Abstract
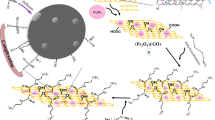
Arsanilic acid (ASA), an organic-arsenic veterinary drug used widely, has greatly attracted attention due to its potential threats. We report the nanocomposites of the α-Fe2O3 nanoparticles growth on reduced graphene oxide (rGO) by a one-pot method. The α-Fe2O3 nanoparticles are densely covered on the surface of rGO according to the observations of transmission and scanning electron microscope. The adsorptive capacity (357.4±11.2 mg g−1) of the Fe2O3@rGO nanocomposites for ASA, which was more than the sum of adsorptive capacities of the pure α-Fe2O3 nanoparticles and rGO, revealed a remarkable enhancement due to the synergetic effect of multiple interactions and the good dispersion of α-Fe2O3 nanoparticles with more active binding sites in the Fe2O3@rGO nanocomposites. The adsorption equilibrium of ASA onto the Fe2O3@rGO nanocomposites was achieved for 60 min, and the adsorption of ASA was dependent of pH and temperature, and independent of the concentration of humic acid ranging from 0 to 20 mg L−1. After five cycles of adsorption-desorption, the adsorptive amounts of ASA by the regenerative sorbent still retained 85% of adsorptive amount by the fresh sorbents. The adsorption process of ASA can be described by the Langmuir and the pseudo-second-order equations and is exothermic and spontaneous according to thermodynamic analysis.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
L. R. Overby and S. Lilian, Toxicol. Appl. Pharmacol., 7, 850 (1965).
D. W. Rutherford, A. J. Bednar, J. R. Garbarino, R. Needham, K. W. Staver and R. L. Wershaw, Environ. Sci. Technol., 37, 1515 (2003).
J. R. Garbarino, A. J. Bednar, D. W. Rutherford, R. S. Beyer and R. L. Wershaw, Environ. Sci. Technol., 37, 1509 (2003).
B. L. Brown, A. D. Slaughter and M. Schreiber, Appl. Geochem., 20, 123 (2005).
J. Xu, X. Shen, D. Wang, C. Zhao, Z. Liu, I. P. Pozdnyakov, F. Wu and J. Xia, Chem. Eng. J., 336, 334 (2018).
B. K. Jung, J. W. Jun, Z. Hasan and S. H. Jhung, Chem. Eng. J., 267, 9 (2015).
S. Chen, J. Deng, C. Ye, C. Xu, L. Huai, J. Li and X. Li, Sci. Total Environ., 742, 140587 (2020).
T. P. Joshi, G. Zhang, R. Koju, Z. Qi, R. Liu, H. Liu and J. Qu, Sci. Total Environ., 601–602, 713 (2017).
W. Liu, Z. Ai, R. A. Dahlgren, L. Zhang and X. Wang, Chem. Eng. J., 330, 1232 (2017).
W. Fan, X. Zhang, Y. Zhang, P. Wang, L. Zhang, Z. Yin, J. Yao and W. Xiang, J. Mol. Recognit., 31, e2625 (2018).
Y.-J. Wang, F. Ji, W. Wang, S.-J. Yuan and Z.-H. Hu, Desalin. Water Treatm., 57, 20520 (2016).
L. Poon, S. Younus and L. D. Wilson, J. Colloid Interface Sci., 420, 136 (2014).
J. Hu, Z. Tong, Z. Hu, G. Chen and T. Chen, J. Colloid Interface Sci., 377, 355 (2012).
D. Kong and L. D. Wilson, Carbohyd. Polym., 169, 282 (2017).
J. Hu, Z. Tong, G. Chen, X. Zhan and Z. Hu, Int. J. Environ. Sci. Technol., 11, 785 (2014).
C. Tian, J. Zhao, J. Zhang, S. Chu, Z. Dang, Z. Lin and B. Xing, Environ. Sci.: Nano, 4, 2134 (2017).
N. You, X.-F. Wang, J.-Y. Li, H.-T. Fan and Q. Zhang, J. Ind. Eng. Chem., 70, 346 (2019).
T. A. Saleh, M. M. Al-Shalalfeh and A. A. Al-Saadi, Sensors Actuat. B: Chem., 254, 1110 (2018).
L. Y. Wang, S. W. Wang and W. R. Chen, Chemosphere, 152, 423 (2016).
W. Tang, Q. Li, S. Gao and J. K. Shang, J. Hazard. Mater., 192, 131 (2011).
X. Zhao, L. Lv, B. Pan, W. Zhang, S. Zhang and Q. Zhang, Chem. Eng. J., 170, 381 (2011).
W. S. Hummers and R. E. Offeman, J. Am. Chem. Soc., 80, 1339 (1958).
N. You, Y. Chen, Q.-X. Zhang, Y. Zhang, Z. Meng and H.-T. Fan, Sci. Total Environ., 735, 139553 (2020).
F.-F. Zhang, W. Wang, S.-J. Yuan and Z.-H. Hu, J. Hazard. Mater., 279, 562 (2014).
