Abstract
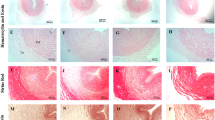
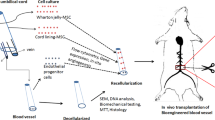
Decellularized human umbilical arteries (dHUA) is an off-the-shelf graft that can potentially serve as vascular scaffolds in tissue engineering of small-diameter vascular grafts. This research aimed to investigate that dHUA could exhibit adequate endothelialization for a long term in xenogenic transplantation. 13 dHUAs were implanted in rat abdominal aortas up to 90 days. Rats were divided into three groups in terms of survival period: Group 1, one to seven days (n = 6); Group 2, 14 to 30 days (n = 4) and Group 3, 90 days (n = 3). The explants were analyzed by histological, immunohistochemistry and magnetic resonance angiography (MRA) examination. Allograft implantation of 12 decellularized rat abdominal aortas’ were processed the same way as the rat in order to make a comparison for survival rates (Group 1, n = 5; Group 2, n = 4; Group 3, n = 3). The results demonstrated that the survival rates of xenograft and allograft implantation were estimated to be 59.2% vs. 58.3% in Group 1, 50.7% vs. 58.3% in Group 2 and 3. Grafts harvested from Group 2 were showed CD31, endothelial nitric oxide synthase expression at intima, and α-smooth muscle actin, CD45, CD68 and CD168 expression at the tunica externa. A layer structure with obvious endothelialization and fiber regeneration/orientation could be inspected from the explants of Group 3. MRA demonstrated the patency of dHUA on day 30 and 90. In conclusion, more than 50% dHUA maintained patency in the xenogenic model till 90 days after surgery. A mature vessel-like functional structure with intact endothelial layer was observed then. This warrants further study in the reinforcement of decellularized vascular scaffolds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ma, X., Z. He, L. Li, G. Liu, Q. Li, D. Yang, Y. Zhang, and N. Li (2017) Development and in vivo validation of tissue-engineered, small-diameter vascular grafts from decellularized aortae of fetal pigs and canine vascular endothelial cells. J. Cardiothorac. Surg. 12: 101.
Kaushal, S., G. E. Amiel, K. J. Guleserian, O. M. Shapira, T. Perry, F. W. Sutherland, E. Rabkin, A. M. Moran, F. J. Schoen, A. Atala, S. Soker, J. Bischoff, and J. E. Mayer Jr. (2001) Functional small-diameter neovessels created using endothelial progenitor cells expanded ex vivo. Nat. Med. 7: 1035–1040.
Tillman, B. W., S. K. Yazdani, L. P. Neff, M. A. Corriere, G. J. Christ, S. Soker, A. Atala, R. L. Geary, and J. J. Yoo (2012) Bioengineered vascular access maintains structural integrity in response to arteriovenous flow and repeated needle puncture. J. Vasc. Surg. 56: 783–793.
Neff, L. P., B. W. Tillman, S. K. Yazdani, M. A. Machingal, J. J. Yoo, S. Soker, B. W. Bernish, R. L. Geary, and G. J. Christ (2011) Vascular smooth muscle enhances functionality of tissue-engineered blood vessels in vivo. J. Vasc. Surg. 53: 426–434.
Lopera Higuita, M. and L. G. Griffiths (2020) Small diameter xenogeneic extracellular matrix scaffolds for vascular applications. Tissue Eng. Part B Rev. 26: 26–45.
Williams, C., J. Liao, E. M. Joyce, B. Wang, J. B. Leach, M. S. Sacks, and J. Y. Wong (2009) Altered structural and mechanical properties in decellularized rabbit carotid arteries. Acta Biomater. 5: 993–1005.
Clarke, D. R., R. M. Lust, Y. S. Sun, K. S. Black, and J. D. Ollerenshaw (2001) Transformation of nonvascular acellular tissue matrices into durable vascular conduits. Ann. Thorac. Surg. 71(5 Suppl): S433–S436.
Gui, L., A. Muto, S. A. Chan, C. K. Breuer, and L. E. Niklason (2009) Development of decellularized human umbilical arteries as small-diameter vascular grafts. Tissue Eng. Part A 15: 2665–2676.
Sharp, M. A., D. Phillips, I. Roberts, and L. Hands (2004) A cautionary case: the SynerGraft vascular prosthesis. Eur. J. Vasc. Endovasc. Surg. 27: 42–44.
Towne, J. B. and L. H. Hollier (2004) Complications in Vascular Surgery. 2nd ed. CRC Press.
Quint, C., Y. Kondo, R. J. Manson, J. H. Lawson, A. Dardik, and L. E. Niklason (2011) Decellularized tissue-engineered blood vessel as an arterial conduit. Proc. Natl. Acad. Sci. U. S. A. 108: 9214–9219.
Allaire, E., R. Forough, M. Clowes, B. Starcher, and A. W. Clowes (1998) Local overexpression of TIMP-1 prevents aortic aneurysm degeneration and rupture in a rat model. J. Clin. Invest. 102: 1413–1420.
