Abstract
Purpose of review
To describe the clinical, laboratory, and MRI features that characterize cognitive decline in the setting of central nervous system (CNS) autoimmunity, and provide an overview of current treatment modalities.
Recent findings
The field of autoimmune neurology is rapidly expanding due to the increasing number of newly discovered autoantibodies directed against specific CNS targets. The clinical syndromes associated with these autoantibodies are heterogeneous but frequently share common, recognizable clinical, and MRI characteristics. While the detection of certain autoantibodies strongly suggest the presence of an underlying malignancy (onconeural autoantibodies), a large proportion of cases remain idiopathic. Cognitive decline and encephalopathy are common manifestations of CNS autoimmunity, and can mimic neurodegenerative disorders. Recent findings suggest that the frequency of autoimmune encephalitis in the population is higher than previously thought, and potentially rivals that of infectious encephalitis. Moreover, emerging clinical scenarios that may predispose to CNS autoimmunity are increasingly been recognized. These include autoimmune dementia/encephalitis post-herpes simplex virus encephalitis, post-transplant and in association with immune checkpoint inhibitor treatment of cancer. Early recognition of autoimmune cognitive impairment is important given the potential for reversibility and disability prevention with appropriate treatment.
Summary
Autoimmune cognitive impairment is treatable and may arise in a number of different clinical settings, with important treatment implications. Several clinical and para-clinical clues may help to differentiate these disorders from dementia of other etiologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The spectrum of neurological manifestations associated with autoantibodies directed against specific neural (neuronal or glial) targets has rapidly evolved during the last 30 years. Autoimmune dementia refers to a specific subgroup of autoimmune neurological disorders where impaired cognition represents the principal clinical manifestation, thus potentially mimicking neurodegenerative disorders (e.g., fronto-temporal dementia, Creutzfeldt-Jacob disease [CJD]) [1,2,3]. In this context, cognitive impairment may range from subtle decline in a single cognitive domain (e.g., isolated behavioral changes resembling psychiatric disorders) to impairment in multiple cognitive domains and a frank dementia [1, 4]. The clinical presentation of autoimmune encephalopathies/encephalitis, which by definition implies altered mental status, frequently overlaps with autoimmune dementia since impaired cognition represents one of the cardinal neurological features making a clear distinction difficult. [5]
Principles of autoimmune neurology
Autoimmune neurological disorders can be generically classified according to the type of antibodies they are associated with. In terms of disease pathophysiology, it is commonly accepted that antibodies directed against antigens expressed on the neural cell-surface are more likely to be pathogenic, inducing cell-damage/dysfunction or receptor internalization by direct binding to their accessible target. Conversely, antibodies targeting intracellular antigens (not accessible for direct binding in physiological conditions) are unlikely to be pathogenic and rather represent disease biomarkers of a cytotoxic T cell process. With few exceptions, disorders associated with antibodies targeting cell-surface antigens generally respond well to immunotherapy and are frequently idiopathic. On the contrary, antibodies targeting intracellular antigens typically predict poor immunotherapy response and high likelihood of underlying malignancies (onconeural antibodies) [6].
Demographic and clinical features of the main autoantibodies associated with cognitive impairment are summarized in Table 1 (cell-surface targets) and Table 2 (intracellular targets).
In clinical practice, detection of specific autoantibodies is not always possible or rapidly available, and it is likely that many autoantibodies are yet to be discovered. To facilitate early diagnosis and treatment, stringent diagnostic criteria for “seronegative” autoimmune encephalitis have been published [42••].
Epidemiology
The exact frequency of autoimmune dementia in the population is unknown. A population-based study conducted in Olmsted County (MN, USA) found the incidence and prevalence of autoimmune encephalitis to be broadly comparable to that of infectious encephalitis: incidence 0.8/100,000; prevalence 13.7/100,000. If antibody-positive cases only are considered, these numbers decrease to 0.4/100,000 and 6.5/100,000, respectively [43•]. Antibodies directed against MOG and GAD65 were the most frequently detected. In a prospective, hospital-based UK study, the autoimmune etiologies accounted for 21% of encephalitis over a 2-year period, while a study from the California encephalitis project found a similar frequency of anti-NMDAR and viral encephalitis in young individuals [44, 45]. Despite these results, it should be recognized that when compared to cognitive impairment of other etiologies (e.g., neurodegenerative, toxic/metabolic, traumatic), autoimmune dementia is much less common and care is needed in the evaluation of such patients to avoid over-diagnosis. Autoimmune dementia typically affects mid-late adulthood (50–70 years of age) but may occur at any age (Tables 1 and 2). Female sex seems overall more affected although certain antibodies are associated with male predominance (e.g., anti-DPPX, anti-CASPR2/LGI1).
Diagnostic approach
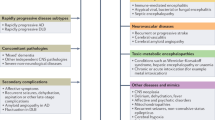
Autoimmune dementia typically presents as a complex clinical syndrome where impaired cognition is accompanied by other neurological manifestations, although isolated cognitive impairment is possible, especially at onset [46, 47]. A recent multicenter study showed that despite 80% of patients with seropositive autoimmune encephalitis having a typical presentation, the disorder was initially suspected in only 32% [48]. Identification of characteristic clinical syndromes associated with autoimmune dementia (see below) is fundamental given the potential reversibility of these diseases, in contrast to neurodegenerative dementias which are generally irreversible. In general, several clinical and para-clinical clues may suggest an autoimmune etiology in patients with new-onset cognitive impairment [49]:
-
Acute/subacute onset (days/weeks) with fluctuating or rapidly progressive course.
-
Strong personal/family history (first-degree relative) of autoimmunity.
-
History of cancer, risk factors for cancer, or recent unexplained weight loss.
-
Viral-like prodrome (e.g., fever, nausea, vomiting, fatigue).
-
Neurological manifestations which are atypical for neurodegenerative disorders (e.g., new-onset seizures or seizures refractory to anti-epileptic drugs).
-
Serologic evidence of systemic autoimmunity.
-
Inflammatory cerebrospinal fluid (CSF) findings (pleocytosis, oligoclonal bands/high IgG index) or MRI (gadolinium enhancement).
-
Suggestive neuroimaging abnormalities (see below).
-
Epileptiform activity on electroencephalogram (EEG).
Objective assessment of cognition (e.g., Kokmen short test of mental status) is helpful to determine the severity of the clinical deficit and serves as a baseline from which to judge immunotherapy response with repeat testing [1].
