Abstract
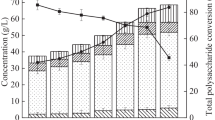
This research investigated the concept of a two-step acidic and thermal treatment for glucose extraction and levulinic acid (LA) production from food waste using dilute hydrochloric acid (DHA) as a catalyst, and subsequently analyzed the properties of the resulting humins. Glucose extraction was performed under various reaction conditions (reaction temperature range: 120-190 °C, DHA concentration range: 0.2-0.5% v/v); the glucose extraction yield of the acidic treatment step reached 83.17% under the optimal conditions (150 °C in 0.5% DHA). LA production was achieved during the thermal treatment step, which was investigated using two independent experiments to determine the influence of the reaction conditions (reaction time: 5-140min, concentration factor: 1.5-3.0, reaction temperature: 160-190 °C). The LA production process was affected by the concentration factor and the reaction temperature due to the low pH of solution and the rapid reaction rate, respectively. The thermal stability of the humins was highest at a concentration factor of 3.0 because of the 13.07 C/H ratio of the humins.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
K. D. Baugh and P. L. McCarty, Biotechnol. Bioeng., 31, 50 (1988).
K. R. Bräutigam, J. Jörissen and C. Priefer, Waste Manage. Res., 32, 683 (2014).
A. Caretto and A. Perosa, ACS Sustainable Chem. Eng., 1, 989 (2013).
F. Cherubini, Energy Convers. Manag., 51, 1412 (2010).
S. P. Das, R. Ravindran, S. Ahmed, D. Das, D. Goyal, C. M. Fontes and A. Goyal, Appl. Biochem. Biotechnol., 167, 1475 (2012).
J. Esteban and M. Ladero, Int. J. Food Sci. Technol., 53, 1095 (2018).
S. W. Fitzpatrick, ACS Symp. Ser., 921, 271 (2006).
S. S. Chen, T. Maneerung, D. C. Tsang, Y. S. Ok and C. H. Wang, Chem. Eng. J., 328, 246 (2017).
B. Girisuta, L.P. B. H. Janssen and H. J. Heeres, Chem. Eng. Res. Des., 84, 339 (2006).
M. Goto, R. Obuchi, T. Hirose, T. Sakaki and M. Shibata, Bioresour. Technol., 93, 279 (2004).
D. J. Hayes, S. Fitzpatrick, M. H. Hayes and J. R. Ross, Biorefineries: Ind. Processes Prod., 1, 139 (2006).
J. Heltzel, S. K. Patil and C. R. Lund, Humin formation pathways, in Reaction pathways and mechanisms in thermocatalytic biomass conversion II, Springer, Singapore, 105 (2016).
