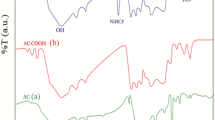
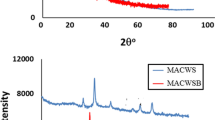
Abstract
Magnetic activated carbons (AC) derived from hazelnut shell using the chemical activation method with ZnCl2 and KOH were prepared in the present work. The adsorption performance of the magnetic sorbents was evaluated for the removal of mercury and arsenic ions in the binary solutions, and the interference of ions with each other during the adsorption process was investigated. The synthesized adsorbents were characterized using XRD, FTIR, BET, XRF, FESEM, TGA and VSM. The XRD results indicated that the small iron oxide crystallites, including goethite and magnetite, were detected on the hazelnut shell-based AC activated by ZnCl2. The extended Langmuir and the modified competitive Langmuir isotherms were applied to fit the competitive adsorption of Hg (II) and As (V) ions using genetic algorithm (GA). The experimental data were in good agreement with the extended Langmuir equation, while the correlation coefficient was measured close to 1. The highest adsorption capacity was calculated to be 80 and 39.31 mg/g for mercury and arsenic ions on the magnetic sample activated by ZnCl2, respectively. The kinetic behavior of carbonaceous adsorbents was studied using pseudo-first and second-order models. The effect of various operating conditions was investigated on the competitive adsorption of metal ions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- Co :
-
initial concentration of heavy metal ions, ppm
- Ce :
-
equilibrium concentration of heavy metal ions, ppm
- K1 :
-
constant parameter of pseudo first order kinetic [min−1]
- K2 :
-
constant parameter of pseudo second order kinetic [g·mg−1· min−1]
- M:
-
the adsorbent dosage [gr]
- qe :
-
equilibrium adsorption capacity [mg/g]
- qexp :
-
experimental adsorption capacity [mg/g]
- qmodel :
-
model adsorption capacity [mg/g]
- qt :
-
adsorption capacity at time t [mg/g]
- Re:
-
removal efficiency
- V:
-
volume of solutions [L]
References
M. Marciniak, J. Goscianska, M. Frankowski and R. Pietrzak, J. Mol. Liq., 276, 630 (2019).
M. Sharma, J. Singh, S. Hazra and S. Basu, Microchem. J., 145, 105 (2019).
E. Demirbaş, M. Kobya, S. Öncel and S. Şencan, Bioresour. Technol., 84(3), 291 (2002).
G. Karaçetin, S. Sivrikaya and M. Imamoğlu, J. Anal. Appl. Pyrol., 110, 270 (2014).
A. Ahmadpour, M. Zabihi, M. Tahmasbi and T. R. Bastami, J. Hazard. Mater., 182(1), 552 (2010).
M. Zabihi, A. Ahmadpour and A. H. Asl, J. Hazard. Mater., 167(1), 230 (2009).
M. Zabihi, A. Haghighi Asl and A. Ahmadpour, J. Hazard. Mater., 174(1), 251 (2010).
M. H. Fatehi, J. Shayegan, M. Zabihi and I. Goodarznia, J. Environ. Chem. Eng., 5(2), 1754 (2017).
J. E. Efome, D. Rana, T. Matsuura and C. Q. Lan, Sci. Total Environ., 674, 355 (2019).
V. Nejadshafiee and M. R. Islami, Mater. Sci. Eng.: C., 101, 42 (2019).
Y. Bayrak, Y. Yesiloglu and U. Gecgel, Micropor. Mesopor. Mater., 91(1), 107 (2006).
D. D. Milenković, P. V. Dašić and V. B. Veljković, Ultrason. Sonochem., 16(4), 557 (2009).
Y. Liu, Z. Huo, Z. Song, C. Zhang, D. Ren, H. Zhong and F. Jin, J. Taiwan Inst. Chem. Engineers, 96, 575 (2019).
Y. Jiang, Q. Xie, Y. Zhang, C. Geng, B. Yu and J. Chi, Int. J. Mining Sci. Technol., 29(3), 513 (2019).
A. C. Fröhlich, E. L. Foletto and G. L. Dotto, J. Cleaner Production, 229, 828 (2019).
S. Lin, C. Lian, M. Xu, W. Zhang, L. Liu and K. Lin, Appl. Surf. Sci., 422, 675 (2017).
F. Cao, C. Lian, J. Yu, H. Yang and S. Lin, Bioresour. Technol., 276, 211 (2019).
I. A. Aguayo-Villarreal, A. Bonilla-Petriciolet and R. Muñiz-Valencia, J. Mol. Liq., 230, 686 (2017).
K. Leus, K. Folens, N. R. Nicomel, J. P. H. Perez, M. Filippousi, M. Meledina, M. M. Dîrtu, S. Turner, G. Van Tendeloo, Y. Garcia and G. Du Laing, J. Hazard. Mater., 353, 312 (2018).
R. Liu and B. Lian, Sci. Total Environ., 659, 122 (2019).
M. Jain, M. Yadav, T. Kohout, M. Lahtinen, V. K. Garg and M. Sillanpää, Water Res. Ind., 20, 54 (2018).
V. K. Rathore, D. K. Dohare, P. Mondal, J. Environ. Chem. Eng., 4(2), 2417 (2016).
C. Qu, M. Ma, W. Chen, P. Cai, X.-Y. Yu, X. Feng and Q. Huang, Chemosphere, 193, 943 (2018).
M. Zabihi, F. Khorasheh and J. Shayegan, RSC Adv., 5(7), 5107 (2015).
M. H. Jannat Abadi, S. M. M. Nouri, R. Zhiani, H. D. Heydarzadeh and A. Motavalizadehkakhky, Int. J. Ind. Chem., 10(4), 291 (2019).
K. Fu, Q. Yue, B. Gao, Y. Wang and Q. Li, Colloids Surf. A: Physicochem. Eng. Aspects, 529, 842 (2017).
S. Asgari, Z. Fakhari and S. Berijani, J. Nanostructures, 4(1), 55 (2014).
S. Cheng, L. Zhang, H. Xia, J. Peng, S. Zhang and S. Wang, J. Porous Mater., 22(6), 1527 (2015).
W. Yu, M. Li, X. Ji, Y. Qiu, Y. Zhu and C. Leng, J. Wuhan Univ. Technol.-Mater Sci. Ed., 31(2), 260 (2016).
K. Yaghmaeian, R. Khosravi Mashizi, S. Nasseri, A. H. Mahvi, M. Alimohammadi and S. Nazmara, J. Environ. Health Sci. Eng., 13(1), 55 (2015).
H. Yuh-Shan, Scientometrics, 59(1), 171 (2004)..
Acknowledgement
The authors gratefully acknowledge Neyshabur Branch, Islamic Azad University, Neyshabur, Iran.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zabihi, M., Omidvar, M., Motavalizadehkakhky, A. et al. Competitive adsorption of arsenic and mercury on nano-magnetic activated carbons derived from hazelnut shell. Korean J. Chem. Eng. 39, 367–376 (2022). https://doi.org/10.1007/s11814-021-0903-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0903-4