Abstract
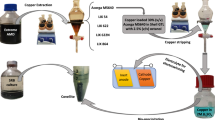
Hazardous hexavalent chromium (Cr(VI)) released from the electroplating industry beyond the discharge concentration limit has created great concern worldwide. Solvent extraction (SX) is an effective method for metal separation from an aqueous system. Throughout this work, novel synergistic solvent extraction of Cr(VI) from rinsed electroplating wastewater was performed using a mixture of two organophosphorus acidic extractants containing di-2,4,4-trimethylpentyl phosphinic acid (Cyanex 272) and di-2,4,4-trimethylpentyl mono-thio-phosphinic acid (Cyanex 302) in kerosene. The results showed almost 99±0.816% of Cr(VI) was synergistically reduced and extracted to the less toxic trivalent chromium (Cr(III)) using the total mixture concentration of 0.1 M (0.04 M Cyanex 272+0.06 M Cyanex 302). For recycling, the organic phase was recycled or reused up to 11 times for excellent extraction (99±0.471%) and stripping (99±0.942%) and became stable afterwards until a certain time. The total concentration of the mixture system was reduced by 50% (0.05 M) using 0.01 M Cyanex 272 and 0.04 M Cyanex 302, with almost 100% of Cr(VI) extraction as less toxic Cr(III). The Cr(III) in the loaded organic phase was completely recovered using 7.0 M hydrochloric acid (HCl). It is concluded that the synergistic extraction using Cyanex 272/Cyanex 302 system improved the Cr(VI) extraction and reduced the chemical consumption in the organic phase.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
S. S. Hosseini, E. Bringas, N. R. Tan, I. Ortiz, M. Ghahramani and M. A. A. Shahmirzadi, J. Water Process Eng., 9, 78 (2016).
Department of Environment. Environmental Quality Report (Industrial Effluent) Regulations. Malaysia, P.U. (A) 434 (2009).
S. S. Hosseini, A. Nazif, M. A. A. Shahmirzadi and I. Ortiz, Sep. Purif. Technol., 187, 46 (2017).
M. R. Muthumareeswaran, M. Alhoshan and G. P. Agarwal, Sci. Rep., 7, 1 (2017).
R. N. R. Sulaiman and N. Othman, Malay. J. Anal. Sci., 21, 416 (2017).
A. Kumar, A. Thakur and P. S. Panesar, J. Ind. Eng. Chem., 70, 394 (2019).
V. K. Bhosale, H. K. Chana, S. P. Kamble and P. S. Kulkarni, J. Water Process Eng., 32, 100925 (2019).
G. Dognani, P. Hadi, H. Ma, F. C. Cabrera, A. E. Job, D. L. S. Agostini and B. S. Hsiao, Chem. Eng. J., 372, 341 (2019).
J. J. Chen, A. L. Ahmad, J. K. Lim and B. S. Ooi, J. Water Process Eng., 32, 100957 (2019).
D. Wang, S. He, C. Shan, Y. Ye, H. Ma, X. Zhang, W. Zhang and B. Pan, J. Hazard. Mater., 316, 169 (2016).
M. D. Lanagan and D. C. Ibana, Miner. Eng., 16, 237 (2003).
R. N. R. Sulaiman and N. Othman, J. Environ. Chem. Eng., 6, 1814 (2018).
Y. Liu, M. Lee and G. Senanayake, J. Mol. Liq., 268, 667 (2018).
R. K. Goyal, N. S. Jayakumar and M. A. Hashim, Desalination, 278, 50 (2011).
M. Rajasimman and R. Sangeetha, J. Hazard. Mater., 168, 291 (2009).
R. T. Bachmann, D. Wiemken, A. B. Tengkiat and M. Wilichowski, Sep. Purif. Technol., 75, 303 (2010).
H. H. Someda, E. A. El-Shazly and R. R. Sheha, J. Hazard. Mater., B117, 213 (2005).
S. Kalidhasan and N. Rajesh, J. Hazard. Mater., 170, 1079 (2009).
G. Zhang, D. Chen, W. Zhao, H. Zhao, L. Wang, W. Wang and T. Qi, Chem. Eng. J., 302, 233 (2016).
V. Eyupoglu, R. A. Kumbasar and M. Isik, J. Dispersion. Sci. Technol., 36, 1309 (2015).
N. K. Batchu, H. S. Jeon and M. S. Lee, J. Ind. Eng. Chem., 26, 285 (2015).
S. Roy, S. Basu, M. Anitha and D. K. Singh, Korean J. Chem. Eng., 34, 1740 (2017).
R. N. R. Sulaiman, H. A. Rahman, N. Othman, M. B. Rosly, N. Jusoh and N. F. M. Noah, Korean J. Chem. Eng., 37, 141 (2020).
