Abstract
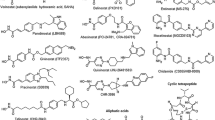
Histone deacetylases (HDACs) remove the acetyl groups of lysine residues of histone tails leading to chromatin compaction and transcriptional repression. In addition, HDACs can also influence transcription-independent events such as mitosis or deoxyribonucleic acid (DNA) repair and deacetylate nonhistone proteins involved in cell proliferation and death, altering their function. Histone deacetylase inhibitors (HDACi) constitute a promising treatment for cancer therapy due to their low toxicity. HDACi have been shown to induce differentiation, cell-cycle arrest, and apoptosis and to inhibit migration, invasion, and angiogenesis in many cancer cell lines. In addition, these compounds inhibit tumor growth in animal models and show antitumor activity in patients. HDACi alone and in combination with a variety of anticancer drugs are being tested in clinical trials, showing significant anticancer activity both in hematological and solid tumors. SAHA (vorinostat, Zolinza) was the first HDACi approved by the US Food and Drug Administration to enter the clinical oncology market for treating cutaneous T-cell lymphoma (CTCL) and is being tested for other malignancies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jenuwein T, Allis CD (2001) Translating the histone code. Science 293(5532):1074–
Riggs MG, Whittaker RG, Neumann JR, Ingram VM (1977). n-Butyrate causes histone modification in HeLa and Friend erythroleukaemia cells. Nature 268(5619):462–464
Struhl K (1998) Histone acetylation and transcriptional regulatory mechanisms. Genes Dev 12(5):599–606
Glaser KB (2007) HDAC inhibitors: clinical update and mechanism-based potential. Biochem Pharmacol 74(5):659–671
Marks PA, Dokmanovic M (2005) Histone deacetylase inhibitors: discovery and development as anticancer agents. Expert Opin Investig Drugs 14(12):1497–1511
Blander G, Guarente L (2004) The Sir2 family of protein deacetylases. Annu Rev Biochem 73:417–435
Dokmanovic M, Clarke C, Marks PA (2007) Histone deacetylase inhibitors: overview and perspectives. Mol Cancer Res 5(10):981–989
Fischle W, Dequiedt F, Hendzel MJ et al (2002) Enzymatic activity associated with class II HDACs is dependent on a multiprotein complex containing HDAC3 and SMRT/N-CoR. Mol Cell 9(1):45–57
Li J, Wang J, Wang J et al (2000) Both corepressor proteins SMRT and N-CoR exist in large protein complexes containing HDAC3. Embo J 19(16):4342–4350
Fraga MF, Ballestar E, Villar-Garea A et al (2005) Loss of acetylation at Lys16 and trimethylation at Lys20 of histone H4 is a common hallmark of human cancer. Nat Genet 37(4):391–400
Glozak MA, Seto E (2007) Histone deacetylases and cancer. Oncogene 26(37):5420–5432
Bolden JE, Peart MJ, Johnstone RW (2006) Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov 5(9):769–784
Minucci S, Pelicci PG (2006) Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer 6(1):38–51
Rosato RR, Grant S (2005) Histone deacetylase inhibitors: insights into mechanisms of lethality. Expert Opin Ther Targets 9(4):809–824
Marks PA, Breslow R (2007) Dimethyl sulfoxide to vorinostat: development of this histone deacetylase inhibitor as an anticancer drug. Nat Biotechnol 25(1):84–90
Yoshida M, Kijima M, Akita M, Beppu T (1990) Potent and specific inhibition of mammalian histone deacetylase both in vivo and in vitro by trichostatin A. J Biol Chem 265(28):17174–17179
Richon VM, Emiliani S, Verdin E et al (1998) A class of hybrid polar inducers of transformed cell differentiation inhibits histone deacetylases. Proc Natl Acad Sci U S A 95(6):3003–3007
Duvic M, Talpur R, Ni X et al (2007) Phase 2 trial of oral vorinostat (suberoylanilide hydroxamic acid, SAHA) for refractory cutaneous T-cell lymphoma (CTCL). Blood 109(1):31–39
Hu E, Dul E, Sung CM et al (2003) Identification of novel isoform-selective inhibitors within class I histone deacetylases. J Pharmacol Exp Ther 307(2):720–728
Haggarty SJ, Koeller KM, Wong JC et al (2003) Domain-selective small-molecule inhibitor of histone deacetylase 6 (HDAC6)-mediated tubulin deacetylation. Proc Natl Acad Sci U S A 100(8): 4389–4394
Peart MJ, Smyth GK, van Laar RK et al (2005) Identification and functional significance of genes regulated by structurally different histone deacetylase inhibitors. Proc Natl Acad Sci U S A 102(10):3697–3702
Xu WS, Parmigiani RB, Marks PA (2007) Histone deacetylase inhibitors: molecular mechanisms of action. Oncogene 26(37):5541–5552
Gui CY, Ngo L, Xu WS et al (2004) Histone deacetylase (HDAC) inhibitor activation of p21 WAF1 involves changes in promoter-associated proteins, including HDAC1. Proc Natl Acad Sci U S A 101(5):1241–1246
Insinga A, Monestiroli S, Ronzoni S et al (2005) Inhibitors of histone deacetylases induce tumorselective apoptosis through activation of the death receptor pathway. Nat Med 11(1):71–76
Ruefli AA, Ausserlechner MJ, Bernhard D et al (2001) The histone deacetylase inhibitor and chemotherapeutic agent suberoylanilide hydroxamic acid (SAHA) induces a cell-death pathway characterized by cleavage of Bid and production of reactive oxygen species. Proc Natl Acad Sci U S A 98(19):10833–10838
Kim MS, Kwon HJ, Lee YM et al (2001) Histone deacetylases induce angiogenesis by negative regulation of tumor suppressor genes. Nat Med 7(4): 437–443
Mori T, Kim J, Yamano T et al (2005) Epigenetic up-regulation of C-C chemokine receptor 7 and C-X-C chemokine receptor 4 expression in melanoma cells. Cancer Res 65(5):1800–1807
Whetstine JR, Ceron J, Ladd B et al (2005) Regulation of tissue-specific and extracellular matrix-related genes by a class I histone deacetylase. Mol Cell 18(4):483–490
Karagiannis TC, El-Osta A (2006) Clinical potential of histone deacetylase inhibitors as stand alone therapeutics and in combination with other chemotherapeutics or radiotherapy for cancer. Epigenetics 1(3):121–126
US National Institutes of Health, US National Library of Medicine (2008). www.clinicaltrials.gov. Accessed
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by an unrestricted educational grant from Pfizer.
Rights and permissions
About this article
Cite this article
Martínez-Iglesias, O., Ruiz-Llorente, L., Sánchez-Martínez, R. et al. Histone deacetylase inhibitors: mechanism of action and therapeutic use in cancer. Clin Transl Oncol 10, 395–398 (2008). https://doi.org/10.1007/s12094-008-0221-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-008-0221-x