dementia: determination of central cholinomimetic effects of physostigmine on cognition and biochemical markers in man. J. Psychopharmacol., 2, 67–79 (1988).
Seo, J. J., Lee, S. H., Lee, Y. S., Kwon, B. M., Ma, Y., Hwang, B. Y., Hong, J. T., and Oh, K. W., Anxiolytic-like effects of obovatol isolated from Magnolia obovata: involvement of GABA/benzodiazepine receptors complex. Prog. Neuropsychopharmacol. Biol. Psychiatry, 31, 1363–1369 (2007).
Uriarte-Pueyo, I. and Calvo, M. I., Flavonoids as acetylcholinesterase inhibitors. Curr. Med. Chem., 18, 5289–5302 (2011).
Weinreb, O., Amit, T., Mandel, S., and Youdim, M. B., Neuroprotective molecular mechanisms of (−)-epigallocatechin-3-gallate: a reflective outcome of its antioxidant, iron chelating and neuritogenic properties. Genes Nutr., DOI 10.1007/s12263-009-0143-4 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, DY., Lee, YJ., Lee, S.Y. et al. Attenuation of scopolamine-induced cognitive dysfunction by obovatol. Arch. Pharm. Res. 35, 1279–1286 (2012). https://doi.org/10.1007/s12272-012-0719-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0719-1