Abstract
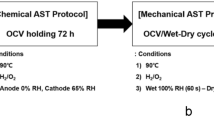
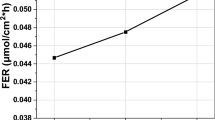
An acceleration stress test (AST) was performed to evaluate the durability of a polymer membrane in a polymer electrolyte membrane fuel cell (PEMFC) for 500 hours. Previous studies have shown that hydrogen crossover measured by linear sweep voltammetry (LSV) increases when the polymer membrane deteriorates in the AST process. On the other hand, hydrogen crossover of the membrane often decreases in the early stages of the AST test. To investigate the cause of this phenomenon, we analyzed the MEA operated for 50 hours using the AST method (OCV, RH 30% and 90 oC). Cyclic voltammetry and transmission electron showed that the electrochemical surface area (ECSA) decreased due to the growth of electrode catalyst particles and that the hydrogen crossover current density measured by LSV could be reduced. Fourier transform infrared spectroscopy and thermogravimetric/differential thermal analysis showed that -S-O-S- crosslinking occurred in the polymer after the 50 hour AST. Gas chromatography showed that the hydrogen permeability was decreased by -S-O-S- crosslinking. The reduction of the hydrogen crossover current density measured by LSV in the early stages of AST could be caused by both reduction of the electrochemical surface area of the electrode catalyst and -S-O-S- crosslinking.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. L. Perry and T. F. Fuller, J. Electrochem. Soc., 149(7), S59 (2002).
J. Kurtz, H. Dinh, G. Saur and C. Ainscough, DOE 2017 Annual Merit Review, Washington, DC, June 8 (2017).
M. P. Rodgers, L. J. Bonville, H. R. Kunz, D. K. Slattery and J. M. Fenton, Chem. Rev., 112, 6075 (2012).
D. P. Wilkinson and J. St-Pierre, Handbook of Fuel Cell: Fundamentals Technology and Applications, Vol. 3, Wiley, Chichester, England, 611 (2003).
S. D. Knights, K. M. Colbow, J. St-Pierre and D. P. Wilkinson, J. Power Sources, 127, 127 (2004).
Z. Luo, D. Li, H. Tang, M. Pan and R. Ruan, Int. J. Hydrogen Energy, 31, 1838 (2006).
A. Pozio, R. F. Silva, M. D. Francesco and L. Giorgi, Electrochim. Acta, 48, 1543 (2003).
S. Chen, H. A. Gasteiger, K. Hayakawa, T. Tada and Y. Shao-Horn, J. Electrochem. Soc., 157, A82 (2010).
D. E. Curtin, R. D. Lousenberg, T. J. Henry, P. C. Tangeman and M. E. Tisack, J. Power Sources, 131, 41 (2004).
A. Collier, H. Wang, X. Yaun, J. Zhang and D. P. Wilison, Int. J. Hydrogen Energy, 31, 1838 (2006).
DOE Fuel Cell Technologies Office, 2016 Multi-Year Research, Development and Demonstration Plan, Protocols for Testing PEM Fuel Cells and Fuel Cell Components, Page 3. 4–46 (2016).
H. Wang, M. Tang and D. Pan, Int. J. Hydrogen Energy, 33(9), 2283 (2008).
T. Kinumoto, M. Inaba, Y. Nakayama, K. Ogata, R. Umebayashi and A. Takaka, J. Power Sources, 158(2), 1222 (2006).
J. Healy, C. Hayden, T. Xie, K. Olson, R. Waldo and M. Brundage, Fuel Cells, 5(2), 302 (2005).
B. P. Pearman, N. Mohajeri, D. K. Slattery, M. D. Hampton, S. Seal and D. A. Cullen, Polym. Degrad. Stab., 98(9), 1766 (2013).
J. Hao, Y. Jiang, X. Gao, F. Xie, Z. Shao and B. Yi, J. Membr. Sci., 522(15), 23 (2017).
H. Zhu, S. Pei, J. Tang, H. Li, L. Wang, W. Yuan and Y. Zhang, J. Membr. Sci., 432, 66 (2013).
Z. Chang, H. Yan, J. Tian, H. Pan and H. Pu, Polym. Degrad. Stab., 138, 98 (2017).
W. Liu, K. Ruth and G. Rusch, J. New Mater. Mater. Electrochem. Syst., 4, 227 (2001).
B. Kieitz, J. Kolde, S. Priester, C. Baczkwski and M. Crum, ECS Trans., 41(1), 1521 (2011).
J. J. Jeong, J. H. Jeong, S. H. Kim, B. K. Ahn, J. J. Ko and K. P. Park, Korean Chem. Eng. Res., 52(4), 425 (2014).
J. Qiao, M. Saito, K. Hayamizu and T. Okada, J. Electrochem. Soc., 153(6), A967 (2006).
E. Endoh, S. Terazono, H. Widjaja and Y. Takimoto, Electrochem., Solid-State Lett., 7(7), A209 (2004).
J. H. Song, S. H. Kim, B. K. Ahn, J. J. Ko and K. P. Park, Korean Chem. Eng. Res., 51(1), 68 (2013).
Z. Liang, W. Chen, J. Liu, S. Wang, Z. Zhou, W. Li, G. Sun and Q. Xin, J. Membr. Sci., 233, 39 (2004).
M. Ludvigsson, J. Lindgren and J. Tegenfeldt, Electrochim. Acta, 45(14), 2267 (2000).
F. D. Cons, ECS Trans., 16(2), 235 (2008).
M. Danilczuk, F. D. Cons and S. Schlick, J. Phys. Chem., B, 113, 8031 (2009).
E. Endoh, S. Terazono, H. Widjaja and Y. Takimoto, Electrochem., Solid-State Lett., 7, 145 (2004).
N. Ohguri, A. Y. Nosaka and Y. Nosaka, J. Power Sources, 195, 4647 (2010).
W. Liu and D. Zuckerbrod, J. Electrochem. Soc., 152, A1165 (2005).
S. Kundu, M. W. Fowler, L. C. Simon, R. Abouatallah and N. Beydokhti, J. Power Sources, 183, 619 (2008).
L. Zhang and S. Mukerjee, J. Electrochem. Soc., 153, A1062 (2006).
S. R. Samms, S. Wasmus and R. F. Savinell, J. Electrochem. Soc., 143(5), 1498 (1996).
S. H. Almeida and Y. Kawano, J. Therm. Anal. Calorim., 58, 569 (1999).
H. J. Lee, M. K. Cho and Y. Y. Jo, Polym. Degrad. Stab., 97, 1010 (2012).
Q. Deng, C. A. Wilkie, R. B. Moore and K. A. Mauritz, J. Appl. Polym. Sci., 68, 747 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, B.C., Oh, S.H., Lee, M.S. et al. Decrease in hydrogen crossover through membrane of polymer electrolyte membrane fuel cells at the initial stages of an acceleration stress test. Korean J. Chem. Eng. 35, 2290–2295 (2018). https://doi.org/10.1007/s11814-018-0142-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-018-0142-5