Allaire, E., C. Guettier, P. Bruneval, D. Plissonnier, and J.-B. Michel (1994) Cell-free arterial grafts: morphologic characteristics of aortic isografts, allografts, and xenografts in rats. J. Vasc. Surg. 19: 446–456.
Hsia, K., C. H. Lin, H. Y. Lee, W. M. Chen, C. L. Yao, C. C. Chen, H. Ma, S. J. Wang, and J. H. Lu (2019) Sphingosine-1- phosphate in endothelial cell recellularization improves patency and endothelialization of decellularized vascular grafts in vivo. Int. J. Mol. Sci. 20: 1641.
Hsia, K., M. J. Yang, W. M. Chen, C. L. Yao, C. H. Lin, C. C. Loong, Y. L. Huang, Y. T. Lin, A. D. Lander, H. Lee, and J. H. Lu (2017) Sphingosine-1-phosphate improves endothelialization with reduction of thrombosis in recellularized human umbilical vein graft by inhibiting syndecan-1 shedding in vitro. Acta Biomater. 51: 341–350.
Crapo, P. M., T. W. Gilbert, and S. F. Badylak (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32: 3233–3243.
Lin, C.-H., Y.-C. Kao, Y.-H. Lin, H. Ma, and R.-Y. Tsay (2016) A fiber-progressive-engagement model to evaluate the composition, microstructure, and nonlinear pseudoelastic behavior of porcine arteries and decellularized derivatives. Acta Biomater. 46: 101–111.
Morris, A. H., D. K. Stamer, and T. R. Kyriakides (2017) The host response to naturally-derived extracellular matrix biomaterials. Semin. Immunol. 29: 72–91.
Human, P. and P. Zilla (2001) Inflammatory and immune processes: the neglected villain of bioprosthetic degeneration? J. Long Term Eff. Med. Implants 11: 199–220.
Rieder, E., A. Nigisch, B. Dekan, M.-T. Kasimir, F. Mühlbacher, E. Wolner, P. Simon, and G. Weigel (2006) Granulocyte-based immune response against decellularized or glutaraldehyde cross-linked vascular tissue. Biomaterials 27: 5634–5642.
Goetzl, E. J., M. J. Banda, and D. Leppert (1996) Matrix metalloproteinases in immunity. J. Immunol. 156: 1–4.
Suma, H., T. Isomura, T. Horii, and T. Sato (2000) Late angiographic result of using the right gastroepiploic artery as a graft. J. Thorac. Cardiovasc. Surg. 120: 496–498.
Chiu, T. and A. Burd (2005) “Xenograft” dressing in the treatment of burns. Clin. Dermatol. 23: 419–423.
Courtman, D. W., B. F. Errett, and G. J. Wilson (2001) The role of crosslinking in modification of the immune response elicited against xenogenic vascular acellular matrices. J. Biomed. Mater. Res. 55: 576–586.
Valentin, J. E., N. J. Turner, T. W. Gilbert, and S. F. Badylak (2010) Functional skeletal muscle formation with a biologic scaffold. Biomaterials 31: 7475–7484.
Gawaz, M., H. Langer, and A. E. May (2005) Platelets in inflammation and atherogenesis. J. Clin. Invest. 115: 3378–3384.
McEver, R. P. (2001) Adhesive interactions of leukocytes, platelets, and the vessel wall during hemostasis and inflammation. Thromb. Haemost. 86: 746–756.
Valentin, J. E., A. M. Stewart-Akers, T. W. Gilbert, and S. F. Badylak (2009) Macrophage participation in the degradation and remodeling of extracellular matrix scaffolds. Tissue Eng. Part A 15: 1687–1694.
Wolf, M. T., K. A. Daly, E. P. Brennan-Pierce, S. A. Johnson, C. A. Carruthers, A. D’Amore, S. P. Nagarkar, S. S. Velankar, and S. F. Badylak (2012) A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials 33: 7028–7038.
Roh, J. D., R. Sawh-Martinez, M. P. Brennan, S. M. Jay, L. Devine, D. A. Rao, T. Yi, T. L. Mirensky, A. Nalbandian, B. Udelsman, N. Hibino, T. Shinoka, W. M. Saltzman, E. Snyder, T. R. Kyriakides, J. S. Pober, and C. K. Breuer (2010) Tissue-engineered vascular grafts transform into mature blood vessels via an inflammation-mediated process of vascular remodeling. Proc. Natl. Acad. Sci. U. S. A. 107: 4669–4674.
Tsai, T.-N., J. P. Kirton, P. Campagnolo, L. Zhang, Q. Xiao, Z. Zhang, W. Wang, Y. Hu, and Q. Xu (2012) Contribution of stem cells to neointimal formation of decellularized vessel grafts in a novel mouse model. Am. J. Pathol. 181: 362–373.