J. Horvat, B. Klaic, B. Metelko and V. Šunjic, Croat. Chemica. Acta, 59, 429 (1986).
I. T. Horváth, H. Mehdi, V. Fábos, L. Boda and L. T. Mika, Green Chem., 10, 238 (2008).
H. Jeong, S. K. Jang, C. Y. Hong, S. H. Kim, S. Y. Lee, S. M. Lee and I. G. Choi, Bioresour. Technol., 225, 183 (2017).
H. Ji, C. Dong, G. Yang and Z. Pang, BioResources, 14, 725 (2019).
S. J. Kim, H. S. Kwon, G. H. Kim and B. H. Um, Ind. Crops Prod., 67, 395 (2015).
Y. S. Kim, J. Y. Jang, S. J. Park and B. H. Um, Waste Manage., 74, 231 (2018).
X. Li, R. Xu, J. Yang, S. Nie, D. Liu, Y. Liu and C. Si, Ind. Crops Prod., 130, 184 (2019).
M. R. Park, H. S. Kim, S. K. Kim and G. T. Jeong, Fuel Process. Technol., 172, 115 (2018).
S. K. Patil and C. R. Lund, Energy Fuels, 25, 4745 (2011).
S. K. Patil, J. Heltzel and C. R. Lund, Energy Fuels, 26, 5281 (2012).
F. D. Pileidis and M. M. Titirici, ChemSusChem, 9, 562 (2016).
D. W. Rackemann and W. O. Doherty, Biofuels, Bioprod. Biorefin., 5, 198 (2011).
D. W. Rackemann, J. P. Bartley and W. O. Doherty, Ind. Crops Prod., 52, 46 (2014).
C. Gong, J. Wei, X. Tang, X. Zeng, Y. Sun and L. Lin, Korean J. Chem. Eng., 36, 740 (2019).
H. Rasmussen, H. R. Sørensen and A. S. Meyer, Carbohydr. Res., 385, 45 (2014).
R. Ravindran and A. K. Jaiswal, Trends Biotechnol., 34, 58 (2016).
T. H. Kim, Y. J. Jeong, K. K. Oh and T. H. Kim, Korean J. Chem. Eng., 30, 1339 (2013).
A. Sluiter, B. Hames, R. Ruiz, C. Scarlata, J. Sluiter and D. Templeton, NREL/TP-510-42623, National Renewable Energy Lab, Golden, CO, USA (2006).
A. Sluiter, R. Ruiz, C. Scarlata, J. Sluiter and D. Templeton, NREL/TP-510-42619, National Renewable Energy Lab, Golden, CO, USA (2008).
A. Sluiter, B. Hames, R. Ruiz, C. Scarlata, J. Sluiter and D. Templeton, NREL/TP-510-42622, National Renewable Energy Lab, Golden, CO, USA (2008).
A. Sluiter, B. Hames, R. Ruiz, C. Scarlata, J. Sluiter, D. Templeton and D. Crocker, NREL/TP-510-42618, National Renewable Energy Lab, Golden, CO, USA (2010).
I. V. Sumerskii, S. M. Krutov and M. Y. Zarubin, Russ. J. Appl. Chem., 83, 320 (2010).
J. Trivedi, A. K. Bhonsle and N. Atray, Academic Press., 19, 427 (2020).
S. Tulaphol, M. A. Hossain, M. S. Rahaman, L. Y. Liu, T. K. Phung, S. Renneckar and N. Sathitsuksanoh, Energy Fuels, 34, 1764 (2019).
G. Tsilomelekis, M. J. Orella, Z. Lin, Z. Cheng, W. Zheng, V. Nikolakis and D. G. Vlachos, Green Chem., 18, 1983 (2016).
B. H. Um, M. N. Karim and L. L. Henk, Appl. Biochem. Biotechnol., 105, 115 (2003).
B. H. Um and G. P. van Walsum, Appl. Biochem. Biotechnol., 168, 406 (2012).
R. J. Van Putten, J. C. Van Der Waal, E. D. De Jong, C. B. Rasrendra, H. J. Heeres and J. G. de Vries, Chem. Rev., 113, 1499 (2013).
R. Weingarten, J. Cho, W. C. Conner Jr. and G. W. Huber, Green Chem., 12, 1423 (2010).
R. Weingarten, J. Cho, R. Xing, W. C. Conner and G. W. Huber, ChemSusChem., 5, 1280 (2012).
R. Weingarten, W. C. Conner and G. W. Huber, Energy Environ. Sci., 5, 7559 (2012).
T. Werpy and G. Petersen, NREL/TP-510-35523, National Renewable Energy Lab, Golden, CO, USA (2004).
R. Xing, W. Qi and G. W. Huber, Energy Environ. Sci., 4, 2193 (2011).
K. Yan, C. Jarvis, J. Gu and Y. Yan, Renew. Sust. Energy Rev., 51, 986 (2015).
Z. Yang, H. Kang, Y. Guo, G. Zhuang, Z. Bai, H. Zhang and Y. Dong, Ind. Crops Prod., 46, 205 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Rights and permissions
About this article
Cite this article
Cha, J.S., Um, B.H. Levulinic acid production through two-step acidic and thermal treatment of food waste using dilute hydrochloric acid. Korean J. Chem. Eng. 37, 1149–1156 (2020). https://doi.org/10.1007/s11814-020-0521-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-020-0521-6