Characteristic clinical syndromes
Limbic encephalitis (LE) is a common clinical manifestation of CNS autoimmunity and can be observed in association with a variety of different autoantibodies (Tables 1 and 2), or in seronegative forms [42]. Anti-NMDAR LE typically presents with rapid development of psychiatric symptoms, for which they are often initially seen by psychiatrists, and may have working memory impairment. A preceding, nonspecific viral-like prodrome (headache, fever, gastrointestinal, and upper respiratory tract symptoms) occurs in 70% of patients. The initial phase is generally followed by seizures, altered mental status, and catatonia which may be followed by central hypoventilation often requiring admission in intensive care units. Hyperkinetic movement disorders (e.g., oro-facial-lingual or limb dyskinesias) and dysautonomia are common accompaniments. In contrast to LE associated with other antibodies, anti-NMDAR LE is frequently seen in young women with an underlying tumor in half of cases (usually ovarian teratoma) [27]. Anti-CASPR2/LGI1 antibodies are also both associated with LE (18% and 43% of cases, respectively), and were formerly identified as anti-voltage-gated potassium channel (VGKC) antibodies [9]. It was later recognized that the clinically relevant antibodies actually bound specific proteins (CASPR2, LGI1) associated with the channel rather than the channel itself, while the clinical relevance of antibodies binding the VGKC but not CASPR2/LGI1 is now uncertain [50]. Facio-brachial dystonic seizures (FBDS) are episodes of dystonic posturing of the face, arm, or both lasting seconds at a time and occurring multiple times per day, and are a hallmark feature of anti-LGI1 autoantibodies [51]. They can mimic paroxysmal dyskinesia [52]. Ictal EEG during FBDS of these patients is frequently normal, possibly due to a deep brain origin of the seizures, and can lead to the erroneous presumption of non-epileptic behavioral spells [9, 51]. When occurring at disease presentation, early treatment of FBDS may prevent cognitive impairment from developing [53••]. Seizures that occur multiple times per day or other paroxysmal events (e.g., paroxysmal dizzy spells) are also common with LGI1 autoantibodies [9]. Peripheral nerve hyper-excitability or Isaac’s syndrome (muscle cramps, fasciculations, and stiffness, occasionally with neuromyotonia) is commonly seen with anti-CASPR2 antibodies, sometimes in association with hyperhidrosis, insomnia, and encephalitis (Morvan’s syndrome) [9]. Importantly, in a large single-center study older age (over 50 years) was able to predict CNS involvement regardless of the antibody subtype (CASPR2 vs LGI1 antibodies) [9]. Diffuse rigidity of central origin is seen with anti-GAD65 and anti-GlyRα1 antibodies and can be accompanied by encephalopathy (progressive encephalopathy with rigidity and myoclonus or stiff-person-syndrome plus) [18, 36]. Status epilepticus is a common initial presentation of anti-GABABR antibodies, while narcolepsy and cataplexy can be an initial manifestation of anti-Ma2 antibodies. The presence of jaw-opening dystonia, opsoclonus myoclonus, and laryngospasm is suggestive of antibodies to ANNA-2/anti-Ri [54].
An insidious clinical presentation over weeks/months is characteristic of anti-DPPX and anti-IgLON5 antibodies. Unexplained weight loss with or without diarrhea is common with anti-DPPX antibodies but dysautonomia and sleep disturbances are also frequently encountered [55]. Sleep disturbances (e.g., insomnia, parasomnias, limb movements, and obstructive sleep apnea) are a hallmark feature of anti-IgLON5 disease, often with bulbar dysfunction and gait/postural instability that may mimic progressive supranuclear palsy [56]. A meningo-encephalo-myelitis, often accompanied by tremor and optic disc edema has been described with CSF anti-GFAP antibodies [38]. These patients typically show excellent response to steroids which is unusual as GFAP is intracellular; further studies are needed to better understand this disorder.
Neuroimaging
Brain MRI with and without gadolinium administration is fundamental to identify abnormalities suggestive of inflammation/autoimmunity, and to exclude structural causes of cognitive impairment. The typical MRI pattern of LE is characterized by unilateral or bilateral T2/fluid-attenuated inversion recovery (FLAIR)-hyperintensity of the mesial temporal lobes, with or without contrast enhancement (Fig. 1a) [42]. This pattern is not specific and may be seen in several other disorders, including epileptic seizures/status epilepticus, gliomas, and herpes simplex virus (HSV) encephalitis [57]. However, a normal MRI is not uncommon in LE [42]. Basal ganglia T2-hyperintensity can be seen with anti-CRMP5 antibodies, typically manifesting clinically as chorea (Fig. 1b), or other antibodies (e.g., anti-NMDAR, anti-Ma2), while T2/T1 hyperintensities may occasionally accompany FBDS associated with anti-LGI1 antibodies [39, 58,59,60]. In contrast to CJD, basal ganglia abnormalities in autoimmune encephalitis typically do not show restricted diffusion on DWI [61]. Diencephalic involvement suggests anti-Ma2 antibodies [39]. Multifocal/extensive white matter involvement and encephalopathy are typically seen with anti-MOG antibodies (Fig. 1c), but may also rarely occur with anti-aquaporin-4 (AQP4) antibodies at disease presentation [62, 63]. Patients with anti-MOG antibody encephalitis may rarely show unilateral or bilateral isolated cortical T2/FLAIR-hyperintensity [64]. Asynchronous, multifocal cortical-subcortical T2/FLAIR-hyperintense lesions in the setting of seizures and cognitive impairment are seen with anti-GABAAR antibodies (Fig. 1d). [13] Progressive cerebral atrophy over time without signal abnormality or enhancement would favor a neurodegenerative etiology over autoimmune dementia; however, a normal MRI or isolated atrophic changes are not uncommon with autoimmune encephalitis and should not dissuade one from neural autoantibody testing in the correct clinical scenario. [65•]
Representative examples of brain MRI abnormalities seen with autoimmune dementia. Axial (a, b, and d) and sagittal (c) fluid-attenuated inversion recovery (FLAIR) images: a Anti-Ma2 limbic encephalitis with mesial temporal signal abnormality (arrow) associated with seminoma; b anti-CRMP5 chorea with caudate and putamen hyperintensity (arrows) associated with small cell lung cancer; c anti-MOG antibody associated ADEM in an adult with multifocal white matter abnormality (arrows) (no cancer detected); d anti-GABA-A antibody autoimmune encephalitis with multifocal cortical and subcortical T2-signal abnormalities (no cancer detected).
Among mimics of autoimmune encephalitis, Wernicke encephalopathy (characterized by T2/FLAIR-hyperintensity of the medial thalami, mammillary bodies, and periaqueductal gray matter) deserves particular attention given the potential for complete reversibility with prompt thiamine administration [66]. Wernicke-like abnormalities with additional striatal involvement occurs in the rare inherited variant due to thiamine transporter impairment [67]. Multifocal areas of restricted diffusion should prompt consideration for intravascular lymphoma, primary CNS vasculitis or cardioembolism [68]. The presence of meningeal enhancement is nonspecific and can be seen in several infectious (e.g., tuberculosis) and inflammatory/autoimmune disorders (e.g., sarcoidosis/CNS vasculitis). Amyloid beta-related angiitis is a recently recognized subtype of CNS vasculitis characterized by micro-hemorrhages, infarcts, and leptomeningeal enhancement on MRI and tends to respond well to immunotherapy. Positive staining for amyloid with vessel wall inflammation confirms the diagnosis and clinicoradiological criteria have also been proposed [69]. Anti-GFAP antibody encephalitis is typically associated with radially oriented, linear perivascular enhancement but a similar pattern can be seen with lymphoma, CNS vasculitis, or neurosarcoidosis [38]. Pachymeningeal enhancement with cranial nerve thickening and orbital pseudotumor are encountered in patients with IgG4-related disease [70].
18F-Fluorodeoxyglucose positron emission tomography (FDG-PET) has a greater sensitivity than MRI for autoimmune encephalitis and may show areas of brain hypometabolism (22%), hypermetabolism (25%), or both (40%) when brain MRI is normal [71]. Abnormal FDG uptake can be regional/syndrome-specific (e.g., mesial temporal lobes, basal ganglia) or diffuse, and generally resolves completely after immunotherapy [71, 72].
CSF analysis
CSF analysis is particularly important in cases of suspected autoimmune dementia since it may reveal inflammatory findings (pleocytosis, oligoclonal bands/high IgG-index) in up to 50% of cases, which are typically absent in neurodegenerative dementias [1]. Pleocytosis (> 5 cells) is extremely rare in patient with neurodegenerative disorders, accounting for < 1% of cases [73]. The overall prevalence of oligoclonal bands among neurodegenerative disorders is 7% [74]. A non-inflammatory CSF does not exclude an autoimmune cause and when present makes the distinction from a neurodegenerative etiology more difficult. Notably, autoimmune encephalitis lacking inflammatory findings on CSF/MRI is not uncommon in the elderly [65•]. CSF levels and ratio of phospho-tau and amyloid-β-42 are useful to diagnose Alzheimer’s disease [75]. CSF real-time quaking-induced conversion (RT-QuIC) is a useful test for prion disorders and has largely superseded the less specific 14-3-3 and neuron-specific enolase [76].
