Abstract
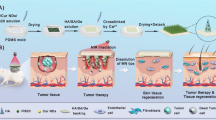
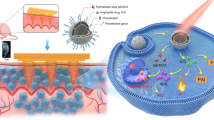
Melanoma is one of the most threatening cancers due to its metastatic capacity and its incidence is recently increasing due to ozone depletion and excessive exposure to ultraviolet. Adjuvant therapy after primary surgical resection is commonly used to prevent tumor recurrence. However, its clinical outcome is not often satisfactory, mainly due to insufficient local accumulation and the systemic toxicity of antitumor drugs. Herein, we reported a self-biodegradable tissue adhesive microneedle (MN) patch for localized and sustained delivery of doxorubicin (DOX) via microchannels for melanoma growth inhibition. DOX-loaded MN patches were fabricated by casting bioengineered mussel adhesive protein (MAP) solution containing DOX onto poly (dimethyl siloxane) molds with MN-shaped cavities under backside vacuum. The fabricated MAP-based MN patch enabled effective tissue insertion for fresh and living mice skins as well as firm surface adhesion. Notably, DOX-laden MN (DOX-MN) patch significantly suppressed B16F10 melanoma cell proliferation in vitro, while drug-free MN patch did not show any cytotoxicity for both NIH3T3 fibroblasts and B16F10 melanoma cells. In a melanoma-bearing mice model, DOX-MN patch treatment induced greater antitumor efficacy as demonstrated by significant decrease in tumor volume and weight and larger necrotic region compared with intratumoral injection of free DOX. We expect that this strategy can also integrate with other immunomodulators to provide further combinative therapy for effectively eradicating tumors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Data Availability
The raw data required to reproduce these findings will be made available upon request.
References
Parker, E. R. (2021) The influence of climate change on skin cancer incidence — a review of the evidence. Int. J. Womens Dermatol. 7: 17–27. (Erratum published 2021, Int. J. Womens Dermatol. 7: 867)
Erdmann, F., J. Lortet-Tieulent, J. Schüz, H. Zeeb, R. Greinert, E. W. Breitbart, and F. Bray (2013) International trends in the incidence of malignant melanoma 1953–2008—are recent generations at higher or lower risk? Int. J. Cancer 132: 385–400.
Naik, P. P. (2021) Cutaneous malignant melanoma: a review of early diagnosis and management. World J. Oncol. 12: 7–19.
Guy, G. P., Jr., S. R. Machlin, D. U. Ekwueme, and K. R. Yabroff (2015) Prevalence and costs of skin cancer treatment in the U.S., 2002–2006 and 2007–2011. Am. J. Prev. Med. 48: 183–187.
Sampogna, F., A. Paradisi, M. L. Iemboli, F. Ricci, G. Sonego, and D. Abeni (2019) Comparison of quality of life between melanoma and non-melanoma skin cancer patients. Eur. J. Dermatol. 29: 185–191.
Miller, K. D., L. Nogueira, A. B. Mariotto, J. H. Rowland, K. R. Yabroff, C. M. Alfano, A. Jemal, J. L. Kramer, and R. L. Siegel (2019) Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin. 69: 363–385.
Rios-Doria, J., N. Durham, L. Wetzel, R. Rothstein, J. Chesebrough, N. Holoweckyj, W. Zhao, C. C. Leow, and R. Hollingsworth (2015) Doxil synergizes with cancer immunotherapies to enhance antitumor responses in syngeneic mouse models. Neoplasia 17: 661–670.
Chitphet, K., S. M. Geary, C. H. Chan, A. L. Simons, G. J. Weiner, and A. K. Salem (2020) Combining doxorubicin-loaded PEGylated poly(lactide-co-glycolide) nanoparticles with checkpoint inhibition safely enhances therapeutic efficacy in a melanoma model. ACS Biomater. Sci. Eng. 6: 2659–2667.
Riley, R. S., C. H. June, R. Langer, and M. J. Mitchell (2019) Delivery technologies for cancer immunotherapy. Nat. Rev. Drug Discov. 18: 175–196.
Hershman, D. L., A. Eisenberger, J. Wang, J. Jacobson, V. Grann, R. McBride, W. Tsai, and A. Neugut (2007) Doxorubicin, cardiac risk factors and cardiac toxicity in elderly patients with diffuse b-cell non-Hodgkin’s lymphoma. J. Clin. Oncol. 25(18 Suppl): 9050.
Renu, K., L. P. Pureti, B. Vellingiri, and A. Valsala Gopalakrishnan (2022) Toxic effects and molecular mechanism of doxorubicin on different organs — an update. Toxin Rev. 41: 650–674.