M. Sun, H. Liu, Y. Liu, J. Qu and J. Li, Nanoscale, 7, 1250 (2015).
K. Urbas, M. Aleksandrzak, M. Jedrzejczak, M. Jedrzejczak, R. Rakoczy, X. Chen and E. Mijowska, Nanoscale Res. Lett., 9, 656 (2014).
C. Z. Zhu, S. J. Guo, Y. X. Fang and S. J. Dong, ACS Nano, 4, 2429 (2010).
C. Wu, H. Zhang, Y. X. Wu, Q. C. Zhuang, L. L. Tian and X. X. Zhang, Electrochim. Acta, 134, 18 (2014).
B. Li, X. Zhu, K. Hu, Y. Li, J. Feng, J. Shi and J. Gu, J. Hazard. Mater., 302, 57 (2016).
I. I. Salame and T. J. Bandosz, J. Colloid Interface Sci., 240, 252 (2001).
J. Deng, Y.-J. Chen, Y.-A. Lu, X.-Y. Ma, S.-F. Feng, N. Gao and J. Li, Environ. Sci. Pollut. Res., 24, 14396 (2017).
J. W. Jun, M. Tong, B. K. Jung, Z. Hasan, C. Zhong and S. H. Jhung, Chem. Eur. J., 21, 347 (2015).
A. Adamescu, I. P. Hamilton and H. A. Al-Abadleh, J. Phys. Chem. A, 118, 5667 (2014).
T. Sun, Z. Zhao, Z. Liang, J. Liu, W. Shi and F. Cui, Chem. Eng. J., 334, 1527 (2018).
W. Mitchell, S. Goldberg and H. A. Al-Abadleh, J. Colloid Interface Sci., 358, 534 (2011).
W. R. Chen and C. H. Huang, J. Hazard. Mater., 227–228, 378 (2012).
Y. Peng, W. Wei, H. Zhou, S. Ge, S. Li, G. Wang and Y. Zhang, J. Dispers. Sci. Technol., 37, 1590 (2016).
T. P. Joshi, G. Zhang, H. Cheng, R. Liu, H. Liu and J. Qu, Water Res., 116, 126 (2017).
H.-T. Fan, J.-B. Wu, X.-L. Fan, D.-S. Zhang, Z.-J. Su, F. Yan and T. Sun, Chem. Eng. J., 198–199, 355 (2012).
H.-T. Fan, X.-T. Sun, Z.-G. Zhang and W.-X. Li, J. Chem. Eng. Data, 59, 2106 (2014).
H. Javadian, P. Vahedian and M. Toosi, Appl. Surf. Sci., 284, 13 (2013).
H.-T. Fan, Q. Tang, Y. Sun, Z.-G. Zhang and W.-X. Li, Chem. Eng. J., 258, 146 (2014).
H.-T. Fan, X.-T. Sun and W.-X. Li, J. Sol-Gel Sci. Technol., 72, 144 (2014).
Y. Liu, J. Chem. Eng. Data, 54, 1981 (2009).
H.-T. Fan, W. Sun, B. Jiang, Q.-J. Wang, D.-W. Li, C.-C. Huang, K.-J. Wang, Z.-G. Zhang and W.-X. Li, Chem. Eng. J., 286, 128 (2016).
H. Fan, X. Fan, J. Li, M. Guo, D. Zhang, F. Yan and T. Sun, Ind. Eng. Chem. Res., 51, 5216 (2012).
H.-T. Fan, Y. Sun, Q. Tang, W.-L. Li and T. Sun, J. Taiwan Inst. Chem. Eng., 45, 2640 (2014).
Acknowledgements
Financial support of this work was provided by the Science and technology development fund of Shenyang Medical College (20191026) and by scientific research foundation of education department of Liaoning province (LN201902).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Rights and permissions
About this article
Cite this article
Sui, LL., Peng, LN. & Xu, HB. Nanocomposites of Fe2O3@rGO for adsorptive removal of arsanilic acid from aqueous solution. Korean J. Chem. Eng. 38, 498–504 (2021). https://doi.org/10.1007/s11814-020-0722-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0722-z