Electroencephalogram
In autoimmune encephalitis, EEG generally shows nonspecific diffuse or focal slowing or epileptiform activity, the latter helping to distinguish from neurodegenerative etiologies in which such a finding is less frequent. Isolated temporal epileptiform abnormalities are common but cannot be distinguished from those of other etiologies (e.g., HSV encephalitis). A pattern of extreme delta brush (rhythmic delta activity at 1–3 Hz with superimposed rhythmic beta bursts at 20–30 Hz) can be found in one third of patients with anti-NMDAR encephalitis [77]. Although suggestive, this pattern is non-specific of anti-NMDAR encephalitis and can be observed in other conditions, including non-autoimmune mesial temporal lobe epilepsy, hypoxic ischemic encephalopathy, and brain tumors [78].
Neural antibody testing
Neural autoantibody detection in the correct clinical setting may confirm the diagnosis of autoimmune dementia. Testing prior to immunotherapy is essential as empiric treatments can impact antibody test results. For example, testing after plasma exchange could result in a false negative, while testing after intravenous immunoglobulins (IVIg) could result in a false positive (due to passive transfer of common autoantibodies [e.g. anti-GAD] with IVIg administration). It is important to recognize that some antibodies are better detected in serum (e.g., anti-LGI1) and others in CSF (e.g., anti-NMDAR) [42]. As many of the clinical syndromes (e.g., LE) can be encountered with a wide variety of antibodies, testing a profile of antibodies is often preferable than individual antibody testing; furthermore, when more than one antibody is positive, the profile can guide cancer search [79]. An initial screening by tissue-based immunofluorescence or immunohistochemistry may show characteristic staining patterns which can guide confirmatory testing, or reveal the presence of unclassified antibodies selectively staining the neural tissue that may help support CNS autoimmunity when testing of known autoantibodies is negative [80]. In certain scenarios (FBDS and anti-LGI1), a single antibody testing may be sufficient although > 1 autoantibodies may sometimes coexist [81]. Refinement in antibody assay techniques and target identification has significantly reduced the risk of false positive results. Older generation techniques (e.g., radioimmunoassays, ELISA) assay have a higher risk of positivity in healthy controls (up to 6%) when compared to newer generation techniques with cell-based assays where the risk of false positives is much less (0.2%) [82]. The titer may be useful in predicting the likelihood of relevance to neurologic disease. For example, anti-GAD65 antibodies are found in up to 8% of the general population at low level, while with CNS autoimmunity, the titer is usually extremely high and CSF detection is often evident. As such, a positive autoantibody result should not replace clinical judgment and indiscriminate testing outside of the correct clinical context significantly reduces the positive predictive value of the test [83]. Most clinically relevant antibodies in autoimmune dementia detected are of the IgG subtype and the utility of IgM/IgA antibodies as a marker of CNS autoimmunity is unproven.
Systemic autoantibodies (e.g., anti-thyroid peroxidase [TPO] antibodies) have been historically associated with encephalopathies with a variety of terms (e.g., Hashimoto’s encephalopathy) and they are frequently detected in patients with CNS autoimmunity [5]. However, care is needed when such antibodies are detected in a patient with cognitive impairment as they are common in the general population (e.g., TPO and thyroglobulin antibodies are found in up to 20–30% of the elderly population). Instead, their presence should be regarded as a marker of general predisposition to autoimmunity and not preclude searching for concomitant CNS-specific autoantibodies or considering alternative non-autoimmune etiologies of cognitive impairment.
Additional diagnostic workup
An extensive search for other potential causes of cognitive impairment is mandatory and includes initial serum/CSF laboratory investigations for toxic/metabolic (complete blood count, kidney, thyroid and liver function, electrolytes, vitamins, drugs/medications, metals), infectious (markers or cultures for bacterial, fungal, parasitic and viral infections), neoplastic (cytology, flow cytometry), and other systemic immune-mediated disorders [5]. Exclusion of CNS infections must be prioritized since immunosuppression might have devastating consequences in such patients. Clinical/MRI findings, geographic area, personal history (e.g., high-risk sexual behavior, intravenous drug use, recent history of travel to endemic areas), and clinical setting (e.g., patients receiving immunosuppression) should guide testing for specific infectious agents, including HIV and syphilis. Brain biopsy can be considered in selected cases, and is more useful to exclude alternative etiologies (e.g., CNS neoplasms) than confirm an autoimmune etiology, but the presence of inflammation can support ongoing use of immunotherapy.
Cancer search
The search for cancer can be guided by age, sex, risk factors (history of smoking), antibody detected, and genetics. In anti-LGI1 autoantibodies, the absence of genetic markers of autoimmune predisposition (HLA-DR7 or HLA-DRB4) may predict a higher risk of underlying tumor [84, 85]. Whole-body CT is often the initial test used due to widespread availability and lower cost, but FDG-PET has a greater sensitivity [86]. Sex-specific tests should not be overlooked (e.g., testicular ultrasound in males with anti-Ma2 antibodies) as localized cancers may not be visible with other testing. In those with antibodies strongly associated with cancer (e.g., ANNA-1/anti-Hu) but initially negative cancer screening, periodic surveillance (e.g., every 6–12 months) should be considered as sometimes the cancer may manifest on subsequent evaluations [87].
Treatment and prognosis
In case of uncertainty, an immunotherapy trial with first-line acute therapies (see below) might support an autoimmune etiology if there is a response [88]. However, other inflammatory (e.g., sarcoidosis, multiple sclerosis) and non-inflammatory (e.g., CNS lymphoma) disorders may respond to corticosteroids and steroid response alone is not sufficient for autoimmune encephalitis/dementia diagnosis.
The natural clinical course of autoimmune encephalitis is not completely understood and varies according to the underlying antibody. Relapses can occur, while certain antibodies are sometimes associated with a steroid-dependent course (e.g., anti-GFAP, anti-MOG) [62, 89]. Cases of untreated patients with autoimmune encephalitis and spontaneous improvement over months after the acute phase have been reported [90, 91]. However, early initiation of immunotherapy and tumor treatment (in paraneoplastic forms) seems critical to reduce long-term disability and prevent relapses [28, 53]. The overall relapse-rate in patients with anti-NMDAR encephalitis at 2 years is 12%, with one third of those patients having multiple relapses [28]. Clinical worsening during immunotherapy should be carefully evaluated since it may be related to opportunistic infection or neoplasm occurrence. In autoimmune encephalitis, relapses tend to be milder and similar to the initial attack [28]. Residual cognitive impairment from structural brain damage is one of the greatest contributors to long-term disability [46, 92,93,94,95]. Decreased quality of life and school performances (often with need of special assistance) are common following anti-NMDAR encephalitis in children [96].
There are no randomized controlled trials on treatment of CNS autoimmunity and recommendations derive from clinical experience and retrospective/prospective series. In general, antibody/B cell-depleting agents are preferred with antibodies targeting cell-surface antigens while T cell-depleting drugs are preferred with antibodies targeting intracellular antigens. Two scores (antibody prevalence in epilepsy and encephalopathy [APE2], and response to immunotherapy in epilepsy and encephalopathy [RITE2]), have recently been shown to be highly accurate in predicting the presence of CNS autoantibodies and immunotherapy response in patients with suspected autoimmune dementia [97].
Acute therapy
First-line immunotherapies with commonly used dosages include:
-
Intravenous methylprednisone (IVMP), 1 g/day for 5 days and possibly weekly for 6–12 weeks.
-
IVIg, 0.4 g/kg/day for 5 days and then weekly for 6–12 weeks.
-
Plasma exchange (PLEX), 1 exchange every other day for 5–7 exchanges.