Roberts, M. S., H. S. Cheruvu, S. E. Mangion, A. Alinaghi, H. A. Benson, Y. Mohammed, A. Holmes, J. van der Hoek, M. Pastore, and J. E. Grice (2021) Topical drug delivery: history, percutaneous absorption, and product development. Adv. Drug Deliv. Rev. 177: 113929.
Nagarkar, R., M. Singh, H. X. Nguyen, and S. Jonnalagadda (2020) A review of recent advances in microneedle technology for transdermal drug delivery. J. Drug Deliv. Sci. Technol. 59: 101923.
Ye, Y., J. Yu, D. Wen, A. R. Kahkoska, and Z. Gu (2018) Polymeric microneedles for transdermal protein delivery. Adv. Drug Deliv. Rev. 127: 106–118.
Jeon, E. Y., J. Lee, B. J. Kim, K. I. Joo, K. H. Kim, G. Lim, and H. J. Cha (2019) Bio-inspired swellable hydrogel-forming double-layered adhesive microneedle protein patch for regenerative internal/external surgical closure. Biomaterials 222: 119439.
Lim, S., T. Y. Park, E. Y. Jeon, K. I. Joo, and H. J. Cha (2021) Double-layered adhesive microneedle bandage based on biofunctionalized mussel protein for cardiac tissue regeneration. Biomaterials 278: 121171.
Waite, J. H. (1987) Nature’s underwater adhesive specialist. Int. J. Adhes. Adhes. 7: 9–14.
Liu, C. (2022) Comparative proteomics for an in-depth understanding of bioadhesion mechanisms and evolution across metazoans. J. Proteomics 256: 104506.
Hwang, D. S., Y. Gim, H. J. Yoo, and H. J. Cha (2007) Practical recombinant hybrid mussel bioadhesive fp-151. Biomaterials 28: 3560–3568.
Choi, B.-H., H. Cheong, Y. K. Jo, S. Y. Bahn, J. H. Seo, and H. J. Cha (2014) Highly purified mussel adhesive protein to secure biosafety for in vivo applications. Microb. Cell Fact. 13: 52.
Jeon, E. Y., B. H. Hwang, Y. J. Yang, B. J. Kim, B.-H. Choi, G. Y. Jung, and H. J. Cha (2015) Rapidly light-activated surgical protein glue inspired by mussel adhesion and insect structural crosslinking. Biomaterials 67: 11–19.
Kim, B. J., D. X. Oh, S. Kim, J. H. Seo, D. S. Hwang, A. Masic, D. K. Han, and H. J. Cha (2014) Mussel-mimetic protein-based adhesive hydrogel. Biomacromolecules 15: 1579–1585.
Lim, S., Y. S. Choi, D. G. Kang, Y. H. Song, and H. J. Cha (2010) The adhesive properties of coacervated recombinant hybrid mussel adhesive proteins. Biomaterials 31: 3715–3722.
Jeong, Y., Y. K. Jo, B. J. Kim, B. Yang, K. I. Joo, and H. J. Cha (2018) Sprayable adhesive nanotherapeutics: mussel-protein-based nanoparticles for highly efficient locoregional cancer therapy. ACS Nano 12: 8909–8919.
Cheong, H., Y.-J. Jun, E. Y. Jeon, J. I. Lee, H. J. Jo, H. Y. Park, E. Kim, J. W. Rhie, K. I. Joo, and H. J. Cha (2022) Sutureless neurorrhaphy system using a macrophage-polarizing in situ visible light-crosslinkable adhesive protein hydrogel for functional nerve regeneration. Chem. Eng. J. 445: 136641.
Wallin, R. F. and E. Arscott (1998) A practical guide to ISO 10993-5: cytotoxicity. Med. Device Diagn. Ind. 20: 96–98.
Park, J.-H., M. G. Allen, and M. R. Prausnitz (2005) Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery. J. Control. Release 104: 51–66.
Ranamukhaarachchi, S. A., S. Lehnert, S. L. Ranamukhaarachchi, L. Sprenger, T. Schneider, I. Mansoor, K. Rai, U. Häfeli, and B. Stoeber (2016) A micromechanical comparison of human and porcine skin before and after preservation by freezing for medical device development. Sci. Rep. 6: 32074.
Lu, Q., E. Danner, J. H. Waite, J. N. Israelachvili, H. Zeng, and D. S. Hwang (2013) Adhesion of mussel foot proteins to different substrate surfaces. J. R. Soc. Interface 10: 20120759.
Kim, S., A. Faghihnejad, Y. Lee, Y. Jho, H. Zeng, and D. S. Hwang (2015) Cation-π interaction in DOPA-deficient mussel adhesive protein mfp-1. J. Mater. Chem. B 3: 738–743.
