Abstract
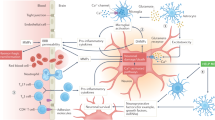
To keep ischemic brain cells alive, neuroprotective agents target events in the ischemic cascade that might be injurious to the cells. They can be divided broadly into groups that restore ion balance, block receptors, prevent reperfusion injury, or promote neuronal healing. To date, neuroprotective agents have either shown a lack of efficacy in clinical stroke trials or been limited by side effects. Ongoing clinical trials with novel agents are trying to enroll a more homogeneous population of stroke patients in an effort to demonstrate treatment benefit.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References and Recommended Reading
The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group: tissue plasminogen activator for acute ischemic stroke. N Engl J Med 1995, 333:1581–1587.
Pulsinelli W: Pathophysiology of acute ischaemic stroke. Lancet 1992, 339:533–536.
Hallenbeck JM, Dutka AJ: Background review and current concepts of reperfusion injury. Arch Neurol 1990, 47:1245–1254.
Lutsep HL, Albers GW, de Crespigny A, et al.: Clinical utility of diffusion weighted MRI in the assessment of ischemic stroke. Ann Neurol 1997, 4:574–580.
Albers GW, Lansberg MG, O’Brien MW, et al.: Evolution of cerebral infarct volume assessed by diffusion-weighted MRI: implications for clinical stroke trials. Presented at the Annual Meeting of the American Academy of Neurology, April 22, 1999, Toronto, Canada.
Fayad P, Davidson WJ, Sullivan MA, et al.: A double-blind, placebo-controlled, safety, efficacy and dose response trial of two intravenous doses of BMS-204352 in patient with acute stroke. Presented at the 24th American Heart Association International Conference on Stroke and Cerebral Circulation, February 4–6, 1999, Nashville, Tennessee.
Horn J, Haan R, Vermeulen M, Limburg M: VENUS — very early nimodipine use in stroke: preliminary trial results. Presented at the 24th American Heart Association International Conference on Stroke and Cerebral Circulation, February 4–6, 1999, Nashville, Tennessee.
Pulsinelli WA, Mann ME, Welch MA, et al.: Fosphenytoin in acute ischemic stroke: efficacy results. Presented at the Annual Meeting of the American Academy of Neurology, April 21, 1999, Toronto, Canada.
Hantson L, Wessel T: Therapeutic benefits of lubeluzole in ischemic stroke. Presented at the 23rd International Joint Conference on Stroke and Cerebral Circulation, February 7, 1998, Orlando, FL; Janssen Corporation.
Diener HC: Lubeluzole in acute ischemic stroke treatment: Lack of efficacy in a large phase III study with an 8-hour window. Presented at the 24th American Heart Association International Conference on Stroke and Cerebral Circulation, February 4, 1999, Nashville, Tennessee.
Grotta JC. Combination therapy stroke trial: rt-PA +/- lubeluzole. Presented at the 25th International Stroke Conference, February 10, 2000, New Orleans, Louisiana.
Albers GW, Atkinson R, Kelley R, et al.: Safety, tolerability, and pharmacokinetics of the N-methyl-D-aspartate antagonist dextrorphan in patients with an acute stroke. Stroke 1995, 26:254–258.
Davis SM, Albers GW, Diener H-C, et al.: Termination of acute stroke studies involving selfotel treatment. Lancet 1997, 349:32.
Dyker AG, Edwards KR, Fayad PB, et al.: Safety and tolerability study of aptiganel hydrochloride in patients with an acute ischemic stroke. Stroke 1999, 30:2038–2042.
Albers GW, Clark WM, Atkinson RP, et al.: Dose escalation study of the NMDA glycine-site antagonist licostinel in acute ischemic stroke. Stroke 1999, 30:508–513. Interesting study
Sacco RL, Lees KR: Glycine antagonist (GV150526) in neuroprotection for patients with acute stroke (The Gain Americas and Gain International Trials). Presented at the 24th American Heart Association International Conference on Stroke and Cerebral Circulation, February 4–6, 1999, Nashville, Tennessee.
Lees KR. GAIN International. Presented at the 25th International Stroke Conference, February 10, 2000, New Orleans, Louisiana.