IVIg and PLEX may be considered as an alternative to IVMP (e.g., diabetic patients), or as an add-on therapy in severe cases [28]. Improvement in cognition was documented in 64% of patients with suspected autoimmune dementia seen at Mayo Clinic after first-line treatment, mostly within the first week [1]. The speed of response may depend on the antibody type. For example, with anti-LGI1 antibodies, a rapid response to immunotherapy is typical with FBDS often resolving within days and a return to normal functioning that may occur within few weeks [53]. A prolonged high-dose oral prednisone often helps to prevent relapses [58]. In a study of anti-NMDAR encephalitis patients, improvement at 4 weeks was observed in 53% of patients after first-line immunotherapies (either alone or in combination), but a return to normal functioning generally takes months. For this reason, second-line immunotherapies (e.g., rituximab, cyclophosphamide) are often administered early with anti-NMDAR encephalitis to help resolve the acute syndrome and then as maintenance therapy to prevent relapse. In the same study, 57% of non-responders received a second line treatment resulting in a better final outcome [28]. The decision to use a second-line agent should be carefully evaluated based on the type of antibody detected (e.g., cell-surface vs intracellular, existing evidence in the literature for each antibody), timing of therapy administration (i.e., patients not treated acutely are less likely to benefit from a late intervention), and the degree of diagnostic certainty (i.e., failure to respond to steroids should always prompt considering another etiology, especially in seronegative cases).
Maintenance therapy
Several drugs might be used based on patient characteristics, disease severity, time taken for drug to become effective, and associated antibody. Azathioprine (2–3 mg/kg/day orally), mycophenolate (500–1000 mg twice/day orally), rituximab (intravenously), and cyclophosphamide (orally or intravenously) are commonly utilized based on our experience in rheumatologic and other autoimmune neurological disorders (e.g., myasthenia gravis) [5]. While azathioprine and mycophenolate require up to 6 months to become effective with slow oral steroids tapering concurrently (e.g., prednisone 20–60 mg/day), rituximab is frequently preferred for the high tolerability and shorter time needed to become effective (4–5 weeks). Thiopurine methyltransferase activity is required prior to azathioprine as reduced or no activity can increase the risk of side effects and may warrant consideration of a lower dose or alternative medication.
Rituximab is an anti CD20 monoclonal antibody that depletes B cells (mostly naive and mature B cells) and is generally administered in one of the following treatment regimens: (1) two 1000 mg infusions separated by 2 weeks; (2) 375 mg/m2/week for 4 weeks. These same dosages are then repeated every 6 months. Serum CD19 (a pan B cell lineage marker) count monitoring with a target of zero might be an alternative approach to guide the frequency of reinfusions [98]. Rituximab is potentially indicated in any antibody-mediated form, although one study on autoimmune LE showed improvement regardless of the antibody status (cell-surface, intracellular, or unknown) [99].
Cyclophosphamide depletes both B and T cells and carries significant side effects including alopecia, blood cytopenias, hemorrhagic cystitis, and infertility. It is generally considered after rituximab failure or for treatment of refractory forms, especially with antibodies targeting intracellular antigens. Administration is generally intravenous with abundant hydration to prevent nephrotoxicity (500–1000 mg/m2/month for 6–12 months).
Other potential treatments include tocilizumab, bortezomib, and low-dose IL-2 [100,101,102]. These immunosuppressants carry an increased risk of infection and rare cases of progressive multifocal leukoencephalopathy have been reported [103]. Prophylaxis against varicella zoster virus (VZV) with oral acyclovir is important with Bortezomib as disseminated infection including VZV vasculopathy has been reported [104]. A full discussion of the precautions/side effects needed with immunotherapy has been reported previously [5].
Emerging clinical settings
It is now recognized that autoimmune encephalitis can follow HSV encephalitis and recent findings from a prospective study showed 27% of patients with HSV encephalitis will eventually develop an autoimmune encephalitis (mostly anti-NMDAR), and usually within 2 months of the infection [105••]. Since these patients are generally responsive to immunotherapy, CNS autoimmunity must be considered in any case of neurological worsening after initial antiviral treatment of HSV encephalitis.
Immune checkpoint inhibitors (ICI) are a family of monoclonal antibodies targeting immune checkpoint proteins (e.g., PD-1/PDL-1, CTLA-4) that are essential for immune system regulation. In oncological practice, inhibition of these proteins by ICI induces a massive immune response which is highly effective against tumors, but may sometimes result in autoimmunity. Neurological autoimmunity has been reported in < 5% of patients treated with ICI but given the increasing use of these therapies in different types of cancer, the number of patients impacted is likely to increase [106]. Cognitive impairment and encephalopathy may occur, generally without detectable neural autoantibodies [107]. Treatment options in these patients are based on expert opinion and include steroids, PLEX, and rituximab. The risk/benefit of ICI discontinuation needs to be carefully weighed as these medications may prolong cancer survival.
Autoimmune encephalitis accompanying antibodies to AMPA, LGI1, MOG, and NMDA have been reported in patients undergoing lymphocyte-depleting immunosuppressants for solid-organ or hematopoietic cell transplantation. Some of these patients were CSF Epstein Barr Virus PCR positive suggesting a possible viral trigger. These rarely reported cases suggest such patients may benefit from additional antibody depleting treatments (e.g., PLEX, rituximab) [108,109,110]. Human Herpes Virus type 6 (HHV6) encephalitis may mimic autoimmune LE in post-transplant patients and evaluation for HHV6 CSF PCR should also be considered in addition to neural autoantibody testing [111]. Future research will help to clarify the optimal treatment strategies for the common and uncommon clinical settings in which CNS autoimmunity may arise.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Flanagan EP, McKeon A, Lennon VA, Boeve BF, Trenerry MR, Tan KM, et al. Autoimmune dementia: clinical course and predictors of immunotherapy response. Mayo Clin Proc. 2010;85(10):881–97. https://doi.org/10.4065/mcp.2010.0326.
Geschwind MD, Tan KM, Lennon VA, Barajas RF Jr, Haman A, Klein CJ, et al. Voltage-gated potassium channel autoimmunity mimicking creutzfeldt-jakob disease. Arch Neurol. 2008;65(10):1341–6. https://doi.org/10.1001/archneur.65.10.1341.
McKeon A, Marnane M, O'Connell M, Stack JP, Kelly PJ, Lynch T. Potassium channel antibody associated encephalopathy presenting with a frontotemporal dementia like syndrome. Arch Neurol. 2007;64(10):1528–30. https://doi.org/10.1001/archneur.64.10.1528.
Mariotto S, Tamburin S, Salviati A, Ferrari S, Zoccarato M, Giometto B, et al. Anti-N-methyl-d-aspartate receptor encephalitis causing a prolonged depressive disorder evolving to inflammatory brain disease. Case Rep Neurol. 2014;6(1):38–43. https://doi.org/10.1159/000358820.
Flanagan EP, Drubach DA, Boeve BF. Autoimmune dementia and encephalopathy. Handb Clin Neurol. 2016;133:247–67. https://doi.org/10.1016/B978-0-444-63432-0.00014-1.
Pittock SJ, Vincent A. Introduction to autoimmune neurology. Handb Clin Neurol. 2016;133:3–14. https://doi.org/10.1016/B978-0-444-63432-0.00001-3.
Hoftberger R, van Sonderen A, Leypoldt F, Houghton D, Geschwind M, Gelfand J, et al. Encephalitis and AMPA receptor antibodies: novel findings in a case series of 22 patients. Neurology. 2015;84(24):2403–12. https://doi.org/10.1212/WNL.0000000000001682.
Lai M, Hughes EG, Peng X, Zhou L, Gleichman AJ, Shu H, et al. AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann Neurol. 2009;65(4):424–34. https://doi.org/10.1002/ana.21589.