P. Zaheri, H. Abolghasemi, T. Mohammadi and M. G. Maraghe, Korean J. Chem. Eng., 32, 1642 (2015).
P. K. Saw, A. K. Prajapati and M. K. Mondal, J. Mol. Liq., 269, 101 (2018).
A. Senol, Sep. Sci. Technol., 50, 1010 (2015).
P. K. Parhi, J. Chem., 2013, 618236 (2013).
J. Rajewski and P. Religa, J. Mol. Liq., 218, 309 (2016).
H. R. Mortaheb, A. Zolfaghari, B. Mokhtarani, M. H. Amini and V. Mandanipour, J. Hazard. Mater., 177, 660 (2010).
A. L. Ahmad, M. M. H. S. Buddin, B. S. Ooi and A. Kusumastuti, J. Water Process Eng., 15, 26 (2017).
R. A. Kumbasar, Sep. Purif. Technol., 63, 592 (2008).
N. F. M. Noah, N. Jusoh, N. Othman, R. N. R. Sulaiman and N. A. M. K. Parker, J. Ind. Eng. Chem., 66, 231 (2018).
R. Sarkar, S. Ray and S. Basu, J. Chem. Bio. Phys. Sci., 4, 3156 (2014).
K. C. Sole and J. B. Hiskey, Hydrometallurgy, 37, 129 (1995).
N. E. El-Hefny and J. A. Daoud, Solvent Extr. Ion Exch., 25, 831 (2007).
V. N. H. Nguyen, T. H. Nguyen and M. S. Lee, Metals, 10, 1 (2020).
P. Zaheri, H. Abolghasemi, M. G. Maraghe and T. Mohammadi, Chem. Eng. Process., 92, 18 (2015).
Y. Liu, H. S. Jeon and M. S. Lee, Korean J. Met. Mater., 54, 592 (2016).
C. Y. Cheng, K. R. Barnard, W. Zhang and D. J. Robinson, Solvent Extr. Ion Exch., 29, 719 (2011).
H. T. Truong, M. S. Lee and S. H. Son, Metals, 7, 1 (2017).
O. Kazak, A. Tor, I. Akin and G. Arslan, J. Environ. Chem. Eng., 3, 1654 (2015).
X. Li, C. Wei, Z. Deng, M. Li, C. Li and G. Fan, Hydrometallurgy, 105, 359 (2011).
S. Yin, W. Wu, X. Bian and F. Zhang, Hydrometallurgy, 131, 133 (2013).
B. D. Pandey, G. Cote and D. Bauer, Hydrometallurgy, 40, 343 (1996).
V. Eyupoglu and R. A. Kumbasar, J. Ind. Eng. Chem., 21, 303 (2015).
R. N. R. Sulaiman and N. Othman, Chem. Eng. Process., 134, 9 (2018).
S. Begum, V. Arelli, G. R. Anupoju, S. Sundergopal, S. K. Bhargava and N. Eshtiaghi, J. Ind. Eng. Chem., 90, 190 (2020).
R. N. R. Sulaiman, N. Othman and N. A. S. Amin, Desalin Water Treat, 57, 3339 (2016).
T. Detchusananard, K. Im-orb, F. Marechal and A. Arpornwichanop, Energy, 207, 118190 (2020).
D. Pugliese, F. Bella, V. Cauda, A. Lamberti, A. Sacco, E. Tresso and S. Bianco, ACS Appl. Mater. Interfaces, 21, 11288 (2013).
B. Miccoli, V. Cauda, A. Bonanno, A. Sanginario, K. Bejtka, F. Bella, M. Fontana and D. Demarchi, Sci. Rep., 6, 29763 (2016).
F. Bella, D. Pugliese, J. R. Nair, A. Sacco, S. Bianco, C. Gerbaldi, C. Barolo and R. Bongiovanni, Phys. Chem. Chem. Phys., 15, 3706 (2013).
M. Huang, C. Hong, C. Ma, Z. Luo, S. Du and F. Yang, Sci. Rep., 10, 9129 (2020).
Acknowledgement
The grants from Universiti Teknologi Malaysia for making this research, including Professional Development Research University (PDRU) (Q. J130000.21A2.04E68), Collaborative Research Grant (CRG) (Q. J130000.2451.08G02) and Collaborative Research Grant (CRG) (Q. J130000.2451.08G03) are highly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sulaiman, R.N.R., Noah, N.F.M., Othman, N. et al. Synergetic formulation of Cyanex 272/Cyanex 302 for hexavalent chromium removal from electroplating wastewater. Korean J. Chem. Eng. 38, 514–522 (2021). https://doi.org/10.1007/s11814-020-0702-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0702-3