Abstract
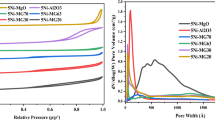
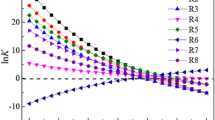
Co-based catalysts were developed for the production of high-calorie synthetic natural gas. The Co reduction in Al2O3- and SiO2-supported catalysts prepared with different Co loading, and their catalytic properties for high-calorie methanation were investigated. The CO conversion of the Co/SiO2 catalysts was superior to that of the Co/ Al2O3 with the same Co loading, due to their better reducibility at 400°C. The activities of both the Al2O3 and SiO2-supported catalysts increased with Co loading, while the growth of hydrocarbon chains decreased as the Co loading increased. As the reduction temperature increased, crystallite size of Co increased in 10Co/SiO2, resulting in decrease of CO conversion and increase of C2+ selectivity. The highest CO conversion (98.7%) was obtained over 10Co/SiO2 reduced at 400 °C. Moreover, the heating value of the product gas (10,405 kcal/Nm3) exceeded the standard heating value without requiring a high reduction temperature (700 °C) or a noble metal (Ru).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. J. Davis, K. Caldeira and H. D. Matthews, Science, 329, 1330 (2010).
S. B. Jo, H. J. Chae, T. Y. Kim, C. H. Lee, J. U. Oh, S.-H. Kang, J. W. Kim, M. Jeong, S. C. Lee and J. C. Kim, Catal. Commun., 117, 74 (2018).
Y. H. Lee, H. Kim, H. S. Choi, D.-W. Lee and K.-Y. Lee, Korean J. Chem. Eng., 32, 2220 (2015).
Y. H. Lee, D.-W. Lee and K.-Y. Lee, J. Mol. Catal. A-Chem., 425, 190 (2016).
I. Czekaj, F. Loviat, F. Raimondi, J. Wambach, S. Biollaz and A. Wokaun, Appl. Catal. A-Gen., 329, 68 (2007).
C. Guo, Y. Wu, H. Qin and J. Zhang, Fuel Process. Technol., 124, 61 (2014).
D. Hu, J. Gao, Y. Ping, L. Jia, P. Gunawan, Z. Zhong, G. Xu, F. Gu and F. Su, Ind. Eng. Chem. Res., 51, 4875 (2012).
K. B. Kester, E. Zagli and J. L. Falconer, Appl. Catal., 22, 311 (1986).
J. Sehested, S. Dahl, J. Jacobsen and J. R. Rostrup-Nielsen, J. Phys. Chem. B, 109, 2432 (2005).
J. Kopyscinski, T. J. Schildhauer and S. M. Biollaz, Fuel, 89, 1763 (2010).
S. Rönsch, J. Schneider, S. Matthischke, M. Schlüter, M. Götz, J. Lefebvre, P. Prabhakaran and S. Bajohr, Fuel, 166, 276 (2016).
B. H. Davis, Ind. Eng. Chem. Res., 46, 8938 (2007).
J. Den Breejen, P. Radstake, G. Bezemer, J. Bitter, V. Frøseth, A. Holmen and K. P. de Jong, J. Am. Chem. Soc., 131, 7197 (2009).
Z.-j. Wang, Z. Yan, C.-j. Liu and D. Goodman, ChemCatChem., 3, 551 (2011).
C. Weststrate, J. Van De Loosdrecht and J. Niemantsverdriet, J. Catal., 342, 1 (2016).
T. Inui, A. Sakamoto, T. Takeguchi and Y. Ishigaki, Ind. Eng. Chem. Res., 28, 427 (1989).
Y. Ishigaki, M. Uba, S. Nishida and T. Inui, Appl. Catal., 47, 197 (1989).
J.-H. Oh, J. W. Bae, S.-J. Park, P. Khanna and K.-W. Jun, Catal. Lett., 130, 403 (2009).
K. S. Park, K. Saravanan, S.-J. Park, Y.-J. Lee, K.-W. Jeon and J. W. Bae, Catal. Sci. Technol., 7, 4079 (2017).
R. C. Reuel and C. H. Bartholomew, J. Catal., 85, 78 (1984).
H. G. Salazar-Contreras, A. Martínez-Hernández, A. A. Boix, G. A. Fuentes and E. Torres-García, Appl. Catal. B-Environ., 244, 414 (2019).
J. Zhang, J. Chen, J. Ren and Y. Sun, Appl. Catal. A-Gen., 243, 121 (2003).
Y. Zhang, S. Nagamori, S. Hinchiranan, T. Vitidsant and N. Tsubaki, Energy Fuel, 20, 417 (2006).
G. T. Armstrong and T. L. Jobe Jr., Heating values of natural gas and its components, U.S. Department of Commerce, Washington, D.C. (1982).
H. M. Torres Galvis and K. P. de Jong, ACS Catal., 3, 2130 (2013).
W. Li, X. Nie, X. Jiang, A. Zhang, F. Ding, M. Liu, Z. Liu, X. Guo and C. Song, Appl. Catal. B-Environ., 220, 397 (2018).
S. Ullah, E. C. Lovell, R. J. Wong, T. H. Tan, J. Scott and R. Amal, ACS Sustain. Chem. Eng., 8, 5056 (2020).
W.-P. Ma, Y.-J. Ding and L.-W. Lin, Ind. Eng. Chem. Res., 43, 2391 (2004).
A. Y. Khodakov, R. Bechara and A. Griboval-Constant, Appl. Catal. A-Gen., 254, 273 (2003).
A. Martínez, C. López, F. Márquez and I. Díaz, J. Catal., 220, 486 (2003).
C. Medina, R. García, P. Reyes, J. Fierro and N. Escalona, Appl. Catal. A-Gen., 373, 71 (2010).
Acknowledgements
This work was supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) and the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (20183010032400).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Supporting Information
11814_2020_588_MOESM1_ESM.pdf
Effect of reducibility on the performance of Co-based catalysts for the production of high-calorie synthetic natural gas
Rights and permissions
About this article
Cite this article
Kim, T.Y., Jo, S.B., Lee, C.H. et al. Effect of reducibility on the performance of Co-based catalysts for the production of high-calorie synthetic natural gas. Korean J. Chem. Eng. 37, 1690–1698 (2020). https://doi.org/10.1007/s11814-020-0588-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0588-0