Gadoth A, Pittock SJ, Dubey D, McKeon A, Britton JW, Schmeling JE, et al. Expanded phenotypes and outcomes among 256 LGI1/CASPR2-IgG-positive patients. Ann Neurol. 2017;82(1):79–92. https://doi.org/10.1002/ana.24979.
van Sonderen A, Arino H, Petit-Pedrol M, Leypoldt F, Kortvelyessy P, Wandinger KP, et al. The clinical spectrum of Caspr2 antibody-associated disease. Neurology. 2016;87(5):521–8. https://doi.org/10.1212/WNL.0000000000002917.
Hara M, Arino H, Petit-Pedrol M, Sabater L, Titulaer MJ, Martinez-Hernandez E, et al. DPPX antibody-associated encephalitis: Main syndrome and antibody effects. Neurology. 2017;88(14):1340–8. https://doi.org/10.1212/WNL.0000000000003796.
Tobin WO, Lennon VA, Komorowski L, Probst C, Clardy SL, Aksamit AJ, et al. DPPX potassium channel antibody: frequency, clinical accompaniments, and outcomes in 20 patients. Neurology. 2014;83(20):1797–803. https://doi.org/10.1212/WNL.0000000000000991.
Spatola M, Petit-Pedrol M, Simabukuro MM, Armangue T, Castro FJ, Barcelo Artigues MI, et al. Investigations in GABAA receptor antibody-associated encephalitis. Neurology. 2017;88(11):1012–20. https://doi.org/10.1212/WNL.0000000000003713.
Hoftberger R, Titulaer MJ, Sabater L, Dome B, Rozsas A, Hegedus B, et al. Encephalitis and GABAB receptor antibodies: novel findings in a new case series of 20 patients. Neurology. 2013;81(17):1500–6. https://doi.org/10.1212/WNL.0b013e3182a9585f.
Jeffery OJ, Lennon VA, Pittock SJ, Gregory JK, Britton JW, McKeon A. GABAB receptor autoantibody frequency in service serologic evaluation. Neurology. 2013;81(10):882–7. https://doi.org/10.1212/WNL.0b013e3182a35271.
Lancaster E, Lai M, Peng X, Hughes E, Constantinescu R, Raizer J, et al. Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol. 2010;9(1):67–76. https://doi.org/10.1016/S1474-4422(09)70324-2.
Spatola M, Sabater L, Planaguma J, Martinez-Hernandez E, Armangue T, Pruss H, et al. Encephalitis with mGluR5 antibodies: symptoms and antibody effects. Neurology. 2018;90(22):e1964–e72. https://doi.org/10.1212/WNL.0000000000005614.
McKeon A, Martinez-Hernandez E, Lancaster E, Matsumoto JY, Harvey RJ, McEvoy KM, et al. Glycine receptor autoimmune spectrum with stiff-man syndrome phenotype. JAMA Neurol. 2013;70(1):44–50. https://doi.org/10.1001/jamaneurol.2013.574.
Carvajal-Gonzalez A, Leite MI, Waters P, Woodhall M, Coutinho E, Balint B, et al. Glycine receptor antibodies in PERM and related syndromes: characteristics, clinical features and outcomes. Brain. 2014;137(Pt 8):2178–92. https://doi.org/10.1093/brain/awu142.
Gaig C, Graus F, Compta Y, Hogl B, Bataller L, Bruggemann N, et al. Clinical manifestations of the anti-IgLON5 disease. Neurology. 2017;88(18):1736–43. https://doi.org/10.1212/WNL.0000000000003887.
Honorat JA, Komorowski L, Josephs KA, Fechner K, St Louis EK, Hinson SR, et al. IgLON5 antibody: neurological accompaniments and outcomes in 20 patients. Neurol Neuroimmunol Neuroinflamm. 2017;4(5):e385. https://doi.org/10.1212/NXI.0000000000000385.
van Sonderen A, Thijs RD, Coenders EC, Jiskoot LC, Sanchez E, de Bruijn MA, et al. Anti-LGI1 encephalitis: clinical syndrome and long-term follow-up. Neurology. 2016;87(14):1449–56. https://doi.org/10.1212/WNL.0000000000003173.
Cobo-Calvo A, Ruiz A, Maillart E, Audoin B, Zephir H, Bourre B, et al. Clinical spectrum and prognostic value of CNS MOG autoimmunity in adults: the MOGADOR study. Neurology. 2018;90(21):e1858–e69. https://doi.org/10.1212/WNL.0000000000005560.
Jurynczyk M, Messina S, Woodhall MR, Raza N, Everett R, Roca-Fernandez A, et al. Clinical presentation and prognosis in MOG-antibody disease: a UK study. Brain. 2017;140(12):3128–38. https://doi.org/10.1093/brain/awx276.
Ramanathan S, Mohammad S, Tantsis E, Nguyen TK, Merheb V, Fung VSC, et al. Clinical course, therapeutic responses and outcomes in relapsing MOG antibody-associated demyelination. J Neurol Neurosurg Psychiatry. 2018;89(2):127–37. https://doi.org/10.1136/jnnp-2017-316880.
Gresa-Arribas N, Planaguma J, Petit-Pedrol M, Kawachi I, Katada S, Glaser CA, et al. Human neurexin-3alpha antibodies associate with encephalitis and alter synapse development. Neurology. 2016;86(24):2235–42. https://doi.org/10.1212/WNL.0000000000002775.
Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Lai M, et al. Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol. 2008;7(12):1091–8. https://doi.org/10.1016/S1474-4422(08)70224-2.
Titulaer MJ, McCracken L, Gabilondo I, Armangue T, Glaser C, Iizuka T, et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 2013;12(2):157–65. https://doi.org/10.1016/S1474-4422(12)70310-1.
Do LD, Chanson E, Desestret V, Joubert B, Ducray F, Brugiere S, et al. Characteristics in limbic encephalitis with anti-adenylate kinase 5 autoantibodies. Neurology. 2017;88(6):514–24. https://doi.org/10.1212/WNL.0000000000003586.
Pittock SJ, Lucchinetti CF, Parisi JE, Benarroch EE, Mokri B, Stephan CL, et al. Amphiphysin autoimmunity: paraneoplastic accompaniments. Ann Neurol. 2005;58(1):96–107. https://doi.org/10.1002/ana.20529.
Dalmau J, Graus F, Rosenblum MK, Posner JB. Anti-Hu--associated paraneoplastic encephalomyelitis/sensory neuronopathy. A clinical study of 71 patients. Medicine (Baltimore). 1992;71(2):59–72.
Lucchinetti CF, Kimmel DW, Lennon VA. Paraneoplastic and oncologic profiles of patients seropositive for type 1 antineuronal nuclear autoantibodies. Neurology. 1998;50(3):652–7.
Pittock SJ, Lucchinetti CF, Lennon VA. Anti-neuronal nuclear autoantibody type 2: paraneoplastic accompaniments. Ann Neurol. 2003;53(5):580–7. https://doi.org/10.1002/ana.10518.
Honnorat J, Cartalat-Carel S, Ricard D, Camdessanche JP, Carpentier AF, Rogemond V, et al. Onco-neural antibodies and tumour type determine survival and neurological symptoms in paraneoplastic neurological syndromes with Hu or CV2/CRMP5 antibodies. J Neurol Neurosurg Psychiatry. 2009;80(4):412–6. https://doi.org/10.1136/jnnp.2007.138016.
Yu Z, Kryzer TJ, Griesmann GE, Kim K, Benarroch EE, Lennon VA. CRMP-5 neuronal autoantibody: marker of lung cancer and thymoma-related autoimmunity. Ann Neurol. 2001;49(2):146–54.
Pittock SJ, Yoshikawa H, Ahlskog JE, Tisch SH, Benarroch EE, Kryzer TJ, et al. Glutamic acid decarboxylase autoimmunity with brainstem, extrapyramidal, and spinal cord dysfunction. Mayo Clin Proc. 2006;81(9):1207–14. https://doi.org/10.4065/81.9.1207.
