Abstract
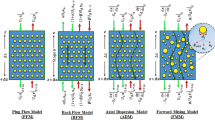
Reactive extraction development was investigated to extract vanadium ions from sulfate solution in the modified rotating disc column (MRDC). It was found from batch experiments that D2EHPA and TBP concentrations, initial aqueous phase pH, and the concentration of NH4OH as a stripping agent were optimized equal to 0.3 M, 0.36 M, 2, and 1 M, respectively. In the continuous experiments, the effects of rotor speed, aqueous and organic phase flow rates, and mass transfer direction were investigated on the dispersed phase holdup, mass transfer coefficients, and vanadium extraction. The experiment design was based on the response surface design to analyze the dependence of responses with the input parameters. The uncertainty analysis by the Monte Carlo simulation indicates that the rotor speed, reaction conditions, and phase flow rates affected the dispersed phase holdup. By applying the axial dispersion model, the performance of mass transfer coefficients in terms of agitation speed, aqueous phase flow rate, organic phase flow rate, and mass transfer direction was evaluated under the chemical reaction system. A new model by considering the dimensionless numbers has been provided to predict overall mass transfer data based on the dispersed phase.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- AARE:
-
average absolute relative error [-]
- a:
-
interfacial area [m2/m3]
- Bn :
-
nth coefficient in Eqs. (1)–(4) [-]
- d32 :
-
sauter mean diameter [m]
- di :
-
drop diameter [m]
- de :
-
equivalent diameter [m]
- Dc :
-
column diameter [m]
- Deff :
-
effective diffusivity [m2/s]
- DR :
-
disc diameter [m]
- Ds :
-
stator ring diameter [m]
- Eö:
-
Eötvös number [-]
- E:
-
axial mixing coefficient [m2/s]
- E%:
-
extraction efficiency [-]
- e:
-
fractional free cross-sectional area [-]
- g:
-
acceleration due to gravity [m/s2]
- hc :
-
compartment height [m]
- K:
-
overall mass transfer coefficient [m/s]
- m:
-
distribution ratio [-]
- N:
-
rotor speed [rps]
- N ox :
-
number of ‘true’ transfer unit [-]
- NDP:
-
number of data points [-]
- Pe:
-
Peclet number [-]
- R:
-
enhancement factor for mass transfer [-]
- RMSE:
-
root mean square error [-]
- Re:
-
Reynolds number [-]
- Sc:
-
Schmidt number [-]
- S%:
-
stripping efficiency [-]
- S.E.F:
-
synergistic enhancement factor [-]
- t:
-
time [s]
- V:
-
superficial velocity [m/s]
- Vt :
-
terminal velocity [m/s]
- Vs :
-
slip velocity [m/s]
- \({\overline {\rm{V}} _c}\) :
-
continuous phase true velocity [m/s]
- ∀:
-
volume of the extraction region [m3]
- x:
-
vanadium mass fraction in continuous phase [-]
- x*:
-
equilibrium mass fraction [-]
- y:
-
vanadium mass fraction in dispersed phase [-]
- ρ c :
-
density of aqueous phase
- ρ d :
-
density of solvent phase
- ρ d :
-
viscosity of solvent phase
- μ c :
-
viscosity of aqueous phase
- φ :
-
dispersed phase holdup
- τ :
