Abstract
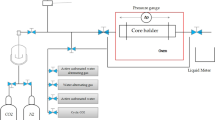
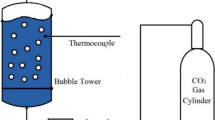
Carbon dioxide (CO2) emitted by fossil energy combustion is related to the greenhouse effect. To further study the motion dynamics of CO2 bubbles in various solutions so as to better absorb them, a CO2 bubble experimental platform was built. The growth and motion of a single CO2 bubble were experimented in five concentrations of NaOH, NaHCO3 solutions and five viscosity deionized waters, photographed with a high-speed camera and imported into PyCharm for analysis. Based on this, four kinds of CO2 bubbles were experimentally studied. The results show that the viscosity leads to the increase of rising time; the maximum rise time is 0.518 s when the viscosity is 100 mPa·s, the aspect ratio δ of CO2 bubble in solution, there will be an “L” distribution, and the minimum rise time is close to rising after the bubble is separated from the injector. NaHCO3 inhibits the reaction between NaOH solution and CO2, resulting in the cross-section ratio κ change decreasing. The concentration of NaOH solution most conducive to CO2 absorption is 0.039 g/ml and 0.058 g/ml.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Binet, J. L. Probst, C. B. Guilhe, J. L. Seidel, C. Emblanch and N. Peyraube, Geochim. Cosmochim. AC, 270, 184 (2020).
Y. Li, H. Y. Wang, Y. Z. Wang and J. Zhao, Huadian Technol., 43, 11 (2021).
D. Schmidt, C. Yildiz, J. Ströhle and B. Epple, Fuel, 284, 15 (2021).
W. Q. Fan, D. Pan, L. Huang and Q. Wang, Mater. Guide, 35, 17 (2021).
X. Qian, L. F. Deng, L. F. Wang, R. Shan and H. R. Yuan, Mater. Guide, 33, 11 (2019).
M. Xu, X. Zhang, J. L. Fan, G. Lin and D. Xu, J. Mining Sci., 6, 06 (2021).
X. P. Bu, Clean Coal Technol., 20, 05 (2014).
A. Y. Ghalya and M. A. Seddeek, Chaos Soliton Fract, 19, 1 (2004).
D. Rodrigue, Can. J. Chem. Eng., 79, 1 (2001).
S. B. Li, Y. G. Ma and T. T. Fu, Chem. Eng. -New York, 39, 10 (2011).
H. Bai and A. C. Yeh, Ind. Eng. Chem. Res., 36, 6 (1997).
Y. Wang, Y. Su, W. X. Hou, T. Y. Fan, Y. Y. Xiao, R. Yin, Y. Li and J. Zhao, Chem. Manage., 31, 19 (2021).
C. B. Liu, Marine Power Technol., 41, 10 (2021).
M. Garcia, H. K. Knuutila, U. E. Aronu and S. Gu, Int. J. Greenh. Gas Con., 78, 11 (2018).
X. S. Li, J. Liu, W. F. Jiang, G. Gao, F. Wu, C. Luo, L. Qi and C. Zhang, Sep. Purif. Technol., 275, 15 (2021).
P. Niegodajew and D. Asendrych, Appl. Math. Model., 40, 23 (2016).
M. F. Kofal, A. Mustafa, A. F. Ismail, M. R. DashtArzhandi and T. Matsuura, J. Nat. Gas Sci. Eng., 31, 4 (2016).
L. Ansaloni, A. Hartono, M. Awais, H. K. Knuutila and L. Deng, Chem. Eng. J., 359, 11 (2018).
L. Xu, Y. Qin, L. Liu, J. Xiao and Z. Ding, Korean J. Chem. Eng., 38, 1032 (2021).
R. K. Nilavuckkarasi, P. Muthumari and B. Ambedkar, Chem. Eng. Process, 151, 3 (2020).
C. Pevida, T. C. Drage and C. E. Snape, Carbon, 46, 11 (2008).
K. Wang, F. Gu and P. T. Clough, Chem. Eng. J., 408, 12 (2020).
M. H. Wang, H. M. Liu, Y. L. Xie, S. Gao, J. Ding and Y.X. Wang, Ion Exchange and Adsorption, 37, 04 (2021).
G. Falzone, I. Mehdipour, N. Neithalath, M. Bauchy and G. Sant, AIChE J., 67, 36 (2021).
B. Silva, D. Silva and A. Silva, J. Sol-Gel Sci. Technol., 97, 6 (2021).
V. Kolachana, K. Cholkar, W. M. Kayani, G. K. Kouassi and N. M. M. Gowda, Am. J. Org. Chem., 2(1), 18 (2012).
L. Zhang, S. X. Ju, Y. F. Yan and Z. E. Zhang, J. Chem. Eng., 65, 06 (2014).
A. Shadloo, K. Peyvandi and A. Shojaeian, J. Mol. Liq., 347, 2 (2021).
J. Saikiaa, A. Saikiab and D. Saikiac, Mater. Today, 09, 535 (2020).
Z. Y. Ding, R. Ding, X. Zheng and X. Chen, J. Fujian Normal University, 37, 06 (2021).
Y. J. Shi, X. Y. Wang, Y. Zhang and L. M. Wei, Micro Nano Electronic Technol., 56, 04 (2019).
P. Ritesh and M. S. Kumar, J. Ind. Eng. Chem., 90, 10 (2020).
H. Li, Z. Liu, J. H. Chen, B. J. Sun, Y. L. Guo and H. K. He, Exp. Therm. Fluid Sci., 88, 11 (2017).
A. Shohei, H. Kosuke, H. Shigeo and T. Akio, Exp. Therm. Fluid Sci., 96, 9 (2018).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Jx., Gao, D., Qi, Yw. et al. Experimental study on CO2 bubble dynamics under different solution viscosity and absorbent concentration. Korean J. Chem. Eng. 39, 2010–2019 (2022). https://doi.org/10.1007/s11814-022-1171-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-022-1171-7