Saiz A, Blanco Y, Sabater L, Gonzalez F, Bataller L, Casamitjana R, et al. Spectrum of neurological syndromes associated with glutamic acid decarboxylase antibodies: diagnostic clues for this association. Brain. 2008;131(Pt 10):2553–63. https://doi.org/10.1093/brain/awn183.
Flanagan EP, Hinson SR, Lennon VA, Fang B, Aksamit AJ, Morris PP, et al. Glial fibrillary acidic protein immunoglobulin G as biomarker of autoimmune astrocytopathy: analysis of 102 patients. Ann Neurol. 2017;81(2):298–309. https://doi.org/10.1002/ana.24881.
Dalmau J, Graus F, Villarejo A, Posner JB, Blumenthal D, Thiessen B, et al. Clinical analysis of anti-Ma2-associated encephalitis. Brain. 2004;127(Pt 8):1831–44. https://doi.org/10.1093/brain/awh203.
Basal E, Zalewski N, Kryzer TJ, Hinson SR, Guo Y, Dubey D, et al. Paraneoplastic neuronal intermediate filament autoimmunity. Neurology. 2018;91(18):e1677–e89. https://doi.org/10.1212/WNL.0000000000006435.
Vernino S, Lennon VA. New Purkinje cell antibody (PCA-2): marker of lung cancer-related neurological autoimmunity. Ann Neurol. 2000;47(3):297–305.
Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15(4):391–404. https://doi.org/10.1016/S1474-4422(15)00401-9 This study proposes diagnostic criteria to facilitate classification and early diagnosis and treatment of autoimmune encephalitis.
Dubey D, Pittock SJ, Kelly CR, McKeon A, Lopez-Chiriboga AS, Lennon VA, et al. Autoimmune encephalitis epidemiology and a comparison to infectious encephalitis. Ann Neurol. 2018;83(1):166–77. https://doi.org/10.1002/ana.25131 This study is the first to show the population-based incidence and prevalence of autoimmune encephalitis and shows that it rivals that of infectious encephalitis.
Gable MS, Sheriff H, Dalmau J, Tilley DH, Glaser CA. The frequency of autoimmune N-methyl-D-aspartate receptor encephalitis surpasses that of individual viral etiologies in young individuals enrolled in the California Encephalitis Project. Clin Infect Dis. 2012;54(7):899–904. https://doi.org/10.1093/cid/cir1038.
Granerod J, Ambrose HE, Davies NW, Clewley JP, Walsh AL, Morgan D, et al. Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis. 2010;10(12):835–44. https://doi.org/10.1016/S1473-3099(10)70222-X.
Arino H, Armangue T, Petit-Pedrol M, Sabater L, Martinez-Hernandez E, Hara M, et al. Anti-LGI1-associated cognitive impairment: presentation and long-term outcome. Neurology. 2016;87(8):759–65. https://doi.org/10.1212/WNL.0000000000003009.
Marquetand J, van Lessen M, Bender B, Reimold M, Elsen G, Stoecker W, et al. Slowly progressive LGI1 encephalitis with isolated late-onset cognitive dysfunction: a treatable mimic of Alzheimer’s disease. Eur J Neurol. 2016;23(5):e28–9. https://doi.org/10.1111/ene.12939.
Baumgartner A, Rauer S, Hottenrott T, Leypoldt F, Ufer F, Hegen H, et al. Admission diagnoses of patients later diagnosed with autoimmune encephalitis. J Neurol. 2018. https://doi.org/10.1007/s00415-018-9105-3.
Lopez-Chiriboga AS, Flanagan EP. Diagnostic and therapeutic approach to autoimmune neurologic disorders. Semin Neurol. 2018;38(3):392–402. https://doi.org/10.1055/s-0038-1660819.
van Sonderen A, Schreurs MW, de Bruijn MA, Boukhrissi S, Nagtzaam MM, Hulsenboom ES, et al. The relevance of VGKC positivity in the absence of LGI1 and Caspr2 antibodies. Neurology. 2016;86(18):1692–9. https://doi.org/10.1212/WNL.0000000000002637.
Irani SR, Michell AW, Lang B, Pettingill P, Waters P, Johnson MR, et al. Faciobrachial dystonic seizures precede Lgi1 antibody limbic encephalitis. Ann Neurol. 2011;69(5):892–900. https://doi.org/10.1002/ana.22307.
Aradillas E, Schwartzman RJ. Kinesigenic dyskinesia in a case of voltage-gated potassium channel-complex protein antibody encephalitis. Arch Neurol. 2011;68(4):529–32. https://doi.org/10.1001/archneurol.2010.317.
Thompson J, Bi M, Murchison AG, Makuch M, Bien CG, Chu K, et al. The importance of early immunotherapy in patients with faciobrachial dystonic seizures. Brain. 2018;141(2):348–56. https://doi.org/10.1093/brain/awx323 This study highlights the importance of early recognition and treatment of anti-LGI1 autoimmunity to prevent long-term cognitive impairment and disability.
Pittock SJ, Parisi JE, McKeon A, Roemer SF, Lucchinetti CF, Tan KM, et al. Paraneoplastic jaw dystonia and laryngospasm with antineuronal nuclear autoantibody type 2 (anti-Ri). Arch Neurol. 2010;67(9):1109–15. https://doi.org/10.1001/archneurol.2010.209.
Boronat A, Gelfand JM, Gresa-Arribas N, Jeong HY, Walsh M, Roberts K, et al. Encephalitis and antibodies to dipeptidyl-peptidase-like protein-6, a subunit of Kv4.2 potassium channels. Ann Neurol. 2013;73(1):120–8. https://doi.org/10.1002/ana.23756.
Sabater L, Gaig C, Gelpi E, Bataller L, Lewerenz J, Torres-Vega E, et al. A novel non-rapid-eye movement and rapid-eye-movement parasomnia with sleep breathing disorder associated with antibodies to IgLON5: a case series, characterisation of the antigen, and post-mortem study. Lancet Neurol. 2014;13(6):575–86. https://doi.org/10.1016/S1474-4422(14)70051-1.
Oyanguren B, Sanchez V, Gonzalez FJ, de Felipe A, Esteban L, Lopez-Sendon JL, et al. Limbic encephalitis: a clinical-radiological comparison between herpetic and autoimmune etiologies. Eur J Neurol. 2013;20(12):1566–70. https://doi.org/10.1111/ene.12249.
Flanagan EP, Kotsenas AL, Britton JW, McKeon A, Watson RE, Klein CJ, et al. Basal ganglia T1 hyperintensity in LGI1-autoantibody faciobrachial dystonic seizures. Neurol Neuroimmunol Neuroinflamm. 2015;2(6):e161. https://doi.org/10.1212/NXI.0000000000000161.
Kinirons P, Fulton A, Keoghan M, Brennan P, Farrell MA, Moroney JT. Paraneoplastic limbic encephalitis (PLE) and chorea associated with CRMP-5 neuronal antibody. Neurology. 2003;61(11):1623–4.
Zhang T, Duan Y, Ye J, Xu W, Shu N, Wang C, et al. Brain MRI characteristics of patients with anti-N-methyl-D-aspartate receptor encephalitis and their associations with 2-year clinical outcome. AJNR Am J Neuroradiol. 2018;39(5):824–9. https://doi.org/10.3174/ajnr.A5593.
Vitali P, Maccagnano E, Caverzasi E, Henry RG, Haman A, Torres-Chae C, et al. Diffusion-weighted MRI hyperintensity patterns differentiate CJD from other rapid dementias. Neurology. 2011;76(20):1711–9. https://doi.org/10.1212/WNL.0b013e31821a4439.
Lopez-Chiriboga AS, Majed M, Fryer J, Dubey D, McKeon A, Flanagan EP, et al. Association of MOG-IgG serostatus with relapse after acute disseminated encephalomyelitis and proposed diagnostic criteria for MOG-IgG-associated disorders. JAMA Neurol. 2018;75(11):1355–63. https://doi.org/10.1001/jamaneurol.2018.1814.
