Abstract
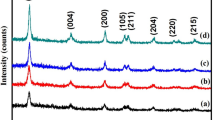
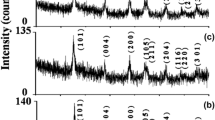
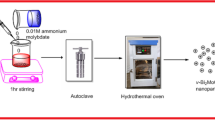
Several mono-and bi-metallic photocatalysts with different loadings have been synthesized using ultrasound assisted sol gel and wet impregnation methods. Synthesized catalysts were characterized using various analytical techniques to determine the bandgap, functional groups, crystallinity, and surface morphology. Photocatalytic performance of the different catalysts was compared in terms of Rhodamine-B dye degradation, and further process optimization studies were carried out to achieve the maximum extent of degradation. Catalyst performance was studied both under UV and visible light irradiation for the degradation of different dyes. Compared to pure TiO2 and monometallic Ag or Bi doped on TiO2, bi-metallic Bi-Ag on TiO2 catalyst synthesized using wet impregnation was found to be highly active under the conditions investigated. Complete degradation (∼100%) was obtained within shorter duration in the presence of minimal catalyst loading (60 mg/L). Overall, this work has clearly demonstrated the higher catalytic activity of bi-metallic catalyst, which is attributed to the synergistic effect of Bi and Ag on TiO2 facilitating the efficient charge separation and improved e− transfer combined with high ability to absorb in visible light region in effectively degrading organic pollutants.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
E. COMMISSION, Publ. Off. Eur. Union (2014).
N. N. Mahamuni and Y. G. Adewuyi, Ultrason. Sonochem., 17, 990 (2010).
Y. G. Adewuyi, Environ. Sci. Technol., 39, 10 (2005).
S. Bagwasi, B. Tian, J. Zhang and M. Nasir, Chem. Eng. J., 217, 108 (2013).
F. Fresno, R. Portela, S. Suárez and J. M. Coronado, J. Mater. Chem. A, 2, 2863 (2014).
X. Wang, Y. Wang and Z. Yan, Int. J. Photoenergy, 2014, 1 (2014).
M. K. Seery, R. George, P. Floris and S. C. Pillai, J. Photochem. Photobiol. A Chem., 189, 258 (2007).
M. A. Behnajady and H. Eskandarloo, Chem. Eng. J., 228, 1207 (2013).
T. S. Natarajan, K. Natarajan, H. C. Bajaj and R. J. Tayade, J. Nanoparticle Res., 15, 1 (2013).
S. I. Mogal, V. G. Gandhi, M. Mishra, S. Tripathi, T. Shripathi, P. A. Joshi and D. O. Shah, Ind. Eng. Chem. Res., 53, 5749 (2014).
N. Sobana, M. Muruganadham and M. Swaminathan, J. Mol. Catal. A Chem., 258, 124 (2006).
H. Zhao, F. Tian, R. Wang and R. Chen, Rev. Adv. Sci. Eng., 3, 3 (2014).
Y. Wu, G. Lu and S. Li, J. Phys. Chem. C, 113, 9950 (2009).
S. Sajjad, S. A. K. Leghari, F. Chen and J. Zhang, Chem. — A Eur. J., 16, 13795 (2010).
J. Xu, M. Chen and D. Fu, Appl. Surf. Sci., 257, 7381 (2011).
K. Atacan, N. Güy and M. Özacar, Colloids Interface Sci. Commun., 40 (2021).
N. Güy, K. Atacan, İ. Yıldırım and M. Özacar, J. Mol. Liq., 326 (2021).
A. Chachvalvutikul, T. Luangwanta, S. Pattisson, G. J. Hutchings and S. Kaowphong, Appl. Surf. Sci., 544 (2021).
V. K. Landge, S. H. Sonawane, M. Sivakumar, S. S. Sonawane, G. Uday Bhaskar Babu and G. Boczkaj, Sustain. Energy Technol. Assessments, 45, 101194 (2021).
C. Zhang, W. Chen, D. Hu, H. Xie, Y. Song, B. Luo, Y. Fang, W. Gao and Z. Zhong, Green Energy Environ. (2021).
S. Rengaraj and X. Z. Li, Int. J. Environ. Pollut., 27, 20 (2006).
V. Vetrivel, K. Rajendran and V. Kalaiselvi, Int. J. ChemTech Res., 7, 1090 (2015).
F. M. Bautista, J. M. Campelo, A. Garcia, D. Luna, J. M. Marinas, M. C. Moreno, A. A. Romero, J. A. Navio and M. Macias, J. Catal., 173, 333 (1998).
S. Rajoriya, S. Bargole, S. George, V. K. Saharan, P. R. Gogate and A. B. Pandit, Sep. Purif. Technol., 209, 254 (2019).
P. S. Sathish Kumar, R. Sivakumar, S. Anandan, J. Madhavan, P. Maruthamuthu and M. Ashokkumar, Water Res., 42, 4878 (2008).
P. C. L. Muraro, S. R. Mortari, B. S. Vizzotto, G. Chuy, C. dos Santos, L. F. W. Brum and W. L. da Silva, Sci. Rep., 10, 1 (2020).
S. Sood, S. K. Mehta, A. Umar and S. K. Kansal, New J. Chem., 38, 3127 (2014).
M. Zimbone, M. A. Buccheri, G. Cacciato, R. Sanz, G. Rappazzo, S. Boninelli, R. Reitano, L. Romano, V. Privitera and M. G. Grimaldi, Appl. Catal. B Environ., 165, 487 (2015).
P. R. Gogate and A. B. Pandit, AIChE J., 50, 5 (2004).
S. Chakrabarti and B. K. Dutta, J. Hazard. Mater., 112, 269 (2004).
U. G. Akpan and B. H. Hameed, Appl. Catal. A Gen., 375, 1 (2010).
U. G. Akpan and B. H. Hameed, J. Hazard. Mater., 170, 2 (2009).
K. Byrappa, A. K. Subramani, S. Ananda, K. M. Lokanatha Rai, R. Dinesh and M. Yoshimura, Bull. Mater. Sci., 29, 433 (2006).
G. Sharma, D. D. Dionysiou, S. Sharma, A. Kumar, A. H. Al-Muhtaseb, M. Naushad and F. J. Stadler, Catal. Today, 335, 437 (2019).
R. Nagaraja, N. Kottam, C. R. Girija and B. M. Nagabhushana, Powder Technol., 215–216, 91 (2012).
M. Z. Bin Mukhlish, F. Najnin, M. M. Rahman and M. J. Uddin, J. Sci. Res., 5, 301 (2013).
K. Yu, S. Yang, H. He, C. Sun, C. Gu and Y. Ju, J. Phys. Chem. A, 113, 10024 (2009).
Acknowledgements
The authors (NC, PRG) would like to acknowledge the Indian National Academy of Engineering (INAE) for mentorship program of engineering faculty. The authors would like to thank Manipal University Jaipur (MUJ) for providing access to the Central Analytical Facilities (CAF) and Sophisticated Analytical Instrumentation Facility (SAIF). One of the authors (AGC) would also like to acknowledge the endowment seed grant (EF/2017-18/QE04-06) provided by the University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Supporting Information
Rights and permissions
About this article
Cite this article
Chakinala, N., Gogate, P.R. & Chakinala, A.G. Highly efficient bi-metallic bismuth-silver doped TiO2 photocatalyst for dye degradation. Korean J. Chem. Eng. 38, 2468–2478 (2021). https://doi.org/10.1007/s11814-021-0890-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0890-5