Hwang, D. S., H. Zeng, Q. Lu, J. Israelachvili, and J. H. Waite (2012) Adhesion mechanism in a DOPA-deficient foot protein from green mussels. Soft Matter 8: 5640–5648.
Tacar, O., P. Sriamornsak, and C. R. Dass (2013) Doxorubicin: an update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 65: 157–170.
Thedrattanawong, C., C. Manaspon, and N. Nasongkla (2018) Controlling the burst release of doxorubicin from polymeric depots via adjusting hydrophobic/hydrophilic properties. J. Drug Deliv. Sci. Technol. 46: 446–451.
Senapati, S., A. K. Mahanta, S. Kumar, and P. Maiti (2018) Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target. Ther. 3: 7.
Li, J. and D. J. Mooney (2016) Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 1: 16071.
de Moura, L. D., L. N. M. Ribeiro, F. V. de Carvalho, G. H. Rodrigues da Silva, P. C. Lima Fernandes, S. Q. Brunetto, C. D. Ramos, L. A. Velloso, D. R. de Araújo, and E. de Paula (2021) Docetaxel and lidocaine co-loaded (NLC-in-hydrogel) hybrid system designed for the treatment of melanoma. Pharmaceutics 13: 1552.
Debreli Coskun, M., T. Sudha, D. J. Bharali, S. Celikler, P. J. Davis, and S. A. Mousa (2020) αvβ3 integrin antagonists enhance chemotherapy response in an orthotopic pancreatic cancer model. Front. Pharmacol. 11: 95.
Alyahya, R., T. Sudha, M. Racz, S. C. Stain, and S. A. Mousa (2015) Anti-metastasis efficacy and safety of non-anticoagulant heparin derivative versus low molecular weight heparin in surgical pancreatic cancer models. Int. J. Oncol. 46: 1225–1231.
Vargason, A. M., A. C. Anselmo, and S. Mitragotri (2021) The evolution of commercial drug delivery technologies. Nat. Biomed. Eng. 5: 951–967.
Li, L., R. Chen, D. Zhou, J. Sun, L. Wang, L. Zhu, H. Shen, W. Xie, and X. Ye (2022) The efficacy and cardiac toxicity of different-dose pegylated liposomal doxorubicin in elderly patients with diffuse large B lymphoma. Cancer Med. Advance online publication. doi: https://doi.org/10.1002/cam4.5280
Barenholz, Y. C. (2012) Doxil®—the first FDA-approved nanodrug: lessons learned. J. Control. Release 160: 117–134.
Kim, H. J., B. Yang, T. Y. Park, S. Lim, and H. J. Cha (2017) Complex coacervates based on recombinant mussel adhesive proteins: their characterization and applications. Soft Matter 13: 7704–7716.
Park, T. Y., J.-M. Oh, J. S. Cho, S. B. Sim, J. Lee, and H. J. Cha (2020) Stem cell-loaded adhesive immiscible liquid for regeneration of myocardial infarction. J. Control. Release 321: 602–615.
Park, T. Y., E. Y. Jeon, H. J. Kim, B.-H. Choi, and H. J. Cha (2019) Prolonged cell persistence with enhanced multipotency and rapid angiogenesis of hypoxia pre-conditioned stem cells encapsulated in marine-inspired adhesive and immiscible liquid micro-droplets. Acta Biomater. 86: 257–268.
Acknowledgements
We acknowledge the financial support by the High Value-added Food Technology Development Program (grant number: 321025051HD060) through the Korea Institute of Planning and Evaluation for Technology in Food, Agriculture and Forestry (IPET), funded by the Ministry of Agriculture, Food & Rural Affairs, Republic of Korea.
Author information
Authors and Affiliations
Contributions
J. Heo: Conceptualization, Validation, Investigation, Writing. E.Y. Jeon: Validation, Methodology, Visualization, Writing. K.I. Joo: Conceptualization, Validation. H.J. Cha: Supervision, Funding acquisition, Resources, Writing.
Corresponding author
Ethics declarations
The authors declare no competing financial interests or personal relationships. All animal studies were conducted according to guidelines of national regulations and the approval of the local Institutional Animal Care and Use Committee (POSTECH-2018-0009).
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Heo, J., Jeon, E.Y., Joo, K.I. et al. Locoregional Melanoma Therapy by Tissue Adhesive Microneedle Patch-assisted Trans-tumoral Delivery of Anticancer Drug. Biotechnol Bioproc E 28, 473–482 (2023). https://doi.org/10.1007/s12257-022-0299-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0299-y