Abstract
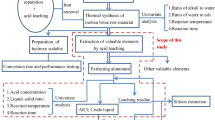
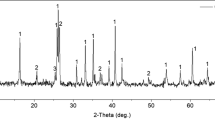
This paper presents a new method of recycling aluminum and iron in boiler slag derived from plants that use coal as fuel. The new method integrates efficient extraction and reuse of the leached pellets together. An elemental analysis of aqueous solutions leached by sulfuric acid was determined by EDTA-Na2-ZnCl2 titration method. The components and microstructures of the samples were examined by means of XRF, XRD and SEM. An aluminum extraction efficiency of 86.50% was achieved when the sintered pellets were leached using 4 mol · L−1 H2SO4 at solid/ liquid [m(g)/V(mL)] ratio of 1: 5 at 80 °C for 24 h. An iron extraction efficiency of 94.60% was achieved in the same conditions for the maximum extraction efficiency of Al. The extraction efficiencies of Al and Fe increased with an increase in temperature, leaching time and acidity. The concentration of alumina and iron hydroxide in the final product was determined to be 99.12% and 92.20% respectively. This product of alumina would be used directly for the production of metallic aluminum.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bailey N T, Chapman R J. Use of Coal Spoils as Feed Materials for Alumina Recovery by Acid Leaching Routes[J]. Hydrometallurgy, 1987, 18(3): 337–350.
Seeley F G, Mcdowell W J, Felker L K, et al. Determination of Extraction Equilibrium for Several Metals in the Development of a Process Designed to Recover Aluminum and Other Metals from Coal Combustion[J]. Hydrometallurgy, 1981, 11(6): 277–290.
Wei Chu. Lead Metal Removal by Recycled Alum Sludge[J]. Water Research, 1999, 33(13): 3019–3025.
Bereza L V, Pechersk N P, Ni L P. Leaching of Ekibastuz Coal Ashes Studied by an Experimental Planning Method[J]. Fuel, 1972, 78(2): 457–462.
Burnet G, Murtha M J, Dunker J W. Recovery of Metals from Coal Ash[J]. Metallurgical Soc of AIME, 1985, 13(4): 747–769.
Seidel A, Zimmels Y. Mechanism and Kinetics of Aluminum and Iron Leaching from Coal Fly Ash by Sulfuric Acid[J]. Chemical Engineering Science, 1998, 53(22): 3835–3851.
Burkin A R. Production of Aluminum and Alumina[M]. 4th ed. New York: John Wiley & Chichester, 1987:235–238.
Kelmers A D, Canon R M. Chemistry of the Direct Acid Leach, Calsinter and Pressure Digestion Acid Leach Methods for the Recovery of Alumina from Fly Ash[J]. Res Conserve, 1982, 22(9): 271–279.
Kelmers A D, Egan B Z, Seeley F G, et al. Direct Acid Dissolution of Aluminum and other Metals from Fly Ash[J]. The Metallurgical Society of AIME, 1981, 26(4): 24–81.
Matjie R H. The Selective Removal of Iron and Titanium from an Aluminum Containing Aqueous Phase[J]. Minerals Engineering, 2005, 18(3): 299–310.
Park Kyun Young, Jeong Jinki, Manufacture of Low-Soda Alumina from Clay[J]. Industrial & Engineering Chemistry Research, 1996, 35(11): 4379–4385
Torma A E. Extraction of Aluminum from Fly Ash[J]. Metals (Berlin), 1983, 37(6): 589–592.
Seidel A. Mechanism of Bioleaching of Coal Fly Ash by Thiobacillus Thiooxidans[J]. Chemical Engineering Journal, 2001, 83(2): 123–130.
Gabler Jr R C, Stoll R L. Removal of Leachable Metals and Recovery of Alumina from Utility Coal Ash[J]. Res Conserve, 1982, 18(9): 131–142.
Matjie R H, Bunt J R, van Heerden J H P. Extraction of Alumina from Coal Fly Ash Generated from a Selected Low Rank Bituminous South African Coal[J]. Minerals Engineering, 2005, 18(7): 299–310.
Maohong Fan, Rabert C B, Thomas D W, et al. Production of a Complex Coagulant from Fly Ash[J]. Chemical Engineering Journal, 2005, 106(3): 269–277.
Mangialardi T. Sintering of MSW Fly Ash for Reuse as a Concrete Aggregate[J]. Journal of Hazardous Materials, 2001, 87(3): 225–239.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Supported by the Communication, Science and Education Foundation of Hubei Province(2005-570)
Biography: LI Jinping (1979–), male, Ph.D. candidate, research direction: further utilization of industrial slag.
Rights and permissions
About this article
Cite this article
Li, J., Hou, H., Gan, J. et al. Extraction of aluminum and iron from boiler slag by sulfuric acid. Wuhan Univ. J. of Nat. Sci. 12, 541–547 (2007). https://doi.org/10.1007/s11859-006-0071-8
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11859-006-0071-8