Abstract
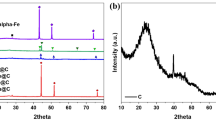
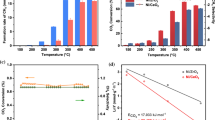
Methane activation through oxychlorination is in the spotlight due to the relatively mild reaction conditions at atmospheric pressure and in the temperature range of 450–550 °C. Although CO2 is known to exhibit good activity for methane oxychlorination, significant amounts of by-products such as CO2, CO and carbon deposits are produced during the reaction over CO2. We investigated the effect of iron in FeOx/CO2 catalysts on methane oxychlorination. FeOx/CO2 with 3 wt% iron shows the maximum yield at 510 °C with 23% conversion of methane and 65% selectivity of chloromethane. XRD and H2 TPR results indicate that iron-cerium solid solution was formed, resulting in the production of more easily reduced cerium oxide and the suppression of catalysts sintering during the reaction. Furthermore, the selectivity of by-products decreased more significantly over FeOx/CO2 than cerium oxide, which can be attributed to the facilitation of HCl oxidation arising from the enhanced reducibility of the former sample.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
V. Paunović, G. Zichittella, M. Moser, A. P. Amrute and J. Pérez-Ramírez, Nat. Chem., (8, 803 (2016).
R. Horn and R. Schlögl, Catal. Lett., (145, 23 (2015).
P. Schwach, X. Pan and X. Bao, Chem. Rev., (117, 8497 (2017).
E. Peringer, S. G. Podkolzin, M. E. Jones, R. Olindo and J. A. Lercher, Top. Catal., (38, 211 (2006).
Z. Li, G. Zhou, C. Li and T. Cheng, Catal. Commun., (40, 42 (2013).
C. Li, G. Zhou, L. Wang, Z. Li, Y. Xue and T. Cheng, Catal. Commun., (13, 22 (2011).
N. B. Muddada, T. Fuglerud, C. Lamberti and U. Olsbye, Top. Catal., (57, 741 (2014).
V. Paunović, G. Zichittella, S. Mitchell, R. Hauert and J. Pérez-Ramírez, ACS Catal., 8, 291 (2017).
V. Paunović, G. Zichittella, R. Verel, A. P. Amrute and J. Pérez-Ramírez, Angew. Chem. Int. Ed., (55, 15619 (2016).
V. Paunović, M. Artusi, R. Verel, F. Krumeich, R. Hauert and J. Pérez-Ramírez, J. Catal., 363, 69 (2018).
L. Xueju, L. Jie, Z. Guangdong, Z. Kaiji, L. Wenxing and C. Tiexin, Catal. Lett., (100, 153 (2005).
W. Taifan and J. Baltrusaitis, Appl. Catal. B Environ., (198, 525 (2016).
R. Lin, A. Amrute and J. Pérez-Ramírez, Chem. Rev., (117, 4182 (2017).
B. Wang, S. Albarracín-Suazo, Y. Pagán-Torres and E. Nikolla, Catal. Today, (285, 147 (2017).
R. Lin, Y. Ding, L. Gong, J. Li, W. Chen, L. Yan and Y. Lu, Appl. Catal. A Gen., (353, 87 (2009).
V. Paunović, R. Lin, M. Scharfe, A. P. Amrute, S. Mitchell, R. Hauert and J. Pérez-Ramírez, Angew. Chem. Int. Ed., (56, 9923 (2017).
G. Zichittella, V. Paunović, A. P. Amrute and J. Pérez-Ramírez, ACS Catal., 7, 1805 (2017).
J. He, T. Xu, Z. Wang, Q. Zhang, W. Deng and Y. Wang, Angew. Chem. Int. Ed., (51, 2438 (2012).
A. P. Amrute, C. Mondelli, M. Moser, G. Novell-Leruth, N. López, D. Rosenthal, R. Farra, M. E. Schuster, D. Teschner and T. Schmidt, J. Catal., 286, 287 (2012).
A. P. Amrute, C. Mondelli, M. A. G. Hevia and J. Pérez-Ramírez, ACS Catal., 1, 583 (2011).
M. Capdevila-Cortada, G. Vilé, D. Teschner, J. Pérez-Ramírez and N. López, Appl. Catal. B Environ., (197, 299 (2016).
C. Li, Y. Sun, I. Djerdj, P. Voepel, C.-C. Sack, T. Weller, R. d. Ellinghaus, J. Sann, Y. Guo and B. M. Smarsly, ACS Catal., 7, 6453 (2017).
W. Wang, Q. Zhu, F. Qin, Q. Dai and X. Wang, Chem. Eng. J., (333, 226 (2018).
A. S. Reddy, C.-Y. Chen, C.-C. Chen, S.-H. Chien, C.-J. Lin, K.-H. Lin, C.-L. Chen and S.-C. Chang, J. Mol. Catal. A Chem., 318, 60 (2010).
S. G. Podkolzin, E. E. Stangland, M. E. Jones, E. Peringer and J. A. Lercher, J. Am. Chem. Soc., 129, 2569 (2007).
Y. Jiang, C. Bao, Q. Liu, G. Liang, M. Lu and S. Ma, Catal. Commun., (103, 96 (2018).
T. Yamashita and P. Hayes, Appl. Surf. Sci., (254, 2441 (2008).
P. Mills and J. Sullivan, J. Phys. D Appl. Phys., 16, 723 (1983).
P. C. Graat and M. A. Somers, Appl. Surf. Sci., (100, 36 (1996).
S. Roosendaal, B. Van Asselen, J. Elsenaar, A. Vredenberg and F. Habraken, Surf. Sci., (442, 329 (1999).
Thermo Fisher Scientific Inc., https://doi.org/xpssimplified.com/elements/iron,php (accessed 22 January 2018).
M. F. Mohamad, A. Ramli and S. Yusup, AIP Conf. Proc., 1502, 288 (2012).
Z. Cui, J. Fan, H. Duan, J. Zhang, Y. Xue and Y. Tan, Korean J. Chem. Eng., (34, 29 (2017).
F. Pérez-Alonso, M. López Granados, M. Ojeda, P. Terreros, S. Rojas, T. Herranz, J. Fierro, M. Gracia and J. Gancedo, Chem. Mater., (17, 2329 (2005).
Y. Li, B. Zhang, X. Tang, Y. Xu and W. Shen, Catal. Commun., (7, 380 (2006).
A. Trovarelli, Commun. Inorg. Chem., (20, 263 (1999).
L. Kongzhai, W. Hua, W. Yonggang and L. Mingchun, J. Rare Earths, 26, 245 (2008).
W. Cai, F. Chen, X. Shen, L. Chen and J. Zhang, Appl. Catal. B Environ., (101, 160 (2010).
L. Tang, D. Yamaguchi, N. Burke, D. Trimm and K. Chiang, Catal. Commun., (11, 1215 (2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J., Ryou, Y., Hwang, G. et al. Oxychlorination of methane over FeOx/CeO2 catalysts. Korean J. Chem. Eng. 35, 2185–2190 (2018). https://doi.org/10.1007/s11814-018-0135-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-018-0135-4