Sechi E, Addis A, Batzu L, Mariotto S, Ferrari S, Conti M, et al. Late presentation of NMOSD as rapidly progressive leukoencephalopathy with atypical clinical and radiological findings. Mult Scler. 2018;24(5):685–8. https://doi.org/10.1177/1352458517721661.
Ogawa R, Nakashima I, Takahashi T, Kaneko K, Akaishi T, Takai Y, et al. MOG antibody-positive, benign, unilateral, cerebral cortical encephalitis with epilepsy. Neurol Neuroimmunol Neuroinflamm. 2017;4(2):e322. https://doi.org/10.1212/NXI.0000000000000322.
Escudero D, Guasp M, Arino H, Gaig C, Martinez-Hernandez E, Dalmau J, et al. Antibody-associated CNS syndromes without signs of inflammation in the elderly. Neurology. 2017;89(14):1471–5. https://doi.org/10.1212/WNL.0000000000004541 This study highlights the importance of recognizing characteristic clinical syndromes associted with CNS autoimmunity as paraclinical findings suggestive of inflammation may be lacking, especially in the elderly.
Sechi G, Sechi E, Fois C, Kumar N. Advances in clinical determinants and neurological manifestations of B vitamin deficiency in adults. Nutr Rev. 2016;74(5):281–300. https://doi.org/10.1093/nutrit/nuv107.
Sechi E, Addis A, Fadda G, Minafra L, Bravata V, Sechi G. Teaching NeuroImages: subacute encephalopathy in a young woman with THTR2 gene mutation. Neurology. 2015;85(14):e108–9. https://doi.org/10.1212/WNL.0000000000002002.
Lopez-Chiriboga AS, Yoon JW, Siegel JL, Harriott AM, Pirris S, Eidelman BH, et al. Granulomatous angiitis of the central nervous system associated with Hodgkin's lymphoma: case report and literature review. J Stroke Cerebrovasc Dis. 2018;27(1):e5–8. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.08.029.
Auriel E, Charidimou A, Gurol ME, Ni J, Van Etten ES, Martinez-Ramirez S, et al. Validation of clinicoradiological criteria for the diagnosis of cerebral amyloid angiopathy-related inflammation. JAMA Neurol. 2016;73(2):197–202. https://doi.org/10.1001/jamaneurol.2015.4078.
Flanagan EP, Chowdhary VR, McCarthy JT, Smyrk TC, Chari ST, Kumar N. IgG4-related (neurologic) disease: diagnostic challenges, clinical clues and expanding spectrum. Int J Rheum Dis. 2015;18(7):807–9. https://doi.org/10.1111/1756-185X.12465.
Probasco JC, Solnes L, Nalluri A, Cohen J, Jones KM, Zan E, et al. Abnormal brain metabolism on FDG-PET/CT is a common early finding in autoimmune encephalitis. Neurol Neuroimmunol Neuroinflamm. 2017;4(4):e352. https://doi.org/10.1212/NXI.0000000000000352.
Rey C, Koric L, Guedj E, Felician O, Kaphan E, Boucraut J, et al. Striatal hypermetabolism in limbic encephalitis. J Neurol. 2012;259(6):1106–10. https://doi.org/10.1007/s00415-011-6308-2.
Jesse S, Brettschneider J, Sussmuth SD, Landwehrmeyer BG, von Arnim CA, Ludolph AC, et al. Summary of cerebrospinal fluid routine parameters in neurodegenerative diseases. J Neurol. 2011;258(6):1034–41. https://doi.org/10.1007/s00415-010-5876-x.
Janssen JC, Godbolt AK, Ioannidis P, Thompson EJ, Rossor MN. The prevalence of oligoclonal bands in the CSF of patients with primary neurodegenerative dementia. J Neurol. 2004;251(2):184–8. https://doi.org/10.1007/s00415-004-0296-4.
Maddalena A, Papassotiropoulos A, Muller-Tillmanns B, Jung HH, Hegi T, Nitsch RM, et al. Biochemical diagnosis of Alzheimer disease by measuring the cerebrospinal fluid ratio of phosphorylated tau protein to beta-amyloid peptide42. Arch Neurol. 2003;60(9):1202–6. https://doi.org/10.1001/archneur.60.9.1202.
McGuire LI, Peden AH, Orru CD, Wilham JM, Appleford NE, Mallinson G, et al. Real time quaking-induced conversion analysis of cerebrospinal fluid in sporadic Creutzfeldt-Jakob disease. Ann Neurol. 2012;72(2):278–85. https://doi.org/10.1002/ana.23589.
Schmitt SE, Pargeon K, Frechette ES, Hirsch LJ, Dalmau J, Friedman D. Extreme delta brush: a unique EEG pattern in adults with anti-NMDA receptor encephalitis. Neurology. 2012;79(11):1094–100. https://doi.org/10.1212/WNL.0b013e3182698cd8.
Baykan B, Gungor Tuncer O, Vanli-Yavuz EN, Baysal Kirac L, Gundogdu G, Bebek N, et al. Delta brush pattern is not unique to NMDAR encephalitis: evaluation of two independent long-term EEG cohorts. Clin EEG Neurosci. 2018;49(4):278–84. https://doi.org/10.1177/1550059417693168.
Horta ES, Lennon VA, Lachance DH, Jenkins SM, Smith CY, McKeon A, et al. Neural autoantibody clusters aid diagnosis of cancer. Clin Cancer Res. 2014;20(14):3862–9. https://doi.org/10.1158/1078-0432.CCR-14-0652.
Hoftberger R, Sabater L, Marignier R, Aboul-Enein F, Bernard-Valnet R, Rauschka H, et al. An optimized immunohistochemistry technique improves NMO-IgG detection: study comparison with cell-based assays. PLoS One. 2013;8(11):e79083. https://doi.org/10.1371/journal.pone.0079083.
Titulaer MJ, Hoftberger R, Iizuka T, Leypoldt F, McCracken L, Cellucci T, et al. Overlapping demyelinating syndromes and anti-N-methyl-D-aspartate receptor encephalitis. Ann Neurol. 2014;75(3):411–28. https://doi.org/10.1002/ana.24117.
Lang K, Pruss H. Frequencies of neuronal autoantibodies in healthy controls: estimation of disease specificity. Neurol Neuroimmunol Neuroinflamm. 2017;4(5):e386. https://doi.org/10.1212/NXI.0000000000000386.
Jarius S, Paul F, Aktas O, Asgari N, Dale RC, de Seze J, et al. MOG encephalomyelitis: international recommendations on diagnosis and antibody testing. J Neuroinflammation. 2018;15(1):134. https://doi.org/10.1186/s12974-018-1144-2.
McCracken L, Zhang J, Greene M, Crivaro A, Gonzalez J, Kamoun M, et al. Improving the antibody-based evaluation of autoimmune encephalitis. Neurol Neuroimmunol Neuroinflamm. 2017;4(6):e404. https://doi.org/10.1212/NXI.0000000000000404.
van Sonderen A, Roelen DL, Stoop JA, Verdijk RM, Haasnoot GW, Thijs RD, et al. Anti-LGI1 encephalitis is strongly associated with HLA-DR7 and HLA-DRB4. Ann Neurol. 2017;81(2):193–8. https://doi.org/10.1002/ana.24858.
McKeon A, Apiwattanakul M, Lachance DH, Lennon VA, Mandrekar JN, Boeve BF, et al. Positron emission tomography-computed tomography in paraneoplastic neurologic disorders: systematic analysis and review. Arch Neurol. 2010;67(3):322–9. https://doi.org/10.1001/archneurol.2009.336.
Pittock SJ, Kryzer TJ, Lennon VA. Paraneoplastic antibodies coexist and predict cancer, not neurological syndrome. Ann Neurol. 2004;56(5):715–9. https://doi.org/10.1002/ana.20269.
