Abstract
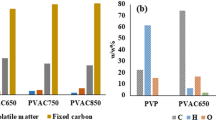
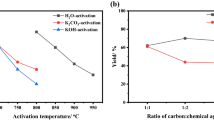
The behavior and characteristics of phenol adsorption by activated carbon produced from co-pyrolysis of coal tar pitch and vinasse were investigated. Coal tar pitch and vinasse (mass ratio of 1:3) were firstly co-pyrolyzed and carbonated at 400 °C for 2 h. The carbonized material produced was then soaked with saturated KOH solution and activated at 800 °C for 1 h to form activated carbon. Results from the phenol wastewater adsorption experiments suggest that pseudo-second-order kinetics and the Weber-Morris model could reflect the time-dependent adsorption behavior of phenol wastewater by the activated carbon, revealing that internal diffusion represents the rate-limiting procedure and dominant process to control the adsorption rate in the early stage of the adsorption. Monolayer adsorption played the key role during the phenol adsorption. The adsorption was an endothermic reaction in temperature ranging from 15 °C to 35 °C, indicating that the adsorption speed could be stimulated by the increasing temperature. This study establishes a theoretical foundation for the usage and the potential application of the activated carbon derived from coal tar pitch and vinasse in wastewater treatment.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. Olivares-Marin, V. Del Prete, E. Garcia-Moruno, C. Fernandez-Gonzalez, A. Macias-Garcia and V. Gomez-Serrano, Food Control, 20, 298 (2009).
A. Martínez de Yuso, M. Teresa Izquierdo, R. Valenciano and B. Rubio, Fuel Process. Technol, 110, 1 (2013).
R. Yavuz, H. Akyildiz, N. Karatepe and E. Etinkaya, Fuel Process. Technol., 91, 80 (2010).
F. Kaouah, S. Boumaza, T. Berrama, M. Trari and Z. Bendjama, J. Clean. Prod., 54, 296 (2013).
C. Peng, X. Yan, R. Wang, J. Lang, Y. Ou and Q. Xue, Electrochim. Acta, 87, 401 (2013).
K. Yaghmaeian, G. Moussavi and A. Alahabadi, Chem. Eng. J., 236, 538 (2014).
Q. Gao, H. Liu, C. Cheng, K. Li, J. Zhang, C. Zhang and Y. Li, Powder Technol., 249, 234 (2013).
A. Yamashita, Y. Mori, T. Oshima and Y. Baba, Carbon, 76, 53 (2014).
J. Xu, L. Chen, H. Qu, Y. Jiao, J. Xie and G. Xing, Appl. Surf. Sci., 320, 674 (2014).
S. Yorgun, N. Vural and H. Demiral, Micropor. Mesopor. Mater., 122, 189 (2009).
Y. Wang, H. Ngo and W Guo, Sci. Total Environ, 533, 32 (2015).
X. Yao, J. Liu, G. Gong, Y. Jiang and Q. Xie, Int. J. Min. Sci. Tech., 23, 395 (2013).
S. Ge, Y. Wu, W Peng, C. Xia, C. Mei, L. Cai, S. Shi, C. Sonne, S. S. Lam and Y. F. Tsang, Chem. Eng. J., 385, 123949 (2020).
S. Ge, S. Y Foong, N. L. Ma, R. K. Liew, W. A. Wan Mahari, C. Xia, P. N. Y. Yek, W. Peng, W. L. Nam, X. Y. Lim, C. M. Liew, C. C. Chong, C. Sonne and S.S. Lam, Renew. Sust. Energ. Rev., 127, 109871 (2020).
Y. Wu, C. Xia, L. Cai and S. Q. Shi, J. Colloid Interface Sci., 518, 41 (2018).
C. Xia and S. Q. Shi, Green Chem., 18, 2063 (2016).
H. Chang, W Wei, Z. Wang, H. Yang and R. Yao, Shanxi Coking Coal Sci. Tech., 2, 39 (2007).
K. S. Yang, D. J. Lee, S. K. Ryu, Y. Korai, Y. J. Kim and I. Mochida, Korean J. Chem. Eng., 16, 518 (1999).
Y. Wu, L. Cai, C. Mei, S. S. Lam, C. Sonne, S. Q. Shi and C. Xia, Mater. Today Commun., 24, 101008 (2020).
M. Ul-Islam, M. W Ullah, S. Khan and J. K Park, Korean J. Chem. Eng., 37, 925 (2020).
Y. Wu, S. Ge, C. Xia, L. Cai, C. Mei, C. Sonne, Y. Park, Y. Kim, W. Chen, J. Chang and S.S. Lam, Bioresour. Technol., 313, 123675 (2020).
Y. Ma, R. Niu, X. Wang, Q. Wang, X. Wang and X. Sun, Waste Manag. Res., 32, 1123 (2014).
H. Wang, R. Xie, J. Zhang and J. Zhao, Adv. Powder Technol., 29, 27 (2018).
X. Zhang, H. Tang, G. Chen, L. Qiao, J. Li, B. Liu, Z. Liu, M. Li and X. Liu, Eur. Food Res. Technol., 245, 2631 (2019).
K. Y. Foo and B. H. Hameed, Bioresour. Technol., 112, 143 (2012).
A. K. Prajapati and M. K. Mondal, Korean J. Chem. Eng., 36, 1900 (2019).
Y. Zhu, W. Du, Y. Su and M. Zhu, Chin. Agr. Sci. Bull., 31, 26 (2015).
B. Tsyntsarski, B. Petrova, T. Budinova, N. Petrov, D. K. Teodosiev, A. Sarbu, T. Sandu, M. Ferhat Yardim and A. Sirkecioglu, Desalin. Water Treat., 52, 3445 (2014).
M. Fan, S. Tong and C. Q. Jia, Int. J. Oil Gas Coal T., 8, 489 (2014).
X. Zhang, W Wang, S. Luo and Q. Lin, J. Colloid Interface Sci., 553, 484 (2019).
I. G. Stoycheva, B. G. Tsyntsarski, B. N. Petrova, B. Kumanek, T. K. Budinova and N. V Petrov, Water Air Soil Pollut., 227, 452 (2016).
P. Yao, J. Cen, M. Fang, T. Wang and Q. Wang, RSC Adv., 8, 17558 (2018).
G. Asgari, B. Roshani and G. Ghanizadeh, J. Hazard. Mater., 217–218, 123 (2012).
Acknowledgements
This work was supported by the Guangxi Key Research and Development Program (Guike AB18126087) and the National Key R&D Program (2018YFC1900904). The support from Sino-US-Japan Joint Laboratory on Organic Solid Waste Resource and Energy Technology of USTB is appreciated. Universiti Malaysia Terengganu under Golden Goose Research Grant Scheme (GGRG) (UMT/RMIC/2-2/25 Jld 5 (64), Vot 55191) for supporting Dr. Lam to perform this joint project.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gao, M., Wang, X., Xia, C. et al. Phenol removal via activated carbon from co-pyrolysis of waste coal tar pitch and vinasse. Korean J. Chem. Eng. 38, 64–71 (2021). https://doi.org/10.1007/s11814-020-0676-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0676-1