-
interfacial tension
References
R. Moskalyk and A. Alfantazi, Miner. Eng., 16, 793 (2003).
M. Tuzen, T. G. Kazi, D. Citak and M. Soylak, J. Anal. Atomic Spect., 28, 1441 (2013).
H. T. Truong, T. H. Nguyen and M. S. Lee, Hydrometallurgy, 171, 298 (2017).
L. Zeng and C. Y. Cheng, Hydrometallurgy, 101, 141 (2010).
Q. Shi, Y. Zhang, J. Huang, T. Liu, H. Liu and L. Wang, Sep. Purif. Technol., 181, 1 (2017).
A. K. Nayak, N. Devi and K. Sarangi, Trans. Indian Inst. Met., 74, 3155 (2021).
X. B. Li, C. Wei, J. Wu, C. X. Li, M. T. Li, Z. G. Deng and H. S. Xu, Trans. Nonferrous Met. Soc. China, 22, 461 (2012).
H. Liu, Y. M. Zhang, J. Huang, T. Liu and D. S. Luo, Sep. Purif. Technol., 215, 335 (2019).
Z. Zhu, K. Tulpatowicz, Y. Pranolo and C. Y. Cheng, Hydrometallurgy, 154, 72 (2015).
M. R. Yaftian, M. I. G. S. Almeida, R. W. Cattrall and S. D. Kolev, J. Mem. Sci., 545, 57 (2018).
G. Hu, D. Chen, L. Wang, J. C. Liu, H. Zhao, Y. Liu, T. Qi, C. Zhang and P. Yu, Sep. Purif. Technol., 125, 59 (2014).
X. Li, C. Wei, Z. Deng, M. Li, C. Li and G. Fan, Hydrometallurgy, 105, 359 (2011).
M. R. Tavakoli and D. B. Dreisinger, Hydrometallurgy, 141, 17 (2014).
P. N. Remya and M. L. Reddy, J. Chem. Eng. Biotechnol., 79, 434 (2004).
M. Asadollahzadeh, A. Ghaemi, M. Torab-Mostaedi and S. Shahhosseini, Chinese J. Chem. Eng., 24, 989 (2016).
R. Ettouney and M. El-Rifai, Chem. Eng. Res. Des., 89, 2228 (2011).
M. Asadollahzadeh, R. Torkaman, M. Torab-Mostaedi and M. Saremi, Inter. J. Heat Mass Trans., 188, 122638 (2022).
M. Asadollahzadeh, A. Hemmati, M. Torab-Mostaedi, M. Shirvani, A. Ghaemi and Z. Mohsenzadeh, Chinese J. Chem. Eng., 25, 53 (2017).
T. Murugesan and I. Regupathi, J. Chem. Eng. Japan, 37, 1293 (2004).
Z. Jia-Wen, Z. Shou-Hua, Z. Xiao-Kui, C. Xiao-Xiang, S. Yuan-Fu and A. Vogelpohl, Chem. Eng. Technol., 14, 167 (1991).
X. Chen, K. Li and Y. Su, Ind. Eng. Chem. Res., 32, 453 (1993).
N. V. Hendre, V. Venkatasubramani, R. A. Farakte and A. W. Patwardhan, Ind. Eng. Chem. Res., 57, 1630 (2018).
B. Kadam, J. Joshi and R. Paul, Chem. Eng. Res. Des., 87, 756 (2009).
N. V. Hendre, S. P. Hinge and A. W. Patwardhan, Ind. Eng. Chem. Res., 60, 5945 (2021).
A. Porto, L. Sarubbo, J. Lima-Filho, M. Aires-Barros, J. Cabral and E. Tambourgi, Bioprocess Eng., 22, 215 (2000).
J. S. Coimbra, F. Mojola and A. J. Meirelles, J. Chem. Eng. Japan, 31, 277 (1998).
M. Carneiro-da-Cunha, M. Aires-Barros, E. Tambourgi and J. Cabral, Biotechnol. Tech., 8, 413 (1994).
A. Hemmati, A. Ghaemi and M. Asadollahzadeh, Sep. Sci. Technol., 57, 225 (2022).
B. Shakib, R. Torkaman, M. Torab-Mostaedi, M. Saremi and M. Asadollahzadeh, Chem. Eng. Process, 171, 108762 (2022).
M. Asadollahzadeh, R. Torkaman and M. Torab-Mostaedi, Chem. Eng. Process, 169, 108608 (2021).
R. Torkaman, M. R. A. Rovais, M. Heydari, M. Torab-Mostaedi and M. Asadollahzadeh, Prog. Nucl. Energy, 147, 104217 (2022).
