Abstract
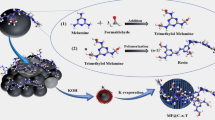
The adsorption of pure gases of carbon dioxide, methane and nitrogen was examined on nitrogen-doped porous carbon (NDC) prepared from polyaniline (PANI) as precursor by chemical activation with KOH (T=650o, IR=2, t=1 h) to determine the potential for the separation of CO2 gas from flue gas or natural gas. Adsorption equilibrium of all gases was determined in a temperature range of 298–318 K and pressure up to 14 bar. Results demonstrated an excellent CO2 adsorption capacity of 3.09 mmol·g−1 owing to high CO2 interaction affinity with NDC surface compared to CH4 (1.43 mmol·g−1) and N2 (0.64 mmol·g−1) under ambient condition (298 K and 1 bar). The ideal adsorbed solution theory (IAST) was used to determine the adsorption selectivity of NDC for CO2/CH4 and CO2/N2 mixtures at different compositions. The NDC had CO2/CH4 (4.42 and 4.08 for CO2/CH4=10/90 and 50/50, respectively) and CO2/N2 (12.81 and 12.08 for CO2/N2=15/85 and 50/50, respectively) IAST selectivity at temperature of 298 K and pressure of 1bar. The moderate CO2 adsorption enthalpy indicates that N-doped activated carbon is a promising material in gas separation such as natural gas and flue gas processing.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Y. Li, R. Xu, B. Wang, J. Wei, L. Wang, M. Shen and J. Yang, Nanomaterials, 9, 266 (2019).
A. Modak and A. Bhaumik, J. Solid State Chem., 232, 157 (2015).
S. Vasudevan, S. Farooq, I. A. Karimi, M. Saeys, M. C. Quah and R. Agrawal, Energy, 103, 709 (2016).
D. Lv, J. Chen, K. Yang, H. Wu, Y. Chen, C. Duan, Y. Wu, J. Xiao, H. Xi and Z. Li, Chem. Eng. J., 375, 122074 (2019).
S. Xian, J. Peng, Z. Zhang, Q. Xia, H. Wang and Z. Li, Chem. Eng. J., 270, 385 (2015).
H. S. Kim, M. S. Kang, S. Lee, Y.-W. Lee and W. C. Yoo, Micropor. Mesopor. Mater., 272, 92 (2018).
M. Hoorfar, Y. Alcheikhhamdon and B. Chen, Comput. Chem. Eng., 117, 11 (2018).
M. Zia-ul-Mustafa, H. Mukhtar, N. Nordin and H. Mannan, Mater. Today. Proc., 16, 1976 (2019).
C. Song, Q. Liu, S. Deng, H. Li and Y. Kitamura, Renew. Sust. Energy Rev., 101, 265 (2019).
W.-T. Zheng, K. Huang and S. Dai, Micropor. Mesopor. Mater., 290, 109653 (2019).
H.-L. Peng, J.-B. Zhang, J.-Y. Zhang, F.-Y. Zhong, P.-K. Wu, K. Huang, J.-P. Fan and F. Liu, Chem. Eng. J., 359, 1159 (2019).
B. Chang, W. Shi, H. Yin, S. Zhang and B. Yang, Chem. Eng. J., 358, 1507 (2019).
S. S. Fiyadh, M. A. AlSaadi, W. Z. Jaafar, M. K. AlOmar, S. S. Fayaed, N. S Mohd, L. S. Hin and A. El-Shafie, J. Clean. Prod., 230, 783 (2019).
M. Xu, S. Chen, D.-K. Seo and S. Deng, Chem. Eng. J., 371, 693 (2019).
S. Jr ibi, T. Miyazaki, B. B. Saha, A. Pal, M. M. Younes, S. Koyama and A. Maalej, Int. J. Heat Mass Transfer, 108, 1941 (2017).
X. Weng, Y. Cui, S. Shaikhutdinov and H.-J. Freund, J. Phys. Chem. C, 123, 1880 (2018).
G. Gómez-Pozuelo, E. Sanz-Pérez, A. Arencibia, P. Pizarro, R. Sanz and D. Serrano, Micropor. Mesopor. Mater., 282, 38 (2019).
R. Kishor and A. K. Ghoshal, Chem. Eng. J., 262, 882 (2015).
J. Wang, R. Krishna, X. Wu, Y. Sun and S. Deng, Langmuir, 31, 9845 (2015).
L. An, S. Liu, L. Wang, J. Wu, Z. Wu, C. Ma, Q. Yu and X. Hu, Ind. Eng. Chem. Res., 58, 3349 (2019).
P. Nugent, Y. Belmabkhout, S. D. Burd, A. J. Cairns, R. Luebke, K. Forrest, T. Pham, S. Ma, B. Space and L. Wojtas, Nature, 495, 80 (2013).