Toledano M, Britton JW, McKeon A, Shin C, Lennon VA, Quek AM, et al. Utility of an immunotherapy trial in evaluating patients with presumed autoimmune epilepsy. Neurology. 2014;82(18):1578–86. https://doi.org/10.1212/WNL.0000000000000383.
Sechi E, Morris PP, McKeon A, Pittock SJ, Hinson SR, Weinshenker BG, et al. Glial fibrillary acidic protein IgG related myelitis: characterisation and comparison with aquaporin-4-IgG myelitis. J Neurol Neurosurg Psychiatry. 2018. https://doi.org/10.1136/jnnp-2018-318004.
Iizuka T, Sakai F, Ide T, Monzen T, Yoshii S, Iigaya M, et al. Anti-NMDA receptor encephalitis in Japan: long-term outcome without tumor removal. Neurology. 2008;70(7):504–11. https://doi.org/10.1212/01.wnl.0000278388.90370.c3.
Szots M, Marton A, Kover F, Kiss T, Berki T, Nagy F, et al. Natural course of LGI1 encephalitis: 3-5 years of follow-up without immunotherapy. J Neurol Sci. 2014;343(1–2):198–202. https://doi.org/10.1016/j.jns.2014.05.048.
Finke C, Kopp UA, Pruss H, Dalmau J, Wandinger KP, Ploner CJ. Cognitive deficits following anti-NMDA receptor encephalitis. J Neurol Neurosurg Psychiatry. 2012;83(2):195–8. https://doi.org/10.1136/jnnp-2011-300411.
Finke C, Pruss H, Heine J, Reuter S, Kopp UA, Wegner F, et al. Evaluation of cognitive deficits and structural hippocampal damage in encephalitis with leucine-rich, glioma-inactivated 1 antibodies. JAMA Neurol. 2017;74(1):50–9. https://doi.org/10.1001/jamaneurol.2016.4226.
McKeon GL, Robinson GA, Ryan AE, Blum S, Gillis D, Finke C, et al. Cognitive outcomes following anti-N-methyl-D-aspartate receptor encephalitis: a systematic review. J Clin Exp Neuropsychol. 2018;40(3):234–52. https://doi.org/10.1080/13803395.2017.1329408.
McKeon GL, Scott JG, Spooner DM, Ryan AE, Blum S, Gillis D, et al. Cognitive and social functioning deficits after anti-N-methyl-D-aspartate receptor encephalitis: an exploratory case series. J Int Neuropsychol Soc. 2016;22(8):828–38. https://doi.org/10.1017/S1355617716000679.
de Bruijn M, Aarsen FK, van Oosterhout MP, van der Knoop MM, Catsman-Berrevoets CE, Schreurs MWJ, et al. Long-term neuropsychological outcome following pediatric anti-NMDAR encephalitis. Neurology. 2018;90(22):e1997–2005. https://doi.org/10.1212/WNL.0000000000005605.
Dubey D, Kothapalli N, McKeon A, Flanagan EP, Lennon VA, Klein CJ, et al. Predictors of neural-specific autoantibodies and immunotherapy response in patients with cognitive dysfunction. J Neuroimmunol. 2018;323:62–72. https://doi.org/10.1016/j.jneuroim.2018.07.009.
Evangelopoulos ME, Andreadou E, Koutsis G, Koutoulidis V, Anagnostouli M, Katsika P, et al. Treatment of neuromyelitis optica and neuromyelitis optica spectrum disorders with rituximab using a maintenance treatment regimen and close CD19 B cell monitoring. A six-year follow-up. J Neurol Sci. 2017;372:92–6. https://doi.org/10.1016/j.jns.2016.11.016.
Lee WJ, Lee ST, Byun JI, Sunwoo JS, Kim TJ, Lim JA, et al. Rituximab treatment for autoimmune limbic encephalitis in an institutional cohort. Neurology. 2016;86(18):1683–91. https://doi.org/10.1212/WNL.0000000000002635.
Lee WJ, Lee ST, Moon J, Sunwoo JS, Byun JI, Lim JA, et al. Tocilizumab in autoimmune encephalitis refractory to rituximab: an institutional cohort study. Neurotherapeutics. 2016;13(4):824–32. https://doi.org/10.1007/s13311-016-0442-6.
Lim JA, Lee ST, Moon J, Jun JS, Park BS, Byun JI, et al. New feasible treatment for refractory autoimmune encephalitis: low-dose interleukin-2. J Neuroimmunol. 2016;299:107–11. https://doi.org/10.1016/j.jneuroim.2016.09.001.
Scheibe F, Pruss H, Mengel AM, Kohler S, Numann A, Kohnlein M, et al. Bortezomib for treatment of therapy-refractory anti-NMDA receptor encephalitis. Neurology. 2017;88(4):366–70. https://doi.org/10.1212/WNL.0000000000003536.
Flanagan EP, Aksamit AJ, Kumar N, Morparia NP, Keegan BM, Weinshenker BG. Simultaneous PML-IRIS and myelitis in a patient with neuromyelitis optica spectrum disorder. Neurol Clin Pract. 2013;3(5):448–51. https://doi.org/10.1212/CPJ.0b013e3182a78f82.
Sharma R, Chakraborty T, Buadi FK, Beam E, Pureza VS, Pagani-Estevez GL, et al. Clinical reasoning: a 56-year-old woman with acute vertigo and diplopia. Neurology. 2018;90(16):748–52. https://doi.org/10.1212/WNL.0000000000005337.
Armangue T, Spatola M, Vlagea A, Mattozzi S, Carceles-Cordon M, Martinez-Heras E, et al. Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol. 2018;17(9):760–72. https://doi.org/10.1016/S1474-4422(18)30244-8 This study reports for the first time a disproportionally high occurrence of autoimmune encephalitis following HSV encephalitis with important therapeutical implications.
Kao JC, Liao B, Markovic SN, Klein CJ, Naddaf E, Staff NP, et al. Neurological complications associated with anti-programmed death 1 (PD-1) antibodies. JAMA Neurol. 2017;74(10):1216–22. https://doi.org/10.1001/jamaneurol.2017.1912.
Larkin J, Chmielowski B, Lao CD, Hodi FS, Sharfman W, Weber J, et al. Neurologic serious adverse events associated with nivolumab plus ipilimumab or nivolumab alone in advanced melanoma, including a case series of encephalitis. Oncologist. 2017;22(6):709–18. https://doi.org/10.1634/theoncologist.2016-0487.
Cohen DA, Lopez-Chiriboga AS, Pittock SJ, Gadoth A, Zekeridou A, Boilson BA, et al. Posttransplant autoimmune encephalitis. Neurol Neuroimmunol Neuroinflamm. 2018;5(6):e497. https://doi.org/10.1212/NXI.0000000000000497.
Rathore GS, Leung KS, Muscal E. Autoimmune encephalitis following bone marrow transplantation. Pediatr Neurol. 2015;53(3):253–6. https://doi.org/10.1016/j.pediatrneurol.2015.05.011.
Zhao CZ, Erickson J, Dalmau J. Clinical reasoning: agitation and psychosis in a patient after renal transplantation. Neurology. 2012;79(5):e41–4. https://doi.org/10.1212/WNL.0b013e3182616fad.
Seeley WW, Marty FM, Holmes TM, Upchurch K, Soiffer RJ, Antin JH, et al. Post-transplant acute limbic encephalitis: clinical features and relationship to HHV6. Neurology. 2007;69(2):156–65. https://doi.org/10.1212/01.wnl.0000265591.10200.d7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Dementia
Rights and permissions
About this article
Cite this article
Sechi, E., Flanagan, E.P. Diagnosis and Management of Autoimmune Dementia. Curr Treat Options Neurol 21, 11 (2019). https://doi.org/10.1007/s11940-019-0550-9
Published:
DOI: https://doi.org/10.1007/s11940-019-0550-9