B. Shakib, R. Torkaman, M. Torab-Mostaedi and M. Asadollahzadeh, Inter. J. Heat Mass Trans., 185, 122337 (2022).
M. Asadollahzadeh, R. Torkaman, M. Torab-Mostaedi and M. Saremi, Sci. Rep., 12, 1609 (2022).
A. Hemmati, A. Ghaemi and M. Asadollahzadeh, Sep. Sci. Technol., 56, 2734 (2020).
B. Shakib, A. Ghaemi, A. Hemmati and M. Asadollahzadeh, Prog. Nucl. Energy, 141, 103969 (2021).
H. Gröber and Z. Var, Dtsch. Ing., 69, 705 (1925).
R. Kronig and J. Brink, Appl. Sci. Res., 2, 142 (1951).
A. Handlos and T. Baron, AIChE J., 3, 127 (1957).
A. I. Johnson and A. Hamielec, AIChE J., 6, 145 (1960).
A. Kumar and S. Hartland, Ind. Eng. Chem. Res., 35, 2682 (1996).
C. Wilke and P. Chang, AIChE J., 1, 264 (1955).
M. Torab-Mostaedi, A. Ghaemi and M. Asadollahzadeh, Chem. Eng. Res. Des., 89, 2742 (2011).
P. V. Danckwerts, Chem. Eng. Sci., 2, 1 (1953).
H. Pratt and G. Stevens, Science and practise of liquid-liquid extraction, Clarendon Press, Oxford (1992).
B. Shakib, R. Torkaman, M. Torab-Mostaedi and M. Asadollahzadeh, Inter. Commun. Heat Mass Trans., 118, 104903 (2020).
B. Shakib, R. Torkaman, M. Torab-Mostaedi and M. Asadollahzadeh, Chem. Pap., 74, 4295 (2020).
I. Macdonald and P. Strachan, Energy Build., 33, 219 (2001).
L. Boyadzhiev, D. Elenkov and G. Kyuchukov, Canadian J. Chem. Eng., 47, 42 (1969).
L. Steiner, Chem. Eng. Sci., 41, 1979 (1986).
A. Hemmati, M. Torab-Mostaedi and M. Asadollahzadeh, Chem. Eng. Res. Des., 93, 747 (2015).
M. Asadollahzadeh, S. Shahhosseini, M. Torab-Mostaedi and A. Ghaemi, Chem. Eng. Res. Des., 100, 104 (2015).
H. Bahmanyar, L. Nazari and A. Sadr, Chem. Eng. Process, 47, 57 (2008).
M. Torab-Mostaedi, A. Ghaemi and M. Asadollahzadeh, Canadian J. Chem. Eng., 90, 1570 (2012).
L. Steiner, A. Kumar and S. Hartland, Canadian J. Chem. Eng., 66, 241 (1988).
A. Rahbar, Z. Azizi, H. Bahmanyar and M. A. Moosavian, Canadian J. Chem. Eng., 89, 508 (2011).
B. Shakib, M. Torab-Mostaedi, M. Outokesh and M. Asadollahzadeh, Heat Mass Trans., 56, 1995 (2020).
M. Torab-Mostaedi, M. Asadollahzadeh and J. Safdari, Chinese J. Chem. Eng., 25, 288 (2017).
R. Torkaman, M. Torab-Mostaedi, J. Safdari, S. M. A. Moosavian and M. Asadollahzadeh, Iranian J. Chem. Chem. Eng., 36, 145 (2017).
M. Amanabadi, H. Bahmanyar, Z. Zarkeshan and M. A. Mousavian, Chinese J. Chem. Eng., 17, 366 (2009).
A. Hemmati, M. Shirvani, M. Torab-Mostaedi and A. Ghaemi, Chem. Eng. Process, 100, 19 (2016).
M. Torab-Mostaedi, J. Safdari, M. Ghannadi-Maragheh and M. A. Moosavian, J. Chem. Eng. Japan, 42, 78 (2009).
M. Torab-Mostaedi and J. Safdari, Brazilian J. Chem. Eng., 26, 685 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shakib, B., Asadollahzadeh, M., Outokesh, M. et al. Reactive extraction evaluation for vanadium (V) removal in the MRDC column using axial dispersion and central composition approach. Korean J. Chem. Eng. 39, 3399–3411 (2022). https://doi.org/10.1007/s11814-022-1239-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-022-1239-4