R. Zhong, Z. Xu, W. Bi, S. Han, X. Yu and R. Zou, Inorg. Chim. Acta, 443, 299 (2016).
M. Kacem, M. Pellerano and A. Delebarre, Fuel Process. Technol., 138, 271 (2015).
S. Khalili, B. Khoshandam and M. Jahanshahi, RSC Adv., 6, 35692 (2016).
J. Singh, S. Basu and H. Bhunia, Micropor. Mesopor. Mater., 280, 357 (2019).
A. C. Dassanayake and M. Jaroniec, Colloids Surf. A Physicochem. Eng. Asp., 549, 147 (2018).
E. Qezelsefloo, S. Khalili, M. Jahanshahi and M. Peyravi, Mater. Chem. Phys., 239, 122304 (2020).
X. Li, Z.-Y. Sui, Y.-N. Sun, P.-W. Xiao, X.-Y. Wang and B.-H. Han, Micropor. Mesopor. Mater., 257, 85 (2018).
J. Pallarés, A. González-Cencerrado and I. Arauzo, Biomass Bioenergy, 115, 64 (2018).
E. Apaydin-Varol and Y. Erülken, J. Taiwan Inst. Chem. Eng., 54, 37 (2015).
J. Singh, S. Basu and H. Bhunia, Micropor. Mesopor. Mater., 280, 357 (2019).
S. Khalili, B. Khoshandam and M. Jahanshahi, RSC Adv., 6, 35692 (2016).
P. Ammendola, F. Raganati and R. Chirone, Chem. Eng. J., 322, 302 (2017).
F. Raganati, M. Alfe, V. Gargiulo, R. Chirone and P. Ammendola, Chem. Eng. Res. Des., 134, 540 (2018).
A. L. Myers and J. M. Prausnitz, AIChE J., 11, 121 (1965).
D. D. Do, Adsorption analysis: Equilibria and kinetics (with cd containing computer MATLAB programs), World Scientific, London (1998).
V. Garshasbi, M. Jahangiri and M. Anbia, Appl. Surf. Sci., 393, 225 (2017).
S. Zohdi, M. Anbia and S. Salehi, Polyhedron, 166, 175 (2019).
A. Heidari, H. Younesi, A. Rashidi and A. A. Ghoreyshi, Chem. Eng. J., 254, 503 (2014).
Y. Huangfu, K. Ruan, H. Qiu, Y. Lu, C. Liang, J. Kong and J. Gu, Compos. Part A Appl. Sci. Manuf., 121, 265 (2019).
S. Daikh, F. Zeggai, A. Bellil and A. Benyoucef, J. Phys. Chem. Solids, 121, 78 (2018).
V. Janaki, K. Vijayaraghavan, B.-T. Oh, K.-J. Lee, K. Muthuchelian, A. Ramasamy and S. Kamala-Kannan, Carbohydr. Polym., 90, 1437 (2012).
M. A. Tofighy and T. Mohammadi, Chem. Eng. Res. Des., 90, 1815 (2012).
J. Stejskal, I. Sapurina and M. Trchová, Prog. Polym. Sci., 35, 1420 (2010).
J. Wang, R. Krishna, X. Wu, Y. Sun and S. Deng, Langmuir, 31, 9845 (2015).
J. Park, N. F. Attia, M. Jung, M. E. Lee, K. Lee, J. Chung and H. Oh, Energy, 158, 9 (2018).
W. Su, L. Yao, M. Ran, Y. Sun, J. Liu and X. Wang, J. Chem. Eng. Data, 63, 2914 (2018).
S. Y. Sawant, R. S. Somani, H. C. Bajaj and S. S. Sharma, J. Hazard. Mater., 227, 317 (2012).
P. S. Bárcia, L. Bastin, E. J. Hurtado, J. A. Silva, A. E. Rodrigues and B. Chen, Sep. Sci. Technol., 43, 3494 (2008).
L. Wang, L. Rao, B. Xia, L. Wang, L. Yue, Y. Liang, H. DaCosta and X. Hu, Carbon, 130, 31 (2018).
N. Álvarez-Gutiérrez, M. Gil, F. Rubiera and C. Pevida, Fuel Process. Technol., 142, 361 (2016).
Acknowledgement
The authors acknowledge the funding support of Babol Noshirvani University of Technology through Grant program No. BNUT/370302/98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalili, S., Jahanshahi, M. Selective CO2 adsorption using N-rich porous carbon derived from KOH-activated polyaniline. Korean J. Chem. Eng. 38, 862–871 (2021). https://doi.org/10.1007/s11814-020-0691-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0691-2