Sacco RL, Phillips SJ, Haley EC, Levin B. Efficacy and safety of GV150526 in acute stroke: results of the GAIN America trial (Glycine Antagonist in Neuroprotection). Presented at the American Academy of Neurology 52nd Annual Meeting, May 2, 2000, San Diego, CA.
Muir KW, Lees KR: Dose optimization of intravenous magnesium sulfate after acute stroke. Stroke 1998, 29:918–923.
Lees KR: Cerestat and other NMDA antagonists in ischemic stroke. Neurology 1997, 49(suppl):66–69.
Muir KW, Lees KR: A randomized, double-blind, placebocontrolled pilot trial of intravenous magnesium sulfate in acute stroke. Stroke 1995, 26:1183–1188.
Bradford AP: IMAGES — Intravenous magnesium efficacy in stroke trial. Presented at the 25th International Stroke Conference, February 10, 2000, New Orleans, Louisiana.
Clark WM, Raps EC, Tong DC, Kelly RE: Cervene (nalmefene) in acute ischemic stroke: final results of a phase III efficacy study. Stroke 2000, 31:1234–1239. Results of nalmefene study.
Wahlgren NG: The clomethiazole acute stroke study (CLASS): Efficacy results in a subgroup of 545 patients with total anterior circulation syndrome. Presented at the 23rd International Joint Conference on Stroke and Cerebral Circulation, February 7, 1998, Orlando, FL.
Bologa M and the Steering Committee on behalf of the BRAIN Study Group: Phase II study of the neuroprotective BAY x3702, in patients with ischemic stroke. Presented at the 23rd American Heart Association International Conference on Stroke and Cerebral Circulation, February 5–7, 1998, Orlando, Florida.
Clark WM, Madden KP, Rothlein R, Zivin JA: Reduction of central nervous system ischemic injury by monoclonal antibody to intercellular adhesion molecule. J Neurosurg 1991, 75:623–627.
The Enlimomab Acute Stroke Trial Investigators: The enlimomab acute stroke trial: final results [abstract]. Neurology 1997, 48:270 (final results presented at the 49th meeting of the American Academy of Neurology, April 16, 1997, Boston, MA).
Ginberg MD, Busto R: Combating hyperthermia in acute stroke: A significant clinical concern. Stroke 1998, 29:529–534.
Clark W, Warach S: Randomized dose response trial of citicoline in acute ischemic stroke patients [abstract]. Neurology 1996, 46:42.
Clark WM, Williams BJ, Selzer KA, et al.: Randomized efficacy trial of citicoline in acute ischemic stroke. Presented at the 23rd International Joint Conference on Stroke and Cerebral Circulation, February 7, 1998, Orlando, FL.
Gammans RE, Sherman DG, ECCO 2000 Investigators. ECCO 2000 study of citicoline for treatment of acute ischemic stroke: Final results. Presented at the 25th International Stroke Conference, February 10, 2000, New Orleans, Louisiana.
Warach S. Use of diffusion-weighted imaging for evaluation of neuroprotective agents. Presented at the 25th International Stroke Conference, February 10, 2000, New Orleans, Louisiana.
The Fiblast Safety Study Group: Clinical safety trial of intravenous basic fibroblast growth factor (bFGF, Fiblast) in acute stroke. Presented at the 23rd International Joint Conference on Stroke and Cerebral Circulation, February 7, 1998, Orlando, FL.
Clark WM, Schim JD, Kasner SE, Victor SJ and the Fiblast Stroke Study Investigators. Trafermin in acute ischemic stroke: results of a phase II/III randomized efficacy study [abstract]. Neurology 2000, 54:88.
Haley EC: High-dose tirilazad for acute stroke (RANTTAS II). Stroke 1998, 29:1256–1257.
De Keyser J, Sulter G, Luiten PG: Clinical trials with neuroprotective drugs in acute ischemic stroke: are we doing the right thing? Trends Neurosci 1999, 22:535–540.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lutsep, H.L., Clark, W.M. Current status of neuroprotective agents in the treatment of acute ischemic stroke. Curr Neurol Neurosci Rep 1, 13–18 (2001). https://doi.org/10.1007/s11910-001-0072-0
Issue Date:
DOI: https://doi.org/10.1007/s11910-001-0072-0