Vorotnikova, E., D. McIntosh, A. Dewilde, J. Zhang, J. E. Reing, L. Zhang, K. Cordero, K. Bedelbaeva, D. Gourevitch, E. Heber-Katz, S. F. Badylak, and S. J. Braunhut (2010) Extracellular matrix-derived products modulate endothelial and progenitor cell migration and proliferation in vitro and stimulate regenerative healing in vivo. Matrix Biol. 29: 690–700.
Avci-Adali, M., G. Ziemer, and H. P. Wendel (2010) Induction of EPC homing on biofunctionalized vascular grafts for rapid in vivo self-endothelialization—a review of current strategies. Biotechnol. Adv. 28: 119–129.
Kristofik, N. J., L. Qin, N. E. Calabro, S. Dimitrievska, G. Li, G. Tellides, L. E. Niklason, and T. R. Kyriakides (2017) Improving in vivo outcomes of decellularized vascular grafts via incorporation of a novel extracellular matrix. Biomaterials 141: 63–73.
Badylak, S. F. (2014) Decellularized allogeneic and xenogeneic tissue as a bioscaffold for regenerative medicine: factors that influence the host response. Ann. Biomed. Eng. 42: 1517–1527.
Funamoto, S., K. Nam, T. Kimura, A. Murakoshi, Y. Hashimoto, K. Niwaya, S. Kitamura, T. Fujisato, and A. Kishida (2010) The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials 31: 3590–3595.
Yamanaka, H., T. Yamaoka, A. Mahara, N. Morimoto, and S. Suzuki (2018) Tissue-engineered submillimeter-diameter vascular grafts for free flap survival in rat model. Biomaterials 179: 156–163.
Robertson, M. J., J. L. Dries-Devlin, S. M. Kren, J. S. Burchfield, and D. A. Taylor (2014) Optimizing recellularization of whole decellularized heart extracellular matrix. PLoS One 9: e90406.
Bilodeau, C., O. Goltsis, I. M. Rogers, and M. Post (2020) Limitations of recellularized biological scaffolds for human transplantation. J. Tissue Eng. Regen. Med. 14: 521–538.
Gong, W., D. Lei, S. Li, P. Huang, Q. Qi, Y. Sun, Y. Zhang, Z. Wang, Z. You, X. Ye, and Q. Zhao (2016) Hybrid small-diameter vascular grafts: anti-expansion effect of electrospun poly ε-caprolactone on heparin-coated decellularized matrices. Biomaterials 76: 359–370.
Negishi, J., Y. Hashimoto, A. Yamashita, Y. Zhang, T. Kimura, A. Kishida, and S. Funamoto (2017) Evaluation of small-diameter vascular grafts reconstructed from decellularized aorta sheets. J. Biomed. Mater. Res. A 105: 1293–1298.
Andreollo, N. A., E. F. Santos, M. R. Araújo, and L. R. Lopes (2012) Rat’s age versus human’s age: what is the relationship? Arq. Bras. Cir. Dig. 25: 49–51.
Acknowledgements
This work was supported by grants R16001 and R16002 from I-MEI FOODS CO., LTD, and Taipei Veterans General Hospital. We thank the Taiwan Mouse Clinic, Academia Sinica and Taiwan Animal Consortium for the technical support in MRA.
Funding
This research received funding support from the Taiwan Ministry of Science and Technology, MOST107-2314-B-075-030, 108-2314-B-075-053, 108-2314-B-075-045, 108WHA0110304, 109WHA0110184, and MOST 109-2314-B-038-143.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author’s Contributions
Kai Hsia: Methodology, Data curation, Writing—Original draft preparation & editing. Tien-Shiang Wang: Data curation, Review and editing. Chin-Su Liu: Investigation, Writing—Review and editing. Chih-Kuan Su: Methodology, Original draft preparation. Chien-Chin Chen: Pathology. Chang-Ching Yeh: Sample collection and preparation. Hsinyu Lee: Supervision. Chao-Ling Yao: Supervision. Tsung-Yu Tseng: Administration. Shih-Hwa Chiou: Supervision, Review and editing. Hsu Ma: Supervision. Chih-Hsun Lin: Methodology, Writing—Original draft preparation. Jen-Her Lu: Reviewing and editing, Funding acquisition, Conceptualization and supervision.
Ethical Statements
The authors declare no conflict of interest.
The study was approved by the Institutional Animal Care and Use Committee of Taipei Veterans General Hospital. All animal care complied with the Guide for the Care and Use of Laboratory Animals (No. 2018-265). Human tissue was obtained using protocols approved by the Institutional Review Board of Taipei Veterans General Hospital. All human subjects signed a consent form that was approved by the Institutional Review Board of Taipei Veterans General Hospital (No. 2020-04-006C).
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hsia, K., Wang, TS., Liu, CS. et al. Decellularized Human Umbilical Artery Exhibits Adequate Endothelialization in Xenogenic Transplantation. Biotechnol Bioproc E 28, 439–450 (2023). https://doi.org/10.1007/s12257-022-0256-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0256-9