Abstract
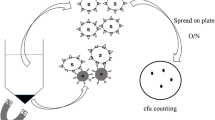
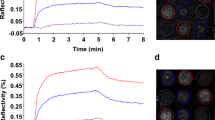
Carbohydrate-protein interaction is one of the most important molecular events deemed critical for numerous biological processes. Therefore, understanding this interaction is essential. In this study, we used bacterial spore display techniques to present multiple copies of streptavidin on the surface of spores to explore carbohydrate-protein interaction in solution and on chips. By applying bacterial spores displaying streptavidin, we developed a new method which allows sensitive, versatile, and passive detection of carbohydrate-protein interactions with a 10-fold increase in sensitivity. The linear relationship of interactions between carbohydrates and labeled concanavalin A (con A) in solution and on functionalized bacterial spore chips has also been confirmed. To the best of our knowledge, this is the first example of development and characterization of binding behavior in carbohydrateprotein interactions using bacterial spore-displayed streptavidin. We believe this strategy may enable new high-throughput screening of carbohydrate interactions as well as establish a basis for monitoring inhibitors of carbohydrate-binding proteins when developing new drugs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhao, Y. Y., M. Takahashi, J. G. Gu, E. Miyoshi, A. Matsumoto, S. Kitazume, and N. Taniguchi (2008) Functional roles of N-glycans in cell signaling and cell adhesion in cancer. Cancer Sci. 99: 1304–1310.
Jelinek, R. and S. Kolusheva (2004) Carbohydrate biosensors. Chem. Rev. 104: 5987–6016.
Smith, E. A., W. D. Thomas, L. L. Kiessling, and R. M. Corn (2003) Surface plasmon resonance imaging studies of proteincarbohydrate interactions. J. Am. Chem. Soc. 125: 6140–6148.
Hone, D. C., A. H. Haines, and D. A. Russell (2003) Rapid, quantitative colorimetric detection of a lectin using mannose-stabilized gold nanoparticles. Langmuir 19: 7141–7144.
Gao, J., D. Liu, and Z. Wang (2008) Microarray-based study of carbohydrate-protein binding by gold nanoparticle probes. Anal. Chem. 80: 8822–8827.
Honda, S. and A. Taga (2003) Studies of carbohydrate-protein interaction by capillary electrophoresis. Methods Enzymol. 362: 434–454.
Yang, Q., M. X. Hu, Z. W. Dai, J. Tian, and Z. K. Xu (2006) Fabrication of glycosylated surface on polymer membrane by UVinduced graft polymerization for lectin recognition. Langmuir 22: 9345–9349.
Kanai, M., K. H. Mortell, and L. L. Kiessling (1997) Varying the size of multivalent ligands: The dependence of concanavalin A binding on neoglycopolymer length. J. Am. Chem. Soc. 119: 9931–9932.
van Dongen, S. F. M., H. -P. M. de Hoog, R. J. R. W. Peters, M. Nallani, R. J. M. Nolte, and J. C. M. van Hest (2009) Biohybrid polymer capsules. Chem. Rev. 109: 6212–6274.
Lee, S. Y., J. H. Choi, and Z. Xu (2003) Microbial cell-surface display. Trends Biotechnol. 21: 45–52.
Kehoe, J. W. and B. K. Kay (2005) Filamentous phage display in the new millennium. Chem. Rev. 105: 4056–4072.
Stahl, S. and M. Uhlen (1997) Bacterial surface display: Trends and progress. Trends Biotechnol. 15: 185–192.
Park, T. J., K.-B. Lee, S. J. Lee, J. P. Park, Z.-W. Lee, S.-K. Choi, H.-C. Jung, J.-G. Pan, S. Y. Lee, and I. S. Choi (2004) Micropatterns of spores displaying heterologous proteins. J. Am. Chem. Soc. 126: 10512–10513.
Jung, H. -C., S. Ko, S. -J. Ju, E. -J. Kim, M. -K. Kim, and J. -G. Pan (2003) Bacterial cell surface display of lipase and its randomly mutated library facilitates high-throughput screening of mutants showing higher specific activities. J. Mol. Catalysis B: Enzymatic. 26: 177–184.
Jose, J., D. Betscheider, and D. Zangen (2005) Bacterial surface display library screening by target enzyme labeling: Identification of new human cathepsin G inhibitors. Anal. Biochem. 346: 258–267.
Gr, C. H., S. J. van Vliet, W. E. Schiphorst, C. M. Bank, S. Meyer, I. van Die, and Y. van Kooyk (2006) One-step biotinylation procedure for carbohydrates to study carbohydrate-protein interactions. Anal. Biochem. 354: 54–63.
Shao, M. -C. (1992) The use of streptavidin-biotinylglycans as a tool for characterization of oligosaccharide-binding specificity of lectin. Anal. Biochem. 205: 77–82.
Driks, A. (1999) Bacillus subtilis spore coat. Microbiol. Mol. Biol. Rev. 63: 1–20.
Park, T. J., S. -K. Choi, H. -C. Jung, S. Y. Lee, and J. -G. Pan (2009) Spore display using Bacillus thuringiensis exosporium protein InhA. J. Microbiol. Biotechnolol. 19: 495–501.
Sambrook, J., E. Fritsch, and T. Maniatis (1989) Molecular Cloning: A Laboratory Manual. 2nd ed., pp. 23–38. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, USA.
Hageman, J. H., G. W. Shankweiler, P. R. Wall, K. Franich, G. W. McCowan, S. M. Cauble, J. Grajeda, and C. Quinones (1984) Single, chemically defined sporulation medium for Bacillus subtilis: Growth, sporulation, and extracellular protease production. J. Bacteriol. 160: 438–441.
Georgiou, G., C. Stathopoulos, P. S. Daugherty, A. R. Nayak, B. L. Iverson, and R. Curtiss (1997) Display of heterologous proteins on the surface of microorganisms: from the screening of combinatorial libraries to live recombinant vaccines. Nat. Biotechnol. 15: 29–34.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, T.J., Park, J.P., Lee, S.J. et al. Quantitative studies of carbohydrate-protein interaction using functionalized bacterial spores in solution and on chips. Biotechnol Bioproc E 16, 190–195 (2011). https://doi.org/10.1007/s12257-010